

Qualitative Data Analysis Methods 101:
The “big 6” methods + examples.
By: Kerryn Warren (PhD) | Reviewed By: Eunice Rautenbach (D.Tech) | May 2020 (Updated April 2023)
Qualitative data analysis methods. Wow, that’s a mouthful.
If you’re new to the world of research, qualitative data analysis can look rather intimidating. So much bulky terminology and so many abstract, fluffy concepts. It certainly can be a minefield!
Don’t worry – in this post, we’ll unpack the most popular analysis methods , one at a time, so that you can approach your analysis with confidence and competence – whether that’s for a dissertation, thesis or really any kind of research project.

What (exactly) is qualitative data analysis?
To understand qualitative data analysis, we need to first understand qualitative data – so let’s step back and ask the question, “what exactly is qualitative data?”.
Qualitative data refers to pretty much any data that’s “not numbers” . In other words, it’s not the stuff you measure using a fixed scale or complex equipment, nor do you analyse it using complex statistics or mathematics.
So, if it’s not numbers, what is it?
Words, you guessed? Well… sometimes , yes. Qualitative data can, and often does, take the form of interview transcripts, documents and open-ended survey responses – but it can also involve the interpretation of images and videos. In other words, qualitative isn’t just limited to text-based data.
So, how’s that different from quantitative data, you ask?
Simply put, qualitative research focuses on words, descriptions, concepts or ideas – while quantitative research focuses on numbers and statistics . Qualitative research investigates the “softer side” of things to explore and describe , while quantitative research focuses on the “hard numbers”, to measure differences between variables and the relationships between them. If you’re keen to learn more about the differences between qual and quant, we’ve got a detailed post over here .
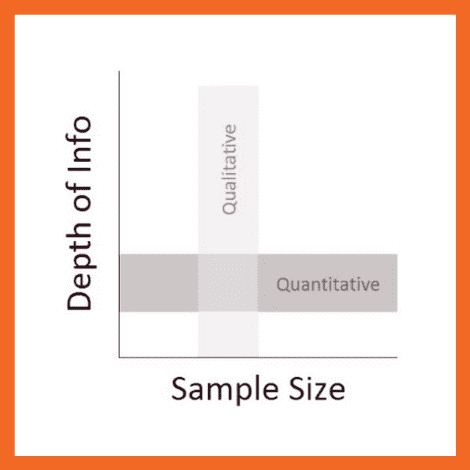
So, qualitative analysis is easier than quantitative, right?
Not quite. In many ways, qualitative data can be challenging and time-consuming to analyse and interpret. At the end of your data collection phase (which itself takes a lot of time), you’ll likely have many pages of text-based data or hours upon hours of audio to work through. You might also have subtle nuances of interactions or discussions that have danced around in your mind, or that you scribbled down in messy field notes. All of this needs to work its way into your analysis.
Making sense of all of this is no small task and you shouldn’t underestimate it. Long story short – qualitative analysis can be a lot of work! Of course, quantitative analysis is no piece of cake either, but it’s important to recognise that qualitative analysis still requires a significant investment in terms of time and effort.
Need a helping hand?
In this post, we’ll explore qualitative data analysis by looking at some of the most common analysis methods we encounter. We’re not going to cover every possible qualitative method and we’re not going to go into heavy detail – