An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List


Treatment of Anorexia Nervosa—New Evidence-Based Guidelines
Gaby resmark.
1 Department of Psychosomatic Medicine and Psychotherapy, University Hospital Tuebingen, Osianderstr. 5, 72076 Tuebingen, Baden-Wuerttemberg, Germany
Stephan Herpertz
2 Department of Psychosomatic Medicine and Psychotherapy, LWL University Hospital, Ruhr-University Bochum, Alexandrinenstr. 1-3, 55791 Bochum, Nordrhein-Westfalen, Germany; [email protected]
Beate Herpertz-Dahlmann
3 Department of Child and Adolescent Psychiatry, Psychosomatics and Psychotherapy, University Hospital of the RWTH Aachen, Neuenhofer Weg 21, 52074 Aachen, Nordrhein-Westfalen, Germany; ed.nehcaaku@ztreprehb
Almut Zeeck
4 Department of Psychosomatic Medicine and Psychotherapy, University Hospital Freiburg, Hauptstr. 8, 79104 Freiburg, Baden-Wuerttemberg, Germany; [email protected]
Anorexia nervosa is the most severe eating disorder; it has a protracted course of illness and the highest mortality rate among all psychiatric illnesses. It is characterised by a restriction of energy intake followed by substantial weight loss, which can culminate in cachexia and related medical consequences. Anorexia nervosa is associated with high personal and economic costs for sufferers, their relatives and society. Evidence-based practice guidelines aim to support all groups involved in the care of patients with anorexia nervosa by providing them with scientifically sound recommendations regarding diagnosis and treatment. The German S3-guideline for eating disorders has been recently revised. In this paper, the new guideline is presented and changes, in comparison with the original guideline published in 2011, are discussed. Further, the German guideline is compared to current international evidence-based guidelines for eating disorders. Many of the treatment recommendations made in the revised German guideline are consistent with existing international treatment guidelines. Although the available evidence has significantly improved in quality and amount since the original German guideline publication in 2011, further research investigating eating disorders in general, and specifically anorexia nervosa, is still needed.
1. Introduction
1.1. anorexia nervosa.
Anorexia nervosa (AN) is a serious illness leading to high morbidity and mortality [ 1 , 2 , 3 , 4 ]. It is characterised by a restriction of energy intake, weight loss, fear of weight gain and distorted body image. According to the diagnostic criteria of the International Classification of Diseases, 11th Revision (ICD-11) [ 5 ] and the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5) [ 6 ], the resulting malnutrition and low body weight may result in massive impairment to health. Often it takes years for patients with AN to achieve a first remission or to recover permanently. A quarter of adult patients go on to develop an enduring form of the disorder, and one-third of patients continue to suffer from residual symptoms in the long-term. The long-term outcome of adolescent-onset AN is more favourable [ 7 ]. Because of its severe and protracted course, AN represents a high emotional and economic burden for sufferers, carers and the society in general [ 8 , 9 ]. Age of onset peaks in middle to late adolescence, which affects educational and professional development. The consequences of starvation can have a negative impact on bone density, growth, and brain maturation, especially in children and adolescents. Many patients are affected by comorbid psychological diseases, such as depression, anxiety or obsessive–compulsive disorder. Additionally, the ego-syntonic nature of AN leads to a strong ambivalence regarding weight gain and recovery, which complicates and often slows down the recovery process. In light of these factors, treatment of AN remains challenging. To improve patients’ chances of recovery, all individuals dealing with this illness should be well informed about the nature and challenges of treating AN.
1.2. Evidence-Based Treatment Guidelines for Eating Disorders
Evidence-based guidelines have been developed in several countries around the world to guide the treatment of different eating disorders, such as AN. These guidelines have the following aims [ 10 ]:
- To support all professionals involved in the diagnosis and treatment of eating disorders, as well as sufferers and their relatives, in deciding on adequate measures of care (prevention, diagnosis, therapy and aftercare);
- To improve health care outcomes;
- To minimise risks;
- To increase treatment safety and efficiency;
- To avoid non-indicated diagnostic and treatment methods.
Further, guidelines can reveal gaps in the health care system [ 11 ] and inspire new paths of research.
Treatment guidelines provide recommendations based on current scientific evidence. In cases where a lack of scientific evidence is available, recommendations are often provided based on expert opinion, influenced by years of clinical experience.
2. The German S3Guideline for the Diagnosis and Treatment of Eating Disorders
2.1. historical development of the s3-guideline.
In 2000, the German Society for Psychiatry, Psychotherapy and Psychosomatics (DGPPN) published a guideline for the diagnosis and treatment of eating disorders in Germany for the first time [ 12 ]. In the same year, a guideline of the German Society of Child and Adolescent Psychiatry, Psychosomatics and Psychotherapy (DGKJP) was also published [ 13 ]. Both guidelines were developed by expert groups using informal consensus (a representative group of experts from the relevant medical society prepares a recommendation which is adopted by the board of the society, development stage one) with the aim of developing recommendations for the diagnosis and treatment of eating disorders. In the autumn of 2003, a conference of members of the German Society for Psychosomatic Medicine and Medical Psychotherapy (DGPM) and the German College of Psychosomatic Medicine (DKPM) decided to develop an evidence-based guideline for eating disorders in Germany according to development stage three (S3, based on all elements of systematic development—logic, decision and outcome analysis, evaluation of the clinical relevance of scientific studies and periodic review).
One year later, in the spring of 2004, a group composed of psychiatrists, child and adolescent psychiatrists, medical specialists in psychosomatic medicine and psychologists with expertise in eating disorders, was formed. The group included representatives of the five professional societies (DGKJP, the German Psychological Society (DGPs), DGPM, DGPPN and DKPM) that are responsible for the care of patients with eating disorders within the German health care system. In 2010, the evidence-based guideline for the diagnosis and treatment of eating disorders was published online by the Association of the Scientific Medical Societies in Germany (AWMF) [ 14 ]. The AWMF advises on matters and tasks of fundamental and interdisciplinary interest in medicine and provides, among other things, a wide range of clinical practice guidelines on its website. The AWMF is the national member for Germany in the Council for International Organisations of Medical Sciences (CIOMS) at the World Health Organisation, Geneva. In 2011, the guideline was made available in book format [ 15 ]. Based on the scientific guideline, a patient guideline was published in 2015 [ 16 ]; this guideline, supported by the German Society for Eating Disorders (DGESS), was designed to communicate the content of the scientific guideline to patients and relatives. The patient guideline, available both online [ 16 ] and in book format [ 17 ], addresses care structures and supports communication with professional health care providers, such as the family doctor, medical or psychological psychotherapists for adults or child and adolescent psychiatrists and psychotherapists.
Over the last two years, the scientific guideline has been revised, and a second edition will be available in German at the beginning of 2019, both online [ 14 ] and in book format. An English version of the guideline is currently in preparation and will be released at a later date. The scientific guideline addresses all age groups and is available in both a short and an extended version. The thematic structure of the recent guideline largely corresponds to the first edition and includes chapters covering epidemiology, diagnostics, the therapeutic relationship, AN, Bulimia nervosa (BN), Binge eating disorder (BED), physical sequelae and methodology. The chapter ‘Diagnostics’ is subdivided into sections on the diagnostics of psychological and somatic symptoms. In line with DSM-5 [ 6 ], two new categories of eating disorders have been added to the revised guideline: the ‘Other Specified Feeding or Eating Disorders’ (OSFED), which also include the ‘Night Eating Syndrome’, and the ‘Avoidant Restrictive Food Intake Disorder’ (ARFID), which replaces the old category of ‘Eating Disorders Not Otherwise Specified’ (EDNOS). With regard to the therapeutic studies on AN [ 18 ], BN [ 19 ] and BED [ 20 ], meta-analyses were performed based on a systematic literature search and assignment of pre-determined quality indicators (evidence level I).
2.2. Recommendation for AN—Differences between the First Version and the Revision
Changes in treatment recommendations were based on a systematic literature search (2008–2017), in which 26 new randomised controlled trials (RCTs) on psychotherapeutic treatments, 13 new RCTs on pharmacotherapy and 2 new RCTs on nutritional management were identified [ 14 , 18 ]. The evidence base has considerably improved since the first version, although studies still show a large heterogeneity in terms of samples (adolescents, adults, severe and enduring AN), setting (outpatient, day hospital, inpatient), treatment phase (acute, maintenance) and outcome measures used. It should be emphasised that an improvement in study quality can be seen. In recent years, for example, studies have been published with sample sizes that allow sufficient statistical power [ 21 , 22 , 23 , 24 , 25 , 26 ].
Treatment recommendations were based on a network-meta-analysis (see Section 2.3 ), newly published RCTs, systematic reviews, or lower levels of evidence (if RCTs or systematic reviews were not available). The guideline group discussed each recommendation in light of the available evidence, clinical relevance and suitability. The most relevant changes in the revised version concerning evidence levels and recommendations are summarised in Table 1 . Evidence levels were assigned using the Oxford Centre of Evidence Based Medicine criteria [ 27 ]: An evidence level of I is given if there is evidence for a specific treatment based on a systematic review (or meta-analysis) of randomised controlled trials (Ia), or one randomised controlled trial with narrow confidence interval (Ib). An evidence level of II is based on cohort-studies (IIa: systematic review, or IIb: individual cohort study). Evidence level III refers to case-control studies and evidence level IV to case-control series.
German guideline—changes in treatment recommendations for AN.
| Original guideline recommendations 2010 [ ] | Guideline-revision recommendations 2019 [ ] |
|---|---|
| General recommendations | |
| No recommendation concerning co-morbid conditions | (Evidence level IV; Clinical consensus point: good clinical practice): Co-morbid conditions should be systematically assessed and taken into consideration when treating patients with AN. |
| Treatment setting | |
| (Evidence level IV; 0): Inpatient treatment should take place in facilities able to offer a specialised multimodal treatment program. | (Evidence level IV; A): Same recommendation as original guideline, recommendation grading updated to A. |
| No recommendation concerning a stabilisation phase | (Evidence level IV; B): In order toreduce the probability of relapse, the final stage of inpatient therapy should aim to ensure that patients at least maintain their weight for a certain period and are prepared for the transition to an outpatient setting. |
| No specific recommendation concerning day hospital treatment for children and adolescents | (Evidence level Ib; A): A transfer to day hospital treatment after short-term inpatient treatment with sufficient physical stabilisation should be considered for children and adolescents, provided eating disorder-specific day hospital treatment can be carried out by the same treatment team, and close involvement of the relatives is ensured (evidence level Ib; A). |
| Psychotherapy | |
| (Evidence level II; B): Patients with AN are highly ambivalent towards change. Addressing ambivalence and motivation to change is a central task and should be maintained throughout the whole treatment process. | (Evidence level Ia; A): Same recommendation as original guideline, recommendation grading updated to A. |
| (Evidence level II; B): The outpatient treatment of first choice for AN should be evidence-based psychotherapy. (Clinical consensus point: good clinical practice): Patients with AN should be offered specialised therapy by a practitioner experienced with eating disorders. The choice of method should take into account the patient’s preference and age. | (Evidence level Ib; B): Outpatient treatment of first choice for patients with AN should be evidence-based psychotherapy (FBT for children and adolescents; FPT, CBT-E, MANTRA or SSCM for adults), administered by practitioners experienced with eating disorders. |
| Nutritional management | |
| (Evidence level: not rated; statement): For orientation during the first days of treatment, the initial food intake (for enteral nutrition) of highly underweight patients can be quantified at approx. 30–40 kcal/kg. | (Evidence level IIa; statement): In patients with mild to moderate AN, an initial low caloric energy supply with gradual increase is not required for safe weight gain (avoidance of refeeding syndrome)—provided that medical monitoring is ensured. |
| No recommendation, but formulation of statements. For example, The basal metabolic rate is initially low and increases significantly with the onset of weight gain. The formulas for calculating basal metabolic rate obtained from normal and overweight people are not suitable for use with AN. | (Evidence level IV; Clinical consensus point: good clinical practice ): The energy supply for the expected weight gain is highly variable and should be individually tailored to the patients as well as to the treatment phase and be continuously monitored. |
| Pharmacotherapy | |
| (Evidence level Ib; B): Neuroleptics are not suitable for achieving weight gain in AN. (Evidence level Ia; A): Antidepressants are not recommended for achieving weight gain in AN. This applies to both initial therapy and relapse prevention. | (Evidence level Ia; A): Same recommendations as original guideline, recommendation grading regarding neuroleptics updated to A. |
| (Evidence level IIa; B): If thinking is considerably restricted to weight phobia and eating and if hyperactivity is not controllable, an attempt to use low-dose neuroleptics (especially olanzapine) may be justified in individual cases. The indication for treatment should be limited to the duration of the symptoms mentioned above (no long-term treatment) and should only be applied within the framework of an overall treatment plan. | Same recommendation as original guideline, with altered recommendation levels: (Evidence level IIa; downgrading to 0): If thinking is considerably restricted to weight phobia and eating and if hyperactivity is not controllable, an attempt to use low-dose neuroleptics (especially olanzapine) may be justified in individual cases. (Clinical consensus point: good clinical practice): The indication for treatment should be limited to the duration of the symptoms mentioned above (no long-term treatment) and should only be applied within the framework of an overall treatment plan. The patient must be informed about the circumstances of the off-label use. |
FBT, Family-Based Treatment; FPT, Focal Psychodynamic Therapy; CBT-E, Enhanced Cognitive Behaviour Therapy; MANTRA, Maudsley Model of Anorexia Nervosa Treatment for Adults; SSCM, Specialist Supportive Clinical Management.
Treatment recommendations in the German treatment guideline were graded according to levels ‘A’, ‘B’, ‘0’ and ‘KKP’ [ 28 ]. ‘A’ is the strongest recommendation, which is usually based on evidence level I (something ‘is to be done’). ‘B’ recommendations are less strong (something ‘should be done’; evidence level II) and ‘0’ recommendations are even less explicit (something ‘may be done’). ‘KKP’ (‘clinical consensus point’) stands for recommendations, which are not based on empirical research and were derived from the experience of experts (good clinical practice). Grading of recommendations was based largely on the evidence level, but also took the following criteria into account: clinical relevance of effect sizes and end points, the balance of benefits and risks, ethical considerations, patient preferences and applicability. Grading of recommendations was discussed in several consensus meetings.
Several key treatment recommendations did not change. They will be referred to in the comparison of evidence-based guidelines from other countries (see Section 3.2 ).
Up and down-grading of recommendations: Only one recommendation was downgraded. It is the recommendation for the use of low-dose neuroleptics in some cases of AN. The decision was based on the consideration that this recommendation should be followed only with caution and not as an overall clinical standard. In contrast, the recommendation not to use neuroleptics for the treatment of AN was upgraded due to an increase in evidence (systematic reviews). The same is true for the recommendation to continuously address motivation to change throughout treatment. Several studies show that motivation to change is a relevant predictor of treatment outcome. Recent high-quality trials made it possible to make specific recommendations regarding the use of specialised psychotherapeutic treatments. However, due to ethical reasons, no study compared an active treatment with untreated control groups. Therefore, it was decided that the recommendation should be classified as ‘B’ and not ‘A’. A further recommendation was upgraded based on clinical relevance; Inpatient treatment should take place in facilities which are able to offer a specialised multimodal treatment programme. In Germany, some adult and child and adolescent psychiatric and psychosomatic hospitals are not specialised and have no experience with the treatment of patients with AN. Treatment in such facilities is, therefore, not recommended, due to high associated risks, not to mention high costs. The new guideline also includes the explicit recommendation to consider co-morbidity in patients with AN. Co-morbid conditions like borderline-personality disorder or post-traumatic stress disorder, for example, might require changes in treatment planning and prioritisation of therapy goals. Although empirical evidence is scarce, a recommendation for a stabilisation phase as a final phase in inpatient treatment was included, as relapse after discharge is common [ 29 , 30 , 31 ], and the transition from one service level to another service level (especially to a level with less supervision and support) is a major challenge for patients with AN. Finally, there was new empirical evidence suggesting that a short inpatient stay for weight stabilisation followed by day hospital treatment is as effective as long-term inpatient treatment for children and adolescents with AN, providing there is continuity in the therapists that are responsible and if there is sufficient support by family members [ 23 ].
2.3. Network-Meta-Analysis
Based on the systematic literature search (see Section 2.2 ), a network-meta-analysis was conducted to answer the following question: What is the comparable effectiveness of different psychotherapeutic treatments for AN? Additionally, two further questions were addressed using standardised mean change statistics: What is the amount of weight gain that can be expected in different treatment settings? And: What is the amount of weight gain that can be expected in adolescents vs. adults?
Predefined inclusion and exclusion criteria were used to select the studies. Each study was rated by two independent researchers and additionally assessed for quality [ 18 ]. For more details on data analysis see [ 18 ].
Network-meta-analysis: 18 randomised controlled studies met inclusion criteria for the data-analysis. Ten studies were on adolescents (625 patients), and 8 studies were on adults (622 patients). No treatment approach was found to be superior. However, there were several limitations to the analysis and interpretation of results. All studies compared active treatments with each other, with no study including an untreated control group. Only a few comparisons were replicated. Furthermore, the majority of studies on adolescents evaluated family-based treatment approaches mostly by the same group of researchers, while interventions in adults were almost exclusively on an individual basis. The manualised treatment approaches that were evaluated in high quality trials comprise the Maudsley Model of Anorexia Nervosa Treatment for Adults (MANTRA) [ 25 ], Focal Psychodynamic Therapy (FPT) [ 26 , 32 , 33 ], Enhanced Cognitive Behaviour Therapy (CBT-E) [ 26 , 34 , 35 ], Specialist Supportive Clinical Management (SSCM) for adults [ 25 , 35 , 36 ], and family-based treatment (FBT) for adolescents [ 21 , 22 , 24 ].
Standardised mean change statistics (SCM): Analyses were conducted with 38 studies (1164 patients). Seventeen of these studies were naturalistic studies, and four studies were on adolescents (350 patients). For a course of up to 27 weeks, significantly higher weight gains can be expected in inpatient treatment compared to outpatient treatment (for adults: mean weight gain of 537 g/week in inpatient treatment vs. 105 g/week in outpatient treatment; for adolescents: mean weight gain of 615 g/week in inpatient treatment vs. 192 g/week in outpatient treatment). The estimated effect sizes for weight gain in adolescents were significantly higher compared to adults (in RCTs: SMC = 1.97 vs. 1.02, in naturalistic studies SMC = 1.84 vs. 1.42, respectively).
In sum, there are several existing manualised psychotherapeutic treatments for AN, which can be considered evidence-based and effective. However, there is a need for replication studies. There are differences regarding treatment response and most suitable treatment approach in adult versus adolescent patients.
3. Comparison of the German S3-Guideline with International Evidence-Based Clinical Treatment Guidelines
3.1. international evidenced-based eating disorders guidelines.
There are currently several additional evidence-based guidelines available, which provide recommendations regarding the diagnosis and treatment of eating disorders. Most of the guidelines were written by multidisciplinary groups (comprising health care professionals and researchers), and most were designed solely for use by health specialists involved in the treatment of eating disorders. The most recent of these guidelines are the Dutch [ 37 ] and the revised British guidelines [ 38 ], both published in 2017. The British guideline [ 38 ], published by the National Institute for Health and Care Excellence (NICE), addresses all age groups (children, adolescents and adults), and all eating disorder categories (AN, BN, BED and Other Specified Feeding or Eating Disorders (OSFED)). Several lay members of the community were involved in the development of this guideline. The Dutch guideline addresses AN, BN and BED [ 39 ]. This guideline, designed to be used by both specialists and population members, is only available in Dutch [ 39 ]. Healthcare professionals collaborated with patients and relatives, as well as health insurance representatives, during the developmental stages of the guideline [ 39 ].
The next most recent guideline, published in 2016, is the Danish guideline [ 40 ]. This ‘quick guide’, provides a brief overview, designed solely for the treatment of AN. The guideline is available in English, and it addresses all age groups. The full-length version of this guideline is only available in Danish. The Australian and New Zealand guideline [ 41 ] was published in 2014 by the Royal Australian and New Zealand College of Psychiatrists. Community members and stakeholders collaborated with healthcare professionals and academics in the development of the guideline. This guideline contains two sections separately addressing AN in children and adolescents, and in adults. BN and BED, as well as avoidant/restrictive food intake disorder, are also addressed.
In 2012, the American Psychiatric Association (APA) released a guideline watch [ 42 ], reviewing new evidence published since the last APA guideline in 2006, but gives no explicit recommendations [ 43 ]. This guideline addresses AN, BN and BED, and also makes reference to EDNOS. The guideline is designed primarily for the treatment of adults, but also briefly addresses the treatment of children and adolescents. The French guideline [ 44 ], published in 2010, is written specifically for AN. It addresses all age groups and is available in English. In 2009, the Spanish guideline [ 45 ] for eating disorders was published. This guideline, which concerns eating disorder patients over 8 years of age, is written not only for healthcare specialists, but also for the population and educational professionals. It addresses AN, BN, BED and EDNOS.
In addition to these national guidelines, several more specific evidence-based guidelines also exist. A guideline, developed specifically for the Canadian province of British Columbia, was released in 2010 [ 46 ]. This guideline addresses AN, BN and EDNOS (except BED), and advisesthe on treatment of all age groups. In 2011, the World Federation of Societies of Biological Psychiatry (WFSBP) released a guideline specifically addressing the pharmacological treatment of eating disorders [ 47 ]. This guideline, written in English, addresses the pharmacological treatment of AN, BN and BED. In 2014, the Management of Really Sick Patients with Anorexia Nervosa (MARSIPAN and Junior MARSIPAN) guideline [ 48 ] was published, a guideline which specifically addresses the treatment of children, adolescents and adult patients with ‘severe’ AN.
In line with an evidence-based approach, most of the guidelines explicitly state that the development of the guideline involved a systematic literature review, a rating of the identified literature, and a complex consensus process, involving collaboration and review by numerous experts [ 14 , 38 , 41 , 43 , 44 , 45 , 46 , 47 , 48 ]. Only the MARSIPAN [ 48 ] and WFSBP guidelines [ 47 ] do not explicitly refer to a complex consensus process, and the British Columbia guideline [ 46 ] does not mention a rating system. The Danish ‘quick guide’ [ 40 ] has a complete absence of information on the methodological process. However, the inclusion of evidence levels in the guide implies that the developmental process was rigorous. A detailed review of the evidence upon which the recommendations are based is only available in the British guideline and the Danish full-length guideline. A more detailed comparison of the methods employed in developing the guidelines goes beyond the scope of this review article. All of the guidelines are available online. The Australian and New Zealand, MARSIPAN, WFSBP and APA guidelines are published in online scientific journals and partly in print versions, and the remainder of the guidelines are available on the relevant publishing society’s website.
3.2. Commonalities and Differences
3.2.1. treatment setting.
For adults: Similar to the German guideline [ 14 ], all remaining guidelines (excluding the Danish [ 40 ] and WFSBP guidelines [ 47 ]) recommend outpatient treatment as a first treatment option, suggesting day patient or inpatient treatment as a more intensive treatment option if outpatient treatment proves ineffective [ 38 , 39 , 41 , 43 , 44 , 46 , 48 ]. The German guideline states, however, that in some cases this ‘stepped-care’ approach may not be appropriate.
Inpatient treatment is recommended in cases with a BMI <15 kg/m², rapid or continuing weight loss (>20% over 6 months), high physical risk, severe co-morbid conditions or denial of illness. If these criteria are met, an inpatient setting may be necessary for initial treatment. Likewise, all remaining guidelines (excluding the Danish and WFSBP guidelines) also suggest more intense treatment settings from the outset in cases of severe medical instability. All of these guidelines provide information regarding hospital admission criteria with varying degrees of detail, but agree on the necessity to judge the need for hospitalisation on an individual and multifactorial basis. Further, they state that compulsory treatment is possible in the case of extreme medical complications. The Danish and WFSBP guidelines do not make reference to treatment setting. For an overview of indicators of high medical risk and the handling of medical complications see the review of Zipfel and colleagues [ 4 ].
For children and adolescents: Corresponding to the treatment recommendations for adults, outpatient treatment is proposed as the first line treatment for children and young people by the German [ 14 ] and most other guidelines [ 38 , 41 , 43 , 44 ] if the patient is in a stable medical state. If more intensive care is needed, several guidelines suggest a graduated procedure from inpatient to partial and finally to outpatient treatment programs [ 40 , 44 , 45 ]. Only the German guideline [ 14 ] gives a special recommendation for a referral to day patient treatment. Interestingly, the British and accordingly the Spanish guidelines advise admitting children and young people to a setting with age-appropriate facilities, which are near to their home and have the capacity to provide appropriate educational activities [ 38 , 45 ].
Regarding medical risk and necessity for inpatient treatment, the Australian and New Zealand, British Columbia, British, APA and French guidelines [ 38 , 41 , 44 , 46 , 49 ] provide exact criteria, such as a BMI below the 3 rd percentile or an expected body weight (EBW) below 75%, an abnormally low heart rate or blood pressure, electrolyte disturbances, etc. However, the exact values vary between countries. As for adults, these guidelines also indicate psychiatric risk factors, such as suicidality or severe self-injurious behaviour. The German and Spanish guidelines [ 14 , 45 ] are more unspecific to indicate hospitalisation (see above). The German and French guidelines [ 14 , 44 ] also refer to psychosocial risks, such as social isolation and family crisis, to consider inpatient treatment.
3.2.2. Psychotherapy
For adults: All guidelines except for the Danish [ 40 ] and WFSBP [ 47 ] address the efficacy of specific psychological interventions. No guideline recommends one single superior treatment option. The German [ 14 ], British [ 38 ] and Dutch guidelines [ 39 ] conclude that cognitive-behavioural therapy (CBT or CBT-E respectively), MANTRA, and SSCM are equally effective treatment options, and so, all treatments are recommended as first-line options. Additionally, the German guideline recommends FPT as another first-line treatment option. The remaining guidelines all review evidence for CBT, as well as a variety of other treatments including SSCM [ 41 ], psychodynamic therapy [ 43 , 44 , 45 , 46 ], interpersonal therapy [ 43 , 45 , 46 ], behaviour therapy [ 45 ] and ‘systematic and strategic therapies’ [ 44 ]. These guidelines all conclude that psychological interventions are effective, however, state that there is insufficient evidence to identify which is the most efficacious. The French [ 44 ], Dutch [ 39 ] and APA guidelines [ 43 ] also suggest that psychological interventions may not be as effective in severely malnourished patients.
The Danish guideline [ 40 ] also recommends the use of psychotherapeutic treatments, however, does not make any recommendations regarding specific interventions. This guideline provides a ‘weak recommendation’ that both group and individual psychotherapeutic treatment be considered as first-line treatment options, based on ‘very low evidence’ which suggests the approaches are equally effective. Recommendations for the inclusion of alternative elements, such as meal support and supervised physical activity, during the treatment phase are mentioned. Other guidelines make specific recommendations against alternative treatments; for example, the German [ 14 ] and the Australian and New Zealand guidelines [ 41 ] state that nutritional counselling alone should not be used as the sole treatment, and the British guideline [ 38 ] recommends against the use of alternative physical therapies, such as yoga, warming therapy, transcranial magnetic stimulation and acupuncture. The Spanish guideline [ 45 ] also advises against the use of excessively rigid behavioural programs for inpatients.
Some guidelines make recommendations regarding the required duration of treatment. The Australian and New Zealand guideline [ 41 ] states that a longer-term follow-up is necessary as relapse is common, and the Spanish guideline [ 45 ] states that duration of treatment should span at least six months for outpatients and twelve months for inpatients. The APA guideline [ 43 ] states that due to the enduring nature of the illness, psychotherapeutic treatment is usually required for at least one year, and the British guideline [ 38 ] makes specific recommendations regarding the time span of treatments, for example suggesting that CBT treatment for eating disorders should consist of 40 sessions over 40 weeks. The French guideline [ 44 ] recommends that treatment should last at least one year after significant clinical improvement, and the German guideline [ 14 ] states that after outpatient treatment, patients should regularly meet with their general practitioner (GP), or other care coordinator, for at least one year. The German guideline also recommends that the last phase of inpatient treatment before transfer to an outpatient setting should include a stabilisation period where patients demonstrate that they can maintain the achieved weight gain for a specified amount of time.
Some treatment guidelines make additional specific recommendations. The German [ 14 ], French [ 44 ], MARSIPAN [ 48 ] and Australian and New Zealand guidelines [ 41 ] all emphasise the importance of adopting a multi-disciplinary, collaborative approach to treatment. In a similar vein, the German [ 14 ], British Columbia [ 46 ] and APA guidelines [ 43 ] highlight the importance of effective communication between all involved health workers, and recommend identifying someone to act as the primary care coordinator, such as the patient’s GP.
The MARSIPAN guideline [ 48 ] is specifically written regarding the treatment of patients who have a severe or enduring form of AN. The Australian and New Zealand [ 41 ] and British Columbia guidelines [ 46 ] also include comprehensive sections which address the treatment of such patients and suggest taking an alternative approach, focused on enhancing quality of life. The French [ 44 ], German [ 14 ] and APA guidelines [ 43 ] also briefly mention the treatment of patients with enduring AN. Other guidelines provide information regarding other additional elements related to AN. For example, both the Spanish [ 45 ] and French guidelines provide information regarding the care required for pregnant patients. Additionally, the APA and British Columbia guidelines include recommendations for therapists and specialists regarding communicating with patients (for example addressing the therapeutic relationship, boundaries). The German guideline does not entail any recommendations, but devotes a separate chapter to this topic.
For children and adolescents: All guidelines strongly recommend the involvement of parents or near caregivers in all treatment settings. The Australian and New Zealand, Spanish, APA and German guidelines explicitly mention family-based treatment or therapy (FBT) [ 14 , 41 , 43 , 45 ]. However, the Australian and British guidelines also propose alternatives if FBT is not appropriate, such as other forms of family therapy [ 41 ], as well as individual treatment, such as adolescent-focused therapy (AFT) or CBT, in older adolescents [ 38 , 41 ]. No guideline gives an explicit advice whether conjoint or separate FBT should be conducted. The French guideline does not refer to FBT, but to family therapy in general [ 44 ]. The British guideline also does not specifically use the term FBT, but has its own terminology instead (anorexia nervosa-focused family therapy, FT-AN) [ 38 ]. Although many key features of this treatment resemble FBT, FT-AN also includes other approaches, such as multi-family therapy, conjoint or separate family therapy and exclusion or inclusion of a family meal, which is a core feature of FBT. The British guideline also requests therapists and staff to be aware of or address carers’ needs [ 38 ].
A summary of guidelines’ essential key recommendations regarding psychotherapy for AN is shown in Table 2 .
International guidelines’ key recommendations regarding psychotherapy for AN.
| Recommendation | AUS [ ] | BC [ ] | DEN [ ] | FR [ ] | GER [ ] | NETH [ ] | SP [ ] | UK [ ] | US [ ] |
|---|---|---|---|---|---|---|---|---|---|
| For adults: | |||||||||
| Psychotherapy in general | + | + | + | + | + | + | + | + | + |
| Not as efficient in severely malnourished patients | N.R. | N.R. | N.R. | ✓ | N.R. | ✓ | N.R. | N.R. | ✓ |
| Specific psychological interventions | ✓ | ✓ | N.R. | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| CBT/CBT-E | + | + | N.R. | + | + | + | + | + | + |
| Psychodynamic Therapy | N.R. | + | N.R. | + | + | N.R. | + | N.R. | + |
| FPT | N.R. | N.R. | N.R. | N.R. | + | N.R. | N.R. | N.R. | N.R. |
| MANTRA | N.R. | N.R. | N.R. | N.R. | + | + | N.R. | + | N.R. |
| SSCM | + | N.R. | N.R. | N.R. | + | + | N.R. | + | N.R |
| IPT | N.R. | + | N.R. | N.R. | N.R. | N.R. | + | N.R. | + |
| For children and adolescents: | |||||||||
| Involvement of parents/near caregivers | + | + | + | + | + | + | + | + | + |
| Family therapy | FBT or other forms of family therapy | FBT | FBT or other forms of family therapy | Family therapy | FBT or other forms of family therapy | FBT? | Family therapy (systemic or not) | FT-AN | FBT or other forms of family therapy |
✓ recommendation given; + explicit recommendation in favour; N.R., no recommendation reported; AUS, Australia and New Zealand; BC, British Columbia; DEN, Denmark; FR, France; GER, Germany; NETH, The Netherlands; SP, Spain; UK, United Kingdom; US, United States; CBT(-E), (Enhanced) Cognitive Behaviour Therapy; FPT, Focal Psychodynamic Therapy; MANTRA, Maudsley Model of Anorexia Nervosa Treatment for Adults; SSCM, Specialist Supportive Clinical Management; IPT, Interpersonal Therapy; FBT, Family-Based Treatment/Therapy; FT-AN, AN-focused Family Therapy; 1 and siblings; ?, ambiguous evidence.
3.2.3. Nutritional Management
For adults: The WFSBP guideline [ 47 ] suggests that nasogastric feeding is effective for malnourished patients, however, does not address risks associated with refeeding, or provide any specific nutritional or weight gain recommendations. All remaining guidelines, (excluding the Danish guideline [ 40 ]), recommend nasogastric feeding for severely malnourished patients, when oral feeding is not an option [ 14 , 38 , 39 , 41 , 43 , 44 , 46 , 48 ]. These guidelines address the risk of refeeding syndrome, recommending that treatment is administered by experienced staff. The APA guideline [ 43 ] recommends nasogastric feeding over parenteral feeding, and the British guideline [ 38 ] explicitly recommends against parenteral nutrition. The German guideline also discusses the use of percutaneous endoscopic gastronomy feeding as a potential alternative, when patients will not tolerate nasogastric feeding [ 14 ]. The Danish guideline does not provide any recommendations regarding refeeding, nutritional intake or weight restoration.
In the original German guideline [ 15 ], an initial food intake of approximately 30 to 40 kcal/kg per day was recommended for highly underweight patients (see Table 1 ), which, upon revision, was considered too strict. The revised German guideline [ 14 ], as well as the Danish [ 40 ], French [ 44 ] and WFSBP guidelines [ 47 ], do not give specific recommendations regarding energy intake during refeeding. Both the British [ 38 ] and MARSIPAN guidelines [ 48 ] recommend commencing refeeding at 5 to 10 kcal/kg/day for severely underweight patients, and gradually increasing to 20 kcal/kg/day within 2 days. The British Columbia guideline [ 46 ] also recommends beginning refeeding at 5 to 10 kcal/kg/day if severity factors (e.g., nasogastric feeding) are involved. In the absence of severity factors, intake of 20 to 25 kcal/kg/day is recommended, and intake should not exceed 70 to 80 kcal/kg/day. The Spanish guideline [ 45 ] recommends a slightly higher caloric intake of 25 to 30 kcal/kg/day for severely malnourished patients, and they also provide a recommended upper limit of 1000 kcal/day. The APA guideline [ 43 ] recommends initiating refeeding at 30 to 40 kcal/kg/day, and also suggests that males may require a significantly higher energy intake to gain weight. The Dutch guideline has an even higher recommended refeeding starting point of 40 to 60 kcal/kg/day for severely underweight patients [ 39 ]. The Australian and New Zealand guideline [ 41 ] does not provide a recommended nutritional intake based on weight, but instead recommends a specific starting intake of 1433 kcal/day, with increases of 478kcal every 2 to 3 days.
Several guidelines also provide recommendations regarding appropriate weekly weight gain goals in inpatient and outpatient settings. Five guidelines recommend a minimum weight gain of 0.5 kg/week in an inpatient setting; the German [ 14 ], French [ 44 ] and Spanish guidelines [ 45 ] recommend weight gain ranging between 0.5 and 1 kg/week, the Australian and New Zealand guideline [ 41 ] recommends weight gain between 0.5 and 1.4 kg/week, and the Dutch guideline suggests weight gain ranging between 0.5 and 1.5 kg/week [ 39 ]. In contrast, the British Columbia guideline [ 46 ] suggests a higher minimum weight gain ranging from 0.8 to 1.4 kg/week, and the APA guideline [ 43 ] suggests a minimum weight gain ranging from 0.9 to 1.4 kg/week. The remaining guidelines [ 38 , 40 , 47 , 48 ] do not provide specific weight gain recommendations. Only four of the guidelines provide recommendations regarding weight gain per week in an outpatient setting. The French guideline recommends a weight gain of 0.25 kg/week, while the German, APA guidelines and Dutch recommend a weekly gain of between 0.2 to 0.5 kg [ 39 ].
For children and adolescents: The British guideline for the management of severely ill young people with AN (Junior MARSIPAN) [ 48 , 50 ] advocates to commence refeeding at about 40 kcal/kg/day and increase the meal plan by 200 kcal/day, while the others do not explicitly give calorie specifications for children and adolescents. Almost all guidelines recommend nasogastric tube feeding, if a meal plan and supplement drink tops are not managed [ 14 , 41 , 43 , 45 , 50 ].
The French, Danish and German guidelines emphasise the necessity of achieving a target weight at which menstruation can reoccur [ 14 , 40 , 44 ]. While the French guideline does not give any threshold criteria, the German guideline defines the 25 th age-adapted BMI-percentile (with the 10th percentile as a minimum) in contrast to the Danish guideline with the 50 th weight-for height percentile as target weight.
Supplementary nutritional counselling is advised by the British, Spanish and German guidelines for children and adolescents and their carers to help young people meet their dietary needs for pubertal development and growth [ 14 , 38 , 45 ]. According to these guidelines, growth and pubertal development should be regularly monitored in this age group.
3.2.4. Psychopharmacology
For adults: Use of pharmacotherapy is addressed in all treatment guidelines excluding the Danish guideline [ 40 ]. All of these guidelines emphasise the lack of evidence surrounding medication use for AN, and most guidelines emphasise that caution must be taken when administering medication, due to the physical complications associated with AN (e.g., cardiac problems). The Spanish [ 45 ], APA [ 43 ] and British guidelines [ 38 ] explicitly state that medication should not be used as the sole treatment. The British guideline also states that there is no proven benefit of combined treatment over psychotherapy alone in treating patients without comorbidities. All guidelines excluding the MARSIPAN [ 48 ], Danish and British guidelines give cautious recommendations for the use of antipsychotic medications. The French guideline [ 44 ] provides a cautionary recommendation, without addressing specific medications or effects. The remaining guidelines all make specific reference to the antipsychotic olanzapine; the German [ 14 ], WFSBP [ 47 ], Dutch [ 39 ], Australian and New Zealand [ 41 ], and APA guidelines recommended it to assist with anxious and obsessional thoughts, the WFSBP and Spanish guidelines suggest that it may be useful for improving general psychological symptoms, and the British Columbian [ 46 ], Spanish and APA guideline cautiously recommended it for improvements in weight gain. In contrast, the German guideline recommends against the use of antipsychotics for weight gain. The German guideline states there is no conclusive evidence to recommend the use of antidepressants for the core symptoms of AN, and the Dutch guideline also explicitly recommends against the use of selective serotonin reuptake inhibitors (SSRIs) [ 39 ]. In contrast, antidepressants are cautiously recommended by the French, WFSBP and APA guidelines, to assist with co-occurring symptoms of depression, obsessive–compulsive or anxiety disorder. Specifically, the APA guideline discusses the advantages of using selective serotonin reuptake inhibitors in combination with psychotherapy to address persistent depressive or anxiety symptoms, but recommends against the use of monoamine oxidase inhibitors and bupropion, due to adverse reactions and health risks. The APA guideline cautiously recommends the use of pro-motility agents for use against bloating, and use of antianxiety agents before eating for some patients. Similarly, the MARSIPAN guideline [ 48 ] discusses the use of benzodiazepines for particularly anxious patients. The WFSBP and APA guidelines discuss potential weight gain benefits of taking zinc supplements, while the German guideline suggests restricting zinc supplementation to cases with proven zinc deficiency.
For children and adolescents: With the exception of hormone replacement therapy the German and most other international guidelines do not give any specific recommendations for this age group. The Junior MARSIPAN guideline concludes that it ‘may be necessary to prescribe regular sedative antipsychotic medication, such as olanzapine’, if the patients are extremely agitated and resist refeeding [ 48 ]. It also gives clear recommendations for ECG monitoring if antipsychotics are applied. Hormone replacement therapy: In several guidelines including the German guideline the prescription of an oral contraceptive is not recommended [ 38 , 41 , 43 ]. The British guideline suggests considering a bone mineral density scan after one year of underweight in children and adolescents. Moreover—in correspondence to the German guideline—the British guideline suggests to consider transdermal estrogen replacement in combination with cyclic progesterone application in girls with a bone age over 15 years and long-term underweight as well as incremental physiological doses of estrogen in those below 15 years [ 14 , 38 ]. Similar indications are mentioned in the APA and the Australian and New Zealand guidelines [ 41 , 43 ].
4. Discussion
This review provides an overview of the newly revised and published German S3-guideline for eating disorders [ 14 ]. In particular, it highlights the changes in recommendations regarding the treatment of AN since the publication of the original guideline in 2011 [ 15 ]. In summary, family-based therapy approaches are recommended for adolescents, whereas individual approaches are suggested for adults. There is no evidence indicating the superiority of one specialised approach over another. In more intensive settings, as well as in adolescents, higher weight gains can be expected. To date, there is no convincing evidence for the positive effect of pharmacotherapy regarding the core symptoms of AN.
The revised German guideline is currently the most recent eating disorder treatment guideline internationally. Recommendations are, therefore, based on the most up to date research findings and evidence available. The development of this guideline involved a rigorous process, including a comprehensive literature review and analysis, and consultation and contribution by many experts in the eating disorder field. The findings of the literature review and network analysis are also available in English [ 18 ].
The German guideline also includes an easily comprehensible guide for sufferers with eating disorders and their relatives [ 17 ], which has been developed with the help of patient representatives. The German guideline, hereby, stresses the necessity of providing information and support to significant others, who often bear a high emotional burden, but also play an important role in helping patients to overcome the eating disorder. The guideline has been published in two different formats—as a scientific book (only the original version so far) and on the website of the Association of the Scientific Medical Societies in Germany (AWMF, awmf.org [ 14 ]), where it is freely available.
Similar to the Dutch guideline, the original version of the German guideline has been published in German only, which limits its distribution and implementation to Germany, Austria and Switzerland. An English translation of the revised version, which is currently in preparation, is, therefore, an invaluable step towards increasing the utility of this guideline.
The review also explores the similarities and differences between the German guideline and other existing international guidelines. There is significant homogeneity among the international guidelines in the recommendations derived from the existing evidence. All agree that there is no superior treatment for AN, if specialised approaches are compared. There are, however, some inconsistencies regarding aspects, such as medication and nutritional management. Most guidelines implemented a thorough methodology. We think there is a need for European research initiatives which aim to enhance the evidence base and clinical guidance regarding AN across the different participating countries. Recommendations must, however, take into account the specificities of the national health care systems.
Overall, evidence for treatment of AN has increased, yet even in the latest German guideline, many of the recommendations are still based on expert opinion. Guidelines do not only mirror the current state of research but also point out gaps that need to be bridged. There is still a need for more research in the field of eating disorders, particularly in AN. In view of the so-called ‘research-practice gap’, it needs to be mentioned that guidelines are not designed to propagate conformist standard therapy, or to restrict clinicians’ individual willingness to learn and innovate. They should not be seen as directives, but as advice.
5. Conclusions
The German S3-guideline is, at present, the most recently revised evidence-based treatment guideline for AN. Based on newly available evidence, several amendments have been made regarding treatment recommendations, since the original guideline publication in 2011. Overall, the recommendations provided in the German guideline are fairly consistent with those provided in other international evidence-based eating disorder guidelines. Adult and adolescent patients should be distinguished in terms of treatment response and the most suitable treatment approach. Although the existing guidelines provide a sound base of information, which can be used by healthcare professionals to guide diagnosis and treatment decisions, further research regarding the treatment of AN is still urgently needed.
Acknowledgments
The support of the publication fund of the University Hospital Tuebingen was greatly appreciated. We would also like to thank all contributors to the German S3-guideline and the Arbeitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften (AWMF). The authors would like to thank Brigid Kennedy for her help in preparing this manuscript.
Author Contributions
Conceptualisation, G.R. and A.Z.; methodology, A.Z.; investigation, G.R.; writing—original draft preparation, G.R., S.H., B.H.-D. and A.Z.; writing–review and editing, G.R.
The S3-guideline was funded by the Christina Barz-Stiftung in the Association of German Academic Foundations.
Conflicts of Interest
The authors declare no conflict of interest.
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- My Bibliography
- Collections
- Citation manager
Save citation to file
Email citation, add to collections.
- Create a new collection
- Add to an existing collection
Add to My Bibliography
Your saved search, create a file for external citation management software, your rss feed.
- Search in PubMed
- Search in NLM Catalog
- Add to Search
State or trait: the neurobiology of anorexia nervosa - contributions of a functional magnetic resonance imaging study
Affiliations.
- 1 Department of Psychosomatic Medicine and Psychotherapy, Medical Center - University of Freiburg, Faculty of Medicine, University of Freiburg, Freiburg, Germany.
- 2 Department of Psychiatry and Psychotherapy, Medical Center - University of Freiburg, Faculty of Medicine, University of Freiburg, Freiburg, Germany. [email protected].
- 3 Department of Psychiatry and Psychotherapy, Medical Center - University of Freiburg, Faculty of Medicine, University of Freiburg, Freiburg, Germany.
- 4 Center for Basics in Neuromodulation, Faculty of Medicine, University of Freiburg, Freiburg, Germany.
- 5 Department of Psychosomatic Medicine and Psychotherapy, Ortenau Klinikum, Lahr, Academic Teaching Hospital of the University of Freiburg, Lahr, Germany.
- PMID: 35641995
- PMCID: PMC9158182
- DOI: 10.1186/s40337-022-00598-7
Background: The understanding of the cerebral neurobiology of anorexia nervosa (AN) with respect to state- versus trait-related abnormalities is limited. There is evidence of restitution of structural brain alterations with clinical remission. However, with regard to functional brain abnormalities, this issue has not yet been clarified.
Methods: We compared women with AN (n = 31), well-recovered female participants (REC) (n = 18) and non-patients (NP) (n = 27) cross-sectionally. Functional magnetic resonance imaging was performed to compare neural responses to food versus non-food images. Additionally, affective ratings were assessed.
Results: Functional responses and affective ratings did not differ between REC and NP, even when applying lenient thresholds for the comparison of neural responses. Comparing REC and AN, the latter showed lower valence and higher arousal ratings for food stimuli, and neural responses differed with lenient thresholds in an occipital region.
Conclusions: The data are in line with some previous findings and suggest restitution of cerebral function with clinical recovery. Furthermore, affective ratings did not differ from NP. These results need to be verified in intra-individual longitudinal studies.
Keywords: Anorexia nervosa; Functional magnetic resonance imaging; Recovery; State; Trait.
Plain language summary
There is abundant evidence of structural and functional brain alterations during the acute stage of anorexia nervosa (AN), although affected brain areas differ based on various study methodologies. Meanwhile, investigations indicate that brain structure normalizes with weight and clinical restitution. The current cross-sectional investigation examines acutely ill AN patients, healthy controls, i.e. non-patients (NP) and well-recovered individuals (REC), with respect to brain function. Functional cerebral responses of participants exposed to food pictures were investigated. Neither in terms of function nor emotional experience of food stimuli, the REC differed from the NP group. This study points to brain function normalizing with clinical and weight restoration, which should be verified in intra-individual longitudinal studies.
© 2022. The Author(s).
PubMed Disclaimer
Conflict of interest statement
SG: no conflict of interest, KN: no conflict of interest, IH: no conflict of interest, DE: no conflict of interest, AZ: no conflict of interest, KD: Member of the ‘Steering Committee Neurosciences’, Janssen Pharmaceuticals, Inc., CL: no conflict of interest, LTvE: Advisory boards, lectures, or travel grants within the last three years: Roche, Eli Lilly, Janssen-Cilag, Novartis, Shire, UCB, GSK, Servier, Janssen and Cyberonics, SM: no conflict of interest, AJ: no conflict of interest.
Examples of food and non-food…
Examples of food and non-food stimuli (cf. [45])
Exclusion flow chart. AN anorexia…
Exclusion flow chart. AN anorexia nervosa, NP non-patients, REC recovered AN
Similar articles
- Food motivation circuitry hypoactivation related to hedonic and nonhedonic aspects of hunger and satiety in women with active anorexia nervosa and weight-restored women with anorexia nervosa. Holsen LM, Lawson EA, Blum J, Ko E, Makris N, Fazeli PK, Klibanski A, Goldstein JM. Holsen LM, et al. J Psychiatry Neurosci. 2012 Sep;37(5):322-32. doi: 10.1503/jpn.110156. J Psychiatry Neurosci. 2012. PMID: 22498079 Free PMC article.
- Increased neural processing of rewarding and aversive food stimuli in recovered anorexia nervosa. Cowdrey FA, Park RJ, Harmer CJ, McCabe C. Cowdrey FA, et al. Biol Psychiatry. 2011 Oct 15;70(8):736-743. doi: 10.1016/j.biopsych.2011.05.028. Epub 2011 Jun 28. Biol Psychiatry. 2011. PMID: 21714958
- Intact value-based decision-making during intertemporal choice in women with remitted anorexia nervosa? An fMRI study. King JA, Bernardoni F, Geisler D, Ritschel F, Doose A, Pauligk S, Pásztor K, Weidner K, Roessner V, Smolka MN, Ehrlich S. King JA, et al. J Psychiatry Neurosci. 2020 Mar 1;45(2):108-116. doi: 10.1503/jpn.180252. J Psychiatry Neurosci. 2020. PMID: 31595737 Free PMC article.
- Functional brain alterations in anorexia nervosa: a scoping review. Fuglset TS, Landrø NI, Reas DL, Rø Ø. Fuglset TS, et al. J Eat Disord. 2016 Nov 28;4:32. doi: 10.1186/s40337-016-0118-y. eCollection 2016. J Eat Disord. 2016. PMID: 27933159 Free PMC article. Review.
- Morphological changes in the brain of acutely ill and weight-recovered patients with anorexia nervosa. A meta-analysis and qualitative review. Seitz J, Bühren K, von Polier GG, Heussen N, Herpertz-Dahlmann B, Konrad K. Seitz J, et al. Z Kinder Jugendpsychiatr Psychother. 2014 Jan;42(1):7-17; quiz 17-8. doi: 10.1024/1422-4917/a000265. Z Kinder Jugendpsychiatr Psychother. 2014. PMID: 24365959 Review.
- Brain Correlates of Eating Disorders in Response to Food Visual Stimuli: A Systematic Narrative Review of FMRI Studies. Celeghin A, Palermo S, Giampaolo R, Di Fini G, Gandino G, Civilotti C. Celeghin A, et al. Brain Sci. 2023 Mar 9;13(3):465. doi: 10.3390/brainsci13030465. Brain Sci. 2023. PMID: 36979275 Free PMC article. Review.
- Steinhausen H-C. The outcome of anorexia nervosa in the 20th century. Am J Psychiatry. 2002;159:1284–1293. doi: 10.1176/appi.ajp.159.8.1284. - DOI - PubMed
- Zipfel S, Giel KE, Bulik CM, Hay P, Schmidt U. Anorexia nervosa: aetiology, assessment, and treatment. Lancet Psychiatry. 2015;2:1099–1111. doi: 10.1016/S2215-0366(15)00356-9. - DOI - PubMed
- Treasure J, Schmidt U. The cognitive-interpersonal maintenance model of anorexia nervosa revisited: a summary of the evidence for cognitive, socio-emotional and interpersonal predisposing and perpetuating factors. J Eat Disord. 2013;1:13. doi: 10.1186/2050-2974-1-13. - DOI - PMC - PubMed
- Seitz J, Herpertz-Dahlmann B, Konrad K. Brain morphological changes in adolescent and adult patients with anorexia nervosa. J Neural Transm. 2016;123:949–959. doi: 10.1007/s00702-016-1567-9. - DOI - PubMed
- Nickel K, Joos A, van Elst LT, Matthis J, Holovics L, Endres D, et al. Recovery of cortical volume and thickness after remission from acute anorexia nervosa. Int J Eat Disord. 2018;51:1056–1069. doi: 10.1002/eat.22918. - DOI - PubMed
Related information
Grants and funding.
- JO 744/2-1/Deutsche Forschungsgemeinschaft
LinkOut - more resources
Full text sources.
- BioMed Central
- Europe PubMed Central
- PubMed Central
Miscellaneous
- NCI CPTAC Assay Portal

- Citation Manager
NCBI Literature Resources
MeSH PMC Bookshelf Disclaimer
The PubMed wordmark and PubMed logo are registered trademarks of the U.S. Department of Health and Human Services (HHS). Unauthorized use of these marks is strictly prohibited.
- Open access
- Published: 31 May 2022

State or trait: the neurobiology of anorexia nervosa — contributions of a functional magnetic resonance imaging study
- Selma Göller 1 na1 ,
- Kathrin Nickel 2 na1 ,
- Isabelle Horster 1 ,
- Dominique Endres 2 ,
- Almut Zeeck 1 ,
- Katharina Domschke 2 , 4 ,
- Claas Lahmann 1 ,
- Ludger Tebartz van Elst 2 ,
- Simon Maier 2 na2 &
- Andreas A. B. Joos 1 , 3 na2
Journal of Eating Disorders volume 10 , Article number: 77 ( 2022 ) Cite this article
3268 Accesses
2 Citations
18 Altmetric
Metrics details
The understanding of the cerebral neurobiology of anorexia nervosa (AN) with respect to state- versus trait-related abnormalities is limited. There is evidence of restitution of structural brain alterations with clinical remission. However, with regard to functional brain abnormalities, this issue has not yet been clarified.
We compared women with AN (n = 31), well-recovered female participants (REC) (n = 18) and non-patients (NP) (n = 27) cross-sectionally. Functional magnetic resonance imaging was performed to compare neural responses to food versus non-food images. Additionally, affective ratings were assessed.
Functional responses and affective ratings did not differ between REC and NP, even when applying lenient thresholds for the comparison of neural responses. Comparing REC and AN, the latter showed lower valence and higher arousal ratings for food stimuli, and neural responses differed with lenient thresholds in an occipital region.
Conclusions
The data are in line with some previous findings and suggest restitution of cerebral function with clinical recovery. Furthermore, affective ratings did not differ from NP. These results need to be verified in intra-individual longitudinal studies.
Plain English summary
There is abundant evidence of structural and functional brain alterations during the acute stage of anorexia nervosa (AN), although affected brain areas differ based on various study methodologies. Meanwhile, investigations indicate that brain structure normalizes with weight and clinical restitution. The current cross-sectional investigation examines acutely ill AN patients, healthy controls, i.e. non-patients (NP) and well-recovered individuals (REC), with respect to brain function. Functional cerebral responses of participants exposed to food pictures were investigated. Neither in terms of function nor emotional experience of food stimuli, the REC differed from the NP group. This study points to brain function normalizing with clinical and weight restoration, which should be verified in intra-individual longitudinal studies.
Anorexia nervosa (AN) is an eating disorder largely affecting young women with high morbidity, chronicity and mortality [ 1 ]. Apart from a restriction of energy intake leading to a significant weight loss, an intense fear of gaining weight and body image disturbance are key symptoms. The etiology is not well understood, though genetic disposition is one important factor, accompanied by psychosocial factors [ 2 ]. Maintaining factors include consequences of malnutrition, as well as consequences of isolation and depression [ 3 ]. Brain imaging studies point towards shrinkage of white (WM) and grey matter (GM) and complementary increase of the cerebrospinal spaces [ 4 , 5 ]. Longitudinal investigations show restitution of GM and WM volumetric alterations following long-term weight restoration [ 4 ]. Apart from structural brain alterations, also the function of various domains has been reported to be affected in acute AN [ 6 , 7 ], although the involved brain areas differed [ 8 ]. Due to significant loss of weight, cerebral aberrations might be associated with metabolic changes, i.e. starvation, and it is difficult to disentangle other factors associated with eating disorder psychopathology and/or predisposing factors [ 9 ].
Hence, the question remains which abnormalities are state phenomena, i.e. occur only during the acute phase of the disease, and which are trait-related. Persistent cerebral aberrations could be a predisposing phenomenon and might therefore represent endophenotypes [ 10 ], but they may also represent sequels, i.e. “scars” of the acute disease. As AN is a disorder with low prevalence [ 2 ], it is methodically difficult to study individuals who might develop AN longitudinally in order to shed light on the question of endophenotypic cerebral aberrations. Therefore, a first step to clarify questions about state and trait is to investigate individuals recovered from AN (REC).
With respect to the issue of state-trait in AN, previous studies examined psychological, cognitive and behavioral variables [ 11 , 12 ] as well as metabolic, structural and functional correlates of the brain [ 13 , 14 , 15 ]. Overall, research on reversibility of psychological, cognitive and behavioral functioning in REC presents a heterogeneous picture [ 16 , 17 , 18 ]. In terms of structural cerebral alterations, neuroimaging studies provide strong evidence of remission with clinical recovery [ 5 , 19 , 20 ]. Findings of functional magnetic resonance imaging (fMRI) studies comparing REC and non-patients (NP) show divergent results. With respect to visual food cues, some authors reported no alteration in brain activation in REC (when compared to NP) [ 21 , 22 , 23 ], while Uher et al. [ 24 ] detected increased activation of prefrontal and anterior cingulate cortices (ACC) and a reduction of activity in parietal regions in a small group of REC. Further studies found hypoactivation of the insula [ 25 ] or increased caudate activation [ 26 ] in response to food pictures. With regard to other disorder specific paradigms (e.g. taste, body shape), both increased and decreased functional neural brain responses for REC (compared to NP) were found in various brain regions [ 13 , 27 , 28 , 29 , 30 ], while two studies detected no altered brain activity in REC [ 31 , 32 ]. However, a majority of fMRI studies using non-disorder specific stimuli (e.g., fear, intimacy, reward) reported no or only minimal functional aberrations in REC [ 33 , 34 , 35 , 36 ]. Longitudinal fMRI studies with non-disorder specific paradigms yielded conflicting results in delay discounting tasks [ 37 , 38 ], a normalization in a working memory and set-shifting task [ 39 , 40 ], and persisting changes in theory of mind and reward learning paradigms [ 41 , 42 ]. For further details and an overview of previous studies, see Additional file 1 .
This investigation focuses on the question of restitution vs. non-restitution of functional brain abnormalities using a cross-sectional design in order to address the topic state and trait of the neurobiology in AN. In this context, the comparison of NP and REC is of particular interest. It complements previous studies on disease-specific food stimuli in REC [ 21 , 22 , 23 , 24 , 25 , 26 ]. The paradigm has already been employed previously in a study of AN with some REC participants [ 24 , 43 ] and a study focusing on NP versus AN [ 44 ]. Data from the current study comparing NP and AN has been reported previously, with a focus on replicability issues [ 45 ]: Group comparisons yielded higher blood oxygenation-level dependent (BOLD) responses of AN compared to NP in midcingulate, pre/postcentral and parietal areas when using a lenient initial threshold, and no significant group differences with a conservative initial threshold.
Based on results of preceding whole-brain analyses [ 23 , 25 ], we expected no differences between REC and NP in neural response to food stimuli or behavioral/experiential response, i.e., affective ratings of stimuli [ 22 , 24 , 46 ]. Additionally, we performed exploratory analyses of affective ratings and insula activation, as earlier studies found a positive correlation between food pleasantness ratings and insula activation in the NP group [ 22 , 25 ], but not in the AN [ 25 ] or REC groups [ 22 ].
Participants
AN and REC participants were recruited via the Department of Psychosomatic Medicine and Psychotherapy of the University Medical Center Freiburg. NP were recruited via local advertisements. The study was performed following written informed consent from the participants. The data was collected between March 2015 and October 2017.
Thirty-one AN, 18 REC and 27 NP were included in the final analysis. AN participants had to fulfill DSM-5 criteria. The following inclusion criteria were defined for the REC group: (1) Absence of eating disorder symptomatology for more than 12 months and an Eating Disorder Examination (EDE) [ 47 ] within one standard deviation of normal; (2) The Body Mass Index (BMI) was aimed at ≥ 20 kg/m 2 , which we achieved for most REC. The BMI of four participants was slightly below 20 kg/m 2 (between 19.3 and 19.7 kg/m 2 ) and of one participant 18.8 kg/m 2 . We decided to include these participants because they were clinically completely recovered and had always had a BMI in this range before the onset of the disease. Three AN patients were of the binge eating/purging type, all other AN and REC were of the restrictive type. Patients with AN were seen in the outpatient clinic for diagnostic reasons while nine were right at the beginning of inpatient treatment. Exclusion criteria had been reported previously [ 45 ].
The participants examined in the current study largely overlap with those of our previous investigations [ 5 , 14 , 19 , 34 , 35 , 45 ].
The study was approved by the local Ethics Committee (EK 520/13). Participants were assessed by means of the SCID interview [ 48 , 49 ], the EDE [ 47 , 50 ] and the following self-report questionnaires: Beck Depression Inventory-II (BDI-II) [ 51 ], Eating Disorder Inventory-2 (EDI-2) [ 52 ], State-Trait Anxiety Inventory (STAI) [ 53 ], and a crystalline intelligence test (MWT-B) [ 54 ]. All participants were studied in the second half of the menstrual cycle or the corresponding phase with estrogen and progesterone when taking oral contraception. In the morning around 8 a.m., participants were provided with a standardized breakfast, the calories consumed were counted, and the feeling of satiety was rated on a Likert-scale from 0 (very hungry) to 9 (very satiated).
Participants viewed via a mirror photographs of food and non-food items of similar structure [ 43 , 44 , 45 ] presented on a BOLDScreen monitor at the rear of the scanner bore. In a block design with five blocks per condition of 30 s each, 10 consecutive food or non-food pictures were presented alternately per block.
Participants were asked to look attentively at the pictures. Examples of the picture stimuli are displayed in Fig. 1 .

Examples of food and non-food stimuli (cf. [ 45 ])
Behavioral data
After scanning, participants rated a selection of images (10 food and 10 non-food images) with respect to three emotion dimensions (valence, arousal and dominance) using “manikin ratings” based on the International Affective Picture System on scales from zero to eight [ 54 ]. The dominance scale assesses how much the viewer feels controlled or in control when watching the images [ 55 ]. Ratings of two AN and two NP could not be included due to incomplete data.
Image acquisition and processing
A T1-weighted Magnetization-prepared rapid gradient echo (MPRAGE) sequence (TR = 2300 ms, TE = 2.98 ms, flip angle = 9°, FOV = 240 * 256 mm 2 , voxel size = 1 × 1 × 1 mm 3 ) was recorded as an anatomical reference. 138 functional echo-planar T2*-weighted (EPI) images (TR = 2500 ms, TE = 30 ms, flip angle = 90°, FOV = 192 * 192 mm 2 , Matrix volume = 64 × 64, 38 slices, voxel size = 3 × 3 × 3 mm 3 ) were recorded with a Siemens 3 T Prisma MAGNETOM (Siemens Medical Systems, Erlangen) using a 20-channel head coil. All EPI images were automatically rigid-body transformed to correct for head motion and a distortion correction algorithm was applied [ 56 ]. Preprocessing and statistical analysis of the functional data was performed with the statistical parametric mapping software SPM12 (Wellcome Trust Centre of Imaging Neuroscience, London; for details, see [ 57 ]). The first two volumes of each run were disregarded and an artifact detection algorithm (ArtRepair toolbox, SPM) was applied to detect head motion and possible spiking artifacts. The functional raw images were realigned to the first volume to generate six head motion parameters (rotation and translation in x, y, z direction), which were used as regressors of no interest in the first-level statistical analysis to correct for influences of head motion. The ‘Artrepair’ tool implemented in SPM12 was used to correct movement artifacts over half a voxel size by interpolating the measurement time points before and after the movement. Participants whose head movements were larger than half a voxel size (corresponding to 1.5 mm for a voxel size of 3 × 3 × 3 mm 3 ) were excluded from the analysis if more than two consecutive measurement time points (= "volumes") were affected or more than two corrections had to be made in the time series. In the case of spiking artifacts again the ‘Artrepair’ toolbox was used to correct single slices by interpolating the slice below and above the affected slice. If several slices of a single volume were affected, we interpolated (correspondingly to motion artifacts) the measurement time points before and after the volume affected by spiking artifacts. If two consecutive volumes or more than two volumes in total were affected by spiking artifacts, the subject was excluded. The motion corrected images were spatially normalized into the MNI (Montreal Neurological Institute) reference system applying the anatomical MPRAGE image. To increase the signal-to-noise ratio and to compensate for inter-individual differences in location of corresponding functional areas, the data was spatially smoothed with a three-dimensional isotropic Gaussian kernel (8 mm FWHM). Low frequency artifacts across the time-series were removed applying a high-pass filter (128 s).
Statistical analysis
Demographic, clinical and behavioral data were assessed by analyses of variance with a level of significance of p < 0.05 (two-sided).
Food and non-food regressors were convolved with a canonical hemodynamic response function and fitted together with the six regressors for head motion parameters in a linear regression model (general linear model (GLM)) with the functional signal time courses for each voxel and participant.
Within-group activation
In the second-level whole brain analysis, we tested for within-group differences (group activation) by performing a one-sample t -test for the food > non-food contrast of the first-level beta estimates of the food and non-food regressors.
Between-group comparison
For group comparisons, the first-level food > non-food contrast was used to compare AN > REC, REC > AN, NP > REC, and REC > NP in a two-sample t-test.
For both, the within- and between-group analysis: (1) We added age as a covariate. (2) We performed whole brain analyses with a cluster-defining threshold, i.e., initial height threshold, of p uncorr. < 0.001 and a minimum cluster size of 10 voxels (k ≥ 10) (3) Results were corrected for multiple comparisons on a cluster level applying family-wise error correction with a threshold of p corr. < 0.05.
For the between-group analysis, we additionally performed analyses with a cluster-defining threshold of p uncorr. < 0.01 and a minimum cluster size of 10 voxels, as previous studies had used lenient thresholds of p uncorr. < 0.01 [ 24 , 44 ] or even lower ( p uncorr. < 0.05) [ 22 , 25 ].
Moreover, we performed ROI-based (region of interest) small volume correction (SVC) for insula and amygdala ROIs according to the AAL3 atlas [ 58 ].
Comparisons of the AN and NP group are not reported as they have already previously been published [ 45 ].
Multiple regression analysis
For all groups and for each group separately, SPM multiple regression analyses were performed to calculate the correlation of the first-level BOLD contrast (food and non-food) with the valence and arousal ratings of the food stimuli. We tested for positive and negative linear effects of stimulus ratings and included the factor group as a regressor of non-interest (to adjust for possible group differences). We set up an interaction model with the factors group and stimulus ratings as regressors of interest and age as regressor of non-interest and compared the regression slopes of AN > NP, AN > REC, and NP > REC and vice versa.
We set-up three separate Analysis of Variance (ANOVA) models for valence/arousal/dominance (dependent variable) with the independent factor group followed by post-hoc Tukey Kramer tests to assess for between-group differences.
One hundred and eight female participants (40 AN, 24 REC, 44 NP) were recruited. Thirty-two data sets had to be discarded due to insufficient data quality, spiking head motion or incomplete data (Fig. 2 ). The food paradigm was performed towards the end of the imaging session (after approximately 35 min). This might have resulted in increased head motion (three subjects) and termination of the session by the participants (five). Spiking artifacts were also more likely to occur at the end of a scanning session, possibly due to the scanner heating up, requiring the exclusion of further 22 participants (Fig. 2 ). Seventy-six functional data sets could finally be analyzed: 31 AN, 18 REC and 27 NP.

Exclusion flow chart. AN anorexia nervosa, NP non-patients, REC recovered AN
Post-hoc power calculation
A post-hoc power calculation for the food > non-food contrast with Random Field Theory control was performed separately for the final AN (power 0.81 with cluster-defining threshold p < 0.001, power 0.59 with cluster-defining threshold p < 0.01), REC (power 0.32 with cluster-defining threshold p < 0.001, power 0.24 with cluster-defining threshold p < 0.01) and NP (power 0.88 with cluster-defining threshold p < 0.001, power 0.54 with cluster-defining threshold p < 0.01) samples applying the “Neuro-Powertool” ( http://neuropowertools.org/ ; retrieval date 05th May, 2022).
Demographic and clinical characteristics
Patients with AN had typical features of psychopathology and a lower BMI compared to the REC and NP group. REC participants showed good restitution of all clinical data (Table 1 ). The NP group was matched to the AN group and therefore younger than REC (Table 1 ). Lowest BMI and duration of illness did not differ between REC and AN. Patients with AN consumed fewer calories than REC and NP, but did not differ in feeling of satiety.
The contrast food > non-food showed increased BOLD activation of frontoinsular cortices in all three groups (Table 2 , Fig. 3 a). Within-group analyses showed no significant results in any of the three groups for the contrast non-food > food.
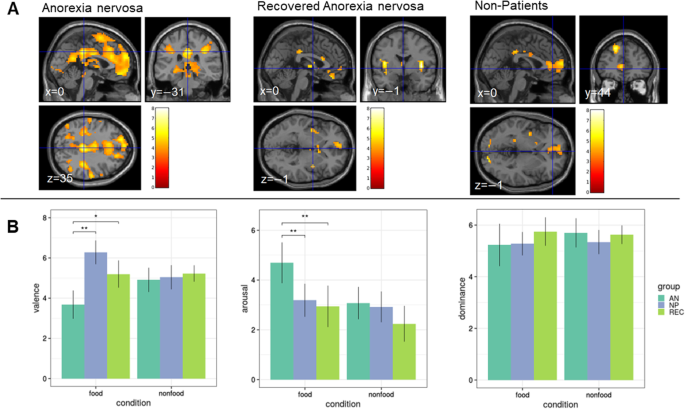
A T-maps of within-group differences for the contrast food > non-food for AN, REC and NP, p uncorr. < 0.001, k ≥ 10 voxels. Color bars represent the t-scores (white/yellow = high, red = low). B Behavioral data. Mean valence (0 = negative to 8 = positive valence), arousal (0 = unaroused to 8 = very aroused) and dominance (0 = not dominant to 8 = very dominant) ratings for the three groups (AN, REC, NP) and the two conditions (food and non-food). * p < 0.01, ** p < 0.001. AN anorexia nervosa; NP non-patients, REC recovered AN (cf. [ 45 ])
Applying the initial threshold of p uncorr. < 0.001, the two-sample t-test revealed no significant BOLD differences between groups for any of the comparisons (AN > REC, REC > AN, NP > REC, REC > NP).
At a lenient cluster-forming initial threshold of p uncorr. < 0.01, there were no differences between REC and NP neither. AN showed a higher BOLD response of an occipital area compared to REC (Additional file 2 ). Comparisons of AN and NP have been reported previously [ 45 ], where AN showed higher BOLD responses compared to NP in midcingulate, pre/postcentral and parietal areas when using a lenient initial threshold, and no significant group differences with recommended more stringent initial cluster-forming thresholds [ 45 , 59 ]. Neither the SVC analysis for the insula nor for the amygdala ROIs survived family-wise error correction.
None of the regression models yielded results that exceeded the significance threshold. Additional file 3 depicts the results of the performed multiple regression models for the affective ratings (arousal/valence).
The AN group had lower valence and higher arousal ratings compared to both REC and NP, while REC did not differ from NP (Table 3 ; Fig. 3 b). There were no group differences in the ratings of non-food stimuli.
This cross-sectional fMRI study on functional neural correlates towards disease-related stimuli (food vs. non-food images) revealed no group differences between REC and NP with neither a conservative initial height threshold [ 45 ] nor a lenient one. REC and NP also showed no difference in affective ratings, i.e. with respect to the subjective experience of the stimuli. Furthermore, there was no significant correlation of the valence and arousal ratings with the BOLD response towards food stimuli in the insular cortex.
The lack of group differences between REC and NP in functional neural correlates to visual food cues is in line with results of previous studies, which also detected no differences between REC and NP [ 21 , 22 , 23 ]. However, some studies reported differences between groups [ 24 , 25 , 26 ], which was only the case in region-of-interest analyses, but not whole-brain in the Holsen et al. sample [ 25 ]. These inconsistencies are likely due to a variety of reasons, some of which are associated with limited reliability and replicability [ 45 ]. Further, it is likely that studies yielded false-positive results due to small sample sizes as well as different statistical methods (e.g., region of interest vs. whole brain analyses, statistical thresholds, etc.) [ 60 , 61 ]. Comparability of studies is hampered due to heterogeneity across participants (BMI, duration of illness and recovery, etc.), within participants (daytime, hormonal level, etc.), and across study sites (study design, scanner hardware, etc.) [ 45 , 62 ]. This, once again, supports the need of replication studies [ 63 ], especially in the field of fMRI [ 64 , 65 ]. With respect to the early study of Uher et al. [ 24 ], which reported group differences of functional neural correlates and which used visual stimuli similar to ours, these factors likely account for the differing results.
Naturally, recovery criteria are another important topic with respect to state and trait. Recovery in AN in principle requires the absence of eating disorder psychopathology and a minimal BMI for a certain period of time [ 66 , 67 , 68 , 69 ], although the exact quantitative values vary.
For further detailed discussion of this topic with respect to recovery, remission and relapse see for example Khalsa et al. [ 69 ]. We used conservative measures in the current study, i.e. absence of eating disorder pathology for at least 12 months, an EDE score within one standard deviation of normal. Most REC were in the BMI ≥ 20 kg/m 2 range with a few exceptions. In consequence, our sample of REC represents clinically stable long-term recovered individuals, which seems important in order to avoid cerebral aberrations and dysfunction—which might still be present after short term—due to insufficient clinical recovery.
However, most previous studies used such recovery criteria (see Additional file 1 ), and we could not detect any pattern with respect to the rather divergent study results.
We aimed to put behavioral and cerebral response to visual food cues in the context of state and trait. In contrast to AN, REC participants did not differ psychometrically or in their valence and arousal ratings to food stimuli from NP, indicating unimpaired cognitive and emotional processing of food stimuli. This is accompanied by a cerebral neural activation pattern which is similar to NP, i.e. which is not different between groups, even when applying a very lenient initial height threshold statistically. These data are comparable to two recent studies using a food paradigm [ 21 , 23 ].
In contrast to previous studies [ 22 , 25 ], correlation and interaction analysis showed no significant results. However, although our study has a larger sample size than the previous ones [ 22 , 25 ], it is possible that the effects are still too small to be detected with the group size of this study [ 70 ].
Comparing REC and AN, experiential data differed. The BOLD responses showed no regions of higher activation of REC as suggested by Uher et al. [ 24 ]. Applying a lenient initial height threshold, we however also found increased activation in occipital areas in AN when compared to REC, but this was located on the left side, while in the other study it was right-sided [ 24 ]. With respect to the meaning of possible occipital differences, these do not represent brain areas associated with emotional and motivational processing, but visual processing and might be unspecific [ 43 ]. Finally, we point to a recent report on neurobiological markers of 55 REC, which did not find differences between NP and REC, which can be regarded as a complementary finding to our results [ 71 ].
Not only neuronal and glial damage appear to be state phenomena [ 4 , 71 ] but, according to our results, neural processing and affective rating of food stimuli also seem to be state-related. Certain neuropsychological characteristics seem to persist in some patients with AN after clinical recovery, such as difficulties in set-shifting abilities and weaker central coherence [ 72 ]. Consequently, functional cerebral aberrations are likely no candidates for endophenotypes in AN.
The study has limitations. REC were slightly older than the other groups, which is due to the course of the illness. When including age as a covariate, the results remained significant. Larger cohorts and in particular longitudinal intra-individual designs will complement our knowledge — ideally through repeated measurements. However, AN is a disease with low prevalence and high chronicity, and therefore it is difficult to recruit large enough samples — in particular in single-center studies. A further limitation of the current study represents that AN consumed less calories before the imaging protocol — which, however, is also difficult to compensate/equalize methodically. Due to the differences in calorie intake between AN and REC/NP, food processing might be due to a temporary situation of hunger during measurement and not representing a stable finding in acute AN. Although our findings suggest a restitution of brain function with regard to food stimuli, the paradigm might not be sensitive enough to detect weaker effects. Furthermore, it cannot be ruled out that there may be other areas where abnormalities persist, such as in the perception of one's own body.
In summary, similar to the restitution of structural cerebral abnormalities [ 5 , 20 ] and serum neuronal biomarkers [ 71 ], functional brain aberrations also seem to be a state phenomenon , at least in terms of processing of food stimuli. However, this should be proven experimentally by longitudinal studies in AN and larger cohorts, which is not easily feasible methodically. Replicability is affected by several methodological issues, which we discussed in more detail elsewhere [ 45 ]. From a clinical perspective, the restitution of structural and functional cerebral alterations is an interesting issue concerning the transition of neuroscientific knowledge into clinical practice [ 73 ].
Availability of data and materials
Data are available from the senior authors on reasonable request.
Abbreviations
- Anorexia nervosa
Beck depression inventory
Blood oxygenation level dependent
Body mass index
Eating disorder examination
Eating disorder inventory
- Functional magnetic resonance imaging
Field of view
Magnetization-prepared rapid gradient echo
Montreal Neurological Institute
Multiple choice vocabulary test
Non-patients
Structured clinical interview for DSM
Statistical parametric mapping
State trait anxiety inventory
Repetition time
Steinhausen H-C. The outcome of anorexia nervosa in the 20th century. Am J Psychiatry. 2002;159:1284–93.
Article PubMed Google Scholar
Zipfel S, Giel KE, Bulik CM, Hay P, Schmidt U. Anorexia nervosa: aetiology, assessment, and treatment. Lancet Psychiatry. 2015;2:1099–111.
Treasure J, Schmidt U. The cognitive-interpersonal maintenance model of anorexia nervosa revisited: a summary of the evidence for cognitive, socio-emotional and interpersonal predisposing and perpetuating factors. J Eat Disord. 2013;1:13.
Article PubMed PubMed Central Google Scholar
Seitz J, Herpertz-Dahlmann B, Konrad K. Brain morphological changes in adolescent and adult patients with anorexia nervosa. J Neural Transm. 2016;123:949–59.
Nickel K, Joos A, van Elst LT, Matthis J, Holovics L, Endres D, et al. Recovery of cortical volume and thickness after remission from acute anorexia nervosa. Int J Eat Disord. 2018;51:1056–69.
Simon JJ, Stopyra MA, Friederich H-C. Neural processing of disorder-related stimuli in patients with anorexia nervosa: a narrative review of brain imaging studies. J Clin Med [Internet]. 2019 [cited 2020 Apr 25];8. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6678397/
Steward T, Menchón JM, Jiménez-Murcia S, Soriano-Mas C, Fernández-Aranda F. Neural network alterations across eating disorders: a narrative review of fMRI studies. Curr Neuropharmacol. 2018;16:1150–63.
Su T, Gong J, Tang G, Qiu S, Chen P, Chen G, et al. Structural and functional brain alterations in anorexia nervosa: a multimodal meta-analysis of neuroimaging studies. Hum Brain Mapp. 2021;42:5154–69.
King JA, Frank GKW, Thompson PM, Ehrlich S. Structural neuroimaging of anorexia nervosa: future directions in the quest for mechanisms underlying dynamic alterations. Biol Psychiatry. 2018;83:224–34.
Gottesman II, Gould TD. The endophenotype concept in psychiatry: etymology and strategic intentions. AJP Am Psychiatr Publ. 2003;160:636–45.
Article Google Scholar
Erdur L, Weber C, Zimmermann-Viehoff F, Rose M, Deter H-C. Affective responses in different stages of anorexia nervosa: results from a startle-reflex paradigm. Eur Eat Disord Rev. 2017;25:114–22.
Gárriz M, Andrés-Perpiñá S, Plana MT, Flamarique I, Romero S, Julià L, et al. Personality disorder traits, obsessive ideation and perfectionism 20 years after adolescent-onset anorexia nervosa: a recovered study. Eat Weight Disord. 2021;26:667–77.
Kodama N, Moriguchi Y, Takeda A, Maeda M, Ando T, Kikuchi H, et al. Neural correlates of body comparison and weight estimation in weight-recovered anorexia nervosa: a functional magnetic resonance imaging study. Biopsychosoc Med [Internet]. 2018 [cited 2020 Apr 25];12. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6208027/
Maier S, Nickel K, Perlov E, Kukies A, Zeeck A, Tebartz van Elst L, et al. Insular cell integrity markers linked to weight concern in anorexia nervosa—an MR-spectroscopy study. J Clin Med [Internet]. 2020 [cited 2020 Nov 10];9. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7288299/
von Schwanenflug N, von Müller DK, King JA, Ritschel F, Bernardoni F, Mohammadi S, et al. Dynamic changes in white matter microstructure in anorexia nervosa: findings from a longitudinal study. Psychol Med. 2019;49:1555–64.
Bentz M, Jepsen JRM, Kjaersdam Telléus G, Moslet U, Pedersen T, Bulik CM, et al. Neurocognitive functions and social functioning in young females with recent-onset anorexia nervosa and recovered individuals. J Eat Disord. 2017;5:5.
Kanakam N, Treasure J. A review of cognitive neuropsychiatry in the taxonomy of eating disorders: State, trait, or genetic? Cogn Neuropsychiatry. 2013;18:83–114.
Miles S, Gnatt I, Phillipou A, Nedeljkovic M. Cognitive flexibility in acute anorexia nervosa and after recovery: A systematic review. Clin Psychol Rev. 2020;81:101905.
Nickel K, Tebartz van Elst L, Holovics L, Feige B, Glauche V, Fortenbacher T, et al. White matter abnormalities in the corpus callosum in acute and recovered anorexia nervosa patients—a diffusion tensor imaging study. Front Psychiatry [Internet]. 2019 [cited 2020 Nov 10];10. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6628864/
Seitz J, Konrad K, Herpertz-Dahlmann B. Extend, pathomechanism and clinical consequences of brain volume changes in anorexia nervosa. Curr Neuropharmacol. 2018;16:1164–73.
Boehm I, Mohr H, King JA, Steding J, Geisler D, Wronski M-L, et al. Aberrant neural representation of food stimuli in women with acute anorexia nervosa predicts treatment outcome and is improved in weight restored individuals. Transl Psychiatry. 2021;11:1–7.
Oberndorfer TA, Simmons A, McCurdy D, Strigo I, Matthews S, Yang T, et al. Greater anterior insula activation during anticipation of food images in women recovered from anorexia nervosa versus controls. Psychiatry Res [Internet]. 2013 [cited 2020 Jul 9];214. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3880160/
Scaife JC, Godier LR, Reinecke A, Harmer CJ, Park RJ. Differential activation of the frontal pole to high vs low calorie foods: the neural basis of food preference in anorexia nervosa? Psychiatry Res. 2016;258:44–53.
Article PubMed Central Google Scholar
Uher R, Brammer MJ, Murphy T, Campbell IC, Ng VW, Williams SCR, et al. Recovery and chronicity in anorexia nervosa: brain activity associated with differential outcomes. Biol Psychiatry. 2003;54:934–42.
Holsen LM, Lawson EA, Blum J, Ko E, Makris N, Fazeli PK, et al. Food motivation circuitry hypoactivation related to hedonic and nonhedonic aspects of hunger and satiety in women with active anorexia nervosa and weight-restored women with anorexia nervosa. J Psychiatry Neurosci. 2012;37:322–32.
Sanders N, Smeets PAM, van Elburg AA, Danner UN, van Meer F, Hoek HW, et al. Altered food-cue processing in chronically Ill and recovered women with anorexia nervosa. Front Behav Neurosci [Internet]. 2015 [cited 2020 Apr 23];9. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4342866/
Cowdrey FA, Park RJ, Harmer CJ, McCabe C. Increased neural processing of rewarding and aversive food stimuli in recovered anorexia nervosa. Biol Psychiatry. 2011;70:736–43.
Kaye WH, Wierenga CE, Bischoff-Grethe A, Berner LA, Ely AV, Bailer UF, et al. Neural insensitivity to the effects of hunger in women remitted from anorexia nervosa. AJP. 2020;177:601–10.
Oberndorfer TA, Frank GKW, Simmons AN, Wagner A, McCurdy D, Fudge JL, et al. Altered insula response to sweet taste processing after recovery from anorexia and bulimia nervosa. Am J Psychiatry. 2013;170:1143–51.
Wagner A, Aizenstein H, Mazurkewicz L, Fudge J, Frank GK, Putnam K, et al. Altered insula response to taste stimuli in individuals recovered from restricting-type anorexia nervosa. Neuropsychopharmacology. 2008;33:513–23.
Frank GKW, Shott ME, Keffler C, Cornier M-A. Extremes of eating are associated with reduced neural taste discrimination. Int J Eat Disord. 2016;49:603–12.
Radeloff D, Willmann K, Otto L, Lindner M, Putnam K, Leeuwen SV, et al. High-fat taste challenge reveals altered striatal response in women recovered from bulimia nervosa: a pilot study. World J Biol Psychiatry. 2014;15:307–16.
King JA, Bernardoni F, Geisler D, Ritschel F, Doose A, Pauligk S, et al. Intact value-based decision-making during intertemporal choice in women with remitted anorexia nervosa? An fMRI study. J Psychiatry Neurosci. 2020;45:108–16.
Maier S, Schneider K, Stark C, Zeeck A, Tebartz van Elst L, Holovics L, et al. Fear network unresponsiveness in women with anorexia nervosa. PPS. 2019;88:238–40.
Google Scholar
Maier S, Spiegelberg J, van Zutphen L, Zeeck A, van Elst LT, Hartmann A, et al. Neurobiological signature of intimacy in anorexia nervosa. Eur Eat Disord Rev. 2019;27:315–22.
Olsavsky AK, Shott ME, DeGuzman MC, Frank GKW. Neural correlates of taste reward value across eating disorders. Psychiatry Res Neuroimaging. 2019;288:76–84.
Decker JH, Figner B, Steinglass JE. On weight and waiting: delay discounting in anorexia nervosa pre- and post-treatment. Biol Psychiatry. 2015;78:606–14.
Doose A, King JA, Bernardoni F, Geisler D, Hellerhoff I, Weinert T, et al. Strengthened default mode network activation during delay discounting in adolescents with anorexia nervosa after partial weight restoration: a longitudinal fMRI study. J Clin Med. 2020;9:900.
Castro-Fornieles J, de la Serna E, Calvo A, Blázquez A, Moya J, Lázaro L, et al. Functional MRI with a set-shifting task in adolescent anorexia nervosa: a cross-sectional and follow-up study. Neuropsychologia. 2019;131:1–8.
Castro-Fornieles J, Caldú X, Andrés-Perpiñá S, Lázaro L, Bargalló N, Falcón C, et al. A cross-sectional and follow-up functional MRI study with a working memory task in adolescent anorexia nervosa. Neuropsychologia. 2010;48:4111–6.
DeGuzman M, Shott ME, Yang TT, Riederer J, Frank GKW. Association of elevated reward prediction error response with weight gain in adolescent anorexia nervosa. AJP. 2017;174:557–65.
Schulte-Rüther M, Mainz V, Fink GR, Herpertz-Dahlmann B, Konrad K. Theory of mind and the brain in anorexia nervosa: relation to treatment outcome. J Am Acad Child Adolesc Psychiatry. 2012;51:832-841.e11.
Uher R, Murphy T, Brammer MJ, Dalgleish T, Phillips ML, Ng VW, et al. Medial Prefrontal cortex activity associated with symptom provocation in eating disorders. AJP. 2004;161:1238–46.
Joos AAB, Saum B, van Elst LT, Perlov E, Glauche V, Hartmann A, et al. Amygdala hyperreactivity in restrictive anorexia nervosa. Psychiatry Research: Neuroimaging. 2011;191:189–95.
Horster I, Nickel K, Holovics L, Schmidt S, Endres D, Tebartz van Elst L, et al. A Neglected topic in neuroscience: replicability of fMRI results with specific reference to ANOREXIA NERVOSA. Front Psychiatry [Internet]. Frontiers; 2020 [cited 2020 Sep 22];11. Available from: https://www.frontiersin.org/articles/10.3389/fpsyt.2020.00777/full?&utm_source=Email_to_authors_&utm_medium=Email&utm_content=T1_11.5e1_author&utm_campaign=Email_publication&field=&journalName=Frontiers_in_Psychiatry&id=567828
Giel KE, Conzelmann A, Renner TJ, Richter T, Benito SM, Zipfel S, et al. Attention allocation to illness-compatible information discriminates women with active versus weight-recovered anorexia nervosa. Int J Eat Disord. 2020;53:1270–9.
Hilbert A, Tuschen-Caffier B, Ohms M. Eating disorder examination: Deutschsprachige Version des strukturierten Essstörungsinterviews. Diagnostica Hogrefe Verlag. 2004;50:98–106.
Fydrich T, Renneberg B, Schmitz B, Wittchen H-U. SKID-II Strukturiertes klinisches interview für DSM-IV: Achse II: Persönlichkeitsstörungen: Interviewheft. Göttingen: Hogrefe; 1997. p. 36.
Wittchen H-U, Zaudig M, Fydrich T. Strukturiertes Klinisches interview für DSM-IV. Göttingen: Hogrefe; 1997.
Hilbert A, Tuschen-Caffier B, Karwautz A, Niederhofer H, Munsch S. Eating disorder examination-questionnaire. Diagnostica. 2007;53:144–54.
Hautzinger M, Keller F, Kühner C, Beck AT. Beck depressions-inventar BDI II.Revision [Internet]. Revision. Frankfurt am Main: Harcourt Test Services; 2006. Available from: https://katalog.ub.uni-freiburg.de/link?kid=1342959973
Paul T, Thiel A. Eating disorder inventory-2—deutsche Fassung. Göttingen: Hogrefe [Internet]. 2005 [cited 2021 Jan 26]; Available from: http://www.redi-bw.de/db/ebsco.php/search.ebscohost.com/login.aspx%3fdirect%3dtrue%26db%3dpdx%26AN%3dPT9003604%26site%3dehost-live
Laux L, Spielberger CD. Das state-trait-angstinventar STAI [Internet]. Göttingen: Beltz; 2001. Available from: https://katalog.ub.uni-freiburg.de/link?kid=1613862806
Lehrl S. Mehrfachwahl-Wortschatz-Intelligenztest MWT-B [Internet]. 5., unveränd. Aufl. Balingen: Spitta; 2005. Available from: https://katalog.ub.uni-freiburg.de/link?kid=1612168906
Bradley MM, Lang PJ. International affective picture system. In: Zeigler-Hill V, Shackelford TK, editors. Encyclopedia of personality and individual differences [Internet]. Cham: Springer; 2017. p. 1–4. https://doi.org/10.1007/978-3-319-28099-8_42-1 .
Chapter Google Scholar
Zaitsev M, Hennig J, Speck O. Point spread function mapping with parallel imaging techniques and high acceleration factors: fast, robust, and flexible method for echo-planar imaging distortion correction. Magn Reson Med. 2004;52:1156–66.
Friston KJ, Holmes AP, Worsley KJ, Poline J-P, Frith CD, Frackowiak RSJ. Statistical parametric maps in functional imaging: a general linear approach. Hum Brain Mapp. 1994;2:189–210.
Rolls ET, Huang C-C, Lin C-P, Feng J, Joliot M. Automated anatomical labelling atlas 3. Neuroimage. 2020;206:116189.
Roiser JP, Linden DE, Gorno-Tempini ML, Moran RJ, Dickerson BC, Grafton ST. Minimum statistical standards for submissions to Neuroimage: Clinical. NeuroImage Clin. 2016;12:1045–7.
Eklund A, Knutsson H, Nichols TE. Cluster failure revisited: Impact of first level design and physiological noise on cluster false positive rates. Hum Brain Mapp. 2019;40:2017–32.
Woo C-W, Krishnan A, Wager TD. Cluster-extent based thresholding in fMRI analyses: pitfalls and recommendations. Neuroimage. 2014;91:412–9.
Frank GKW, Favaro A, Marsh R, Ehrlich S, Lawson EA. Toward valid and reliable brain imaging results in eating disorders. Int J Eat Disord. 2018;51:250–61.
Hüffmeier J, Mazei J, Schultze T. Reconceptualizing replication as a sequence of different studies: a replication typology. J Exp Soc Psycholo. 2016;66:9–10.
Bennett CM, Miller MB. How reliable are the results from functional magnetic resonance imaging? Ann NY Acad Sci. 2010;1191:133–55.
Gorgolewski KJ, Poldrack RA. A practical guide for improving transparency and reproducibility in neuroimaging research. PLOS Biol. 2016;14:e1002506.
Bardone-Cone AM, Harney MB, Maldonado CR, Lawson MA, Robinson DP, Smith R, et al. Defining recovery from an eating disorder: conceptualization, validation, and examination of psychosocial functioning and psychiatric comorbidity. Behav Res Ther. 2010;48:194–202.
Wade TD, Lock J. Developing consensus on the definition of remission and recovery for research. Int J Eat Disord. 2020;53:1204–8.
Couturier J, Lock J. What is remission in adolescent anorexia nervosa? A review of various conceptualizations and quantitative analysis. Int J Eat Disord. 2006;39:175–83.
Khalsa SS, Portnoff LC, McCurdy-McKinnon D, Feusner JD. What happens after treatment? A systematic review of relapse, remission, and recovery in anorexia nervosa. J Eat Disord [Internet]. 2017 [cited 2020 Nov 3];5. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5470198/
Elliott ML, Knodt AR, Ireland D, Morris ML, Poulton R, Ramrakha S, et al. What is the test-retest reliability of common task-functional MRI measures? New empirical evidence and a meta-analysis. Psychol Sci. 2020;31:792–806.
Doose A, Hellerhoff I, Tam FI, King JA, Seidel M, Geisler D, et al. Neural and glial damage markers in women after long-term weight-recovery from anorexia nervosa. Psychoneuroendocrinology. 2022;135:105576.
Fuglset TS. Set-shifting, central coherence and decision-making in individuals recovered from anorexia nervosa: a systematic review. J Eat Disord. 2019;7:22.
Bang L, Treasure J, Rø Ø, Joos A. Advancing our understanding of the neurobiology of anorexia nervosa: translation into treatment. J Eat Disord. 2017;5:38.
Download references
Open Access funding enabled and organized by Projekt DEAL. German Research Foundation, JO 744/2-1 (Principal Investigator: A. Joos).
Author information
Selma Göller and Kathrin Nickel contributed equally as first authors
Simon Maier and Andreas A. B. Joos contributed equally as senior authors
Authors and Affiliations
Department of Psychosomatic Medicine and Psychotherapy, Medical Center - University of Freiburg, Faculty of Medicine, University of Freiburg, Freiburg, Germany
Selma Göller, Isabelle Horster, Almut Zeeck, Claas Lahmann & Andreas A. B. Joos
Department of Psychiatry and Psychotherapy, Medical Center - University of Freiburg, Faculty of Medicine, University of Freiburg, Freiburg, Germany
Kathrin Nickel, Dominique Endres, Katharina Domschke, Ludger Tebartz van Elst & Simon Maier
Department of Psychosomatic Medicine and Psychotherapy, Ortenau Klinikum, Lahr, Academic Teaching Hospital of the University of Freiburg, Lahr, Germany
Andreas A. B. Joos
Center for Basics in Neuromodulation, Faculty of Medicine, University of Freiburg, Freiburg, Germany
Katharina Domschke
You can also search for this author in PubMed Google Scholar
Contributions
All authors contributed to and reviewed the study and manuscript and gave consent to its publication. All authors approved the final manuscript.
Corresponding author
Correspondence to Kathrin Nickel .
Ethics declarations
Ethics approval and consent to participate.
The study was approved by the Ethics Committee of the University of Freiburg (EK 520/13). Study participants gave written informed consent to participate in the study.
Consent for publication
All authors gave consent to the publication of the manuscript.
Competing interests
SG: no conflict of interest, KN: no conflict of interest, IH: no conflict of interest, DE: no conflict of interest, AZ: no conflict of interest, KD: Member of the ‘Steering Committee Neurosciences’, Janssen Pharmaceuticals, Inc., CL: no conflict of interest, LTvE: Advisory boards, lectures, or travel grants within the last three years: Roche, Eli Lilly, Janssen-Cilag, Novartis, Shire, UCB, GSK, Servier, Janssen and Cyberonics, SM: no conflict of interest, AJ: no conflict of interest.
Additional information
Publisher's note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
file 1: Overview of previous studies.
file 2: Results of between-group differences for the contrast food > non-food.
file 3: Results of the linear regression models for the affective ratings.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Göller, S., Nickel, K., Horster, I. et al. State or trait: the neurobiology of anorexia nervosa — contributions of a functional magnetic resonance imaging study. J Eat Disord 10 , 77 (2022). https://doi.org/10.1186/s40337-022-00598-7
Download citation
Received : 19 March 2022
Accepted : 23 May 2022
Published : 31 May 2022
DOI : https://doi.org/10.1186/s40337-022-00598-7
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
Journal of Eating Disorders
ISSN: 2050-2974
- General enquiries: [email protected]
Anastasiya D. KOROTENKOVA’s research while affiliated with Astrakhan State Technical University and other places
What is this page, publications (3).
January 2022
Finance and Credit

Subject. The article addresses risk diagnostics in the activities of organizations of agro-industrial complex in the Astrakhan Oblast. Objectives. The focus is to develop practical recommendations aimed at minimizing the risks of organizations operating in the agro-industrial complex of the Astrakhan Oblast. Methods. The study rests on the analysis of data provided by the State Statistics Service, the Ministry of Agriculture and Fishing Industry of the Astrakhan Oblast and the calculation of indicators based on these data. We apply the indicative method and calculation of integrated index. Results. We analyzed the main socio-economic indicators and diagnosed risks, using a risk map. The indicative method and the calculation of integrated indicator helped specify a low (pre-crisis) level of economic security in the agro-industrial complex for three years under study. To mitigate the identified risks we proposed measures to be taken by the government, like farm business concessional loans scheme, careful selection of creditors and borrowers, support to unprofitable farms, taking into account the WTO rules. Conclusions. The findings enable to analyze the current state of agriculture in the Astrakhan Oblast and develop the areas of risk mitigation in the agro-industrial complex. It is crucial to stabilize the financial condition through stock reduction, improve the supply chain process, the system of warehouses and storage facilities.
October 2021
Subject. We consider risks to economic security in procurement of a budgetary institution. Objectives. The focus is on diagnostics and monitoring of economic security in procurement for subsequent elaboration of recommendations to minimize the impact of threats in the process of ensuring economic security. Methods. The study draws on the analysis of data from the official website of the ASTU and the Unified Information System for Procurement. We employ statistical and analytical methods to assess the probability of occurrence of adverse events (risk realization). Results. We reviewed procurement activity of a budgetary institution, produced a map indicating the risks of a budgetary institution, calculated the indicators of economic security in procurement of this type of institutions. Conclusions. The analysis identified weaknesses of a budgetary institution and risk exposure in procurement. Control and monitoring of the process and its results will enable contractual procurement system to operate with greater stability in terms of economic security. Preliminary control, affecting the effectiveness and efficiency of procurement by means of quality, will also have a great impact. Quality products, works or services are the key to effective procurement.
August 2021
Subject. The article considers the economic relationship between the banking sector and the population. Objectives. We focus on theoretical and empirical analysis of the activities of PAO Sberbank, identifying threats and measures to minimize any damage to the banking organization. Methods. The study rests on the analysis of data provided by PAO Sberbank on its official website. Our calculation of indicators bases on these data. The main methods employed are expert assessments, the method of threshold values of the main threats to economic security. Results. We performed a horizontal and vertical analysis of the financial statements of PAO Sberbank, considered the structure of income and expenses and the bank's loan portfolio by sector of economy. Using the threshold value method, we unveiled the threats inherent in PAO Sberbank and developed a strategy to minimize the identified threats. The findings may be useful for analyzing the bank’s activities and identifying the threats to the banking sector in a socially oriented market economy. Conclusions. The study reveales that this bank is inherent in such threats as low short-term liquidity and loan impairment risk. We offer a strategy that affects the policy of PAO Sberbank. It includes long-term measures to increase the share of mortgage loans, which, as a result, will enable the development of the construction sector and have a positive effect on society as a whole.

IMAGES
COMMENTS
Anorexia nervosa is a food intake disorder. characterized by a cute weight loss that it could cause. severe psychosomatic problems [1]. Diagnostic criteria for Anorexia nervosa. include an intense ...
anorexia nervosa can be described as a significant loss of weight for age, gender, developmental. pathway, and physical health issue developed due to a restriction of energy intake for body ...
Anorexia Nervosa, often referred to as anorexia, is a complex. and potentially life-threatening eating disorder that a ects mil. lions of individuals worldwide. Characterized by an extreme ...
Abstract. Anorexia nervosa is a complex psychiatric illness associated with food restriction and high mortality. Recent brain research in adolescents and adults with anorexia nervosa has used larger sample sizes compared with earlier studies and tasks that test specific brain circuits. Those studies have produced more robust results and ...
For bulimia nervosa, there has been a decline in overall incidence rate over time. The lifetime prevalence rates of anorexia nervosa might be up to 4% among females and 0.3% among males. Regarding bulimia nervosa, up to 3% of females and more than 1% of males suffer from this disorder during their lifetime.
The Clinical Problem. Anorexia nervosa is a severe psychiatric disorder that is characterized by starvation and malnutrition, a high incidence of coexisting psychiatric conditions, treatment ...
Anorexia nervosa (AN) is classically defined as a condition in which an abnormally low body weight is associated with an intense fear of gaining weight and distorted cognitions regarding weight, shape, and drive for thinness. This article reviews recent evidences from physiology, genetics, epigenetics, and brain imaging which allow to consider ...
Objective: Currently, there is debate in the eating disorders field regarding how to define atypical anorexia (AAN), how prevalent it is in community and clinical settings, and how AAN rates compare with low-weight AN. This systematic review assesses AAN literature from 2007 to 2020, to investigate: (a) the demographic characteristics of AAN studies, (b) the prevalence of AAN compared with AN ...
Anorexia nervosa, as a psychiatric disorder with a progressively high prevalence, is characterized by the following features: high mortality rate, serious harm, long-lasting effects, difficult to ...
Anorexia nervosa is associated with high personal and economic costs for sufferers, their relatives and society. Evidence-based practice guidelines aim to support all groups involved in the care of patients with anorexia nervosa by providing them with scientifically sound recommendations regarding diagnosis and treatment. The German S3 ...
Background: The understanding of the cerebral neurobiology of anorexia nervosa (AN) with respect to state- versus trait-related abnormalities is limited. There is evidence of restitution of structural brain alterations with clinical remission. However, with regard to functional brain abnormalities, this issue has not yet been clarified.
The understanding of the cerebral neurobiology of anorexia nervosa (AN) with respect to state- versus trait-related abnormalities is limited. There is evidence of restitution of structural brain alterations with clinical remission. However, with regard to functional brain abnormalities, this issue has not yet been clarified. We compared women with AN (n = 31), well-recovered female ...
Anorexia nervosa (AN) is characterized by restricted caloric intake and is associated with low body weight, a fear of gaining weight or becoming fat, and, often, a distorted body image [1]. It ...
Methods: Females with DSM-III-R/DSM-IV anorexia nervosa or bulimia nervosa were assessed at 9 and at 20 to 25 years of follow-up (mean [SD] = 22.10 [1.10] years; study initiated in 1987, last ...
Anorexia nervosa is an eating disorder condition characterized by an abnormal fear of gaining weight, driving people to starve themselves and become dangerously thin. It involves restricting food ...
Various forms of eating disorder focused family therapy (FT-ED) have been developed and evaluated for adolescents. FT-ED for adolescent anorexia nervosa (AN) focuses on empowering parents/carers ...
Characterized primarily by a low body-mass index, anorexia nervosa is a complex and serious illness¹, affecting 0.9-4% of women and 0.3% of men2-4, with twin-based heritability estimates of ...
Abstract. Anorexia nervosa is associated with a high incidence of coexisting psychiatric conditions, marked treatment resistance, frequent medical complications, and a substantial risk of death ...
anorexia nervosa include women in the age ranges 13-14 years old and the 18-20 years old. Studies continue to support this research, noting that 95% of those who have eating disorders. are between ...
Join ResearchGate to find the people and research you need to help your work. 25+ million members; 160+ million publication pages; 2.3+ billion citations; Join for free. Top co-authors.
Join ResearchGate to find the people and research you need to help your work. 25+ million members; 160+ million publication pages; 2.3+ billion citations; Join for free. or. Discover by subject area.
Subject. The article considers theoretical aspects of nominal, accrued, real, and disposable wages and savings in the Astrakhan Oblast in various areas of economic activity.
Anastasiya D. KOROTENKOVA's 3 research works with 7 reads, including: Analytical methods and tools for risk diagnostics in the activities of agricultural organizations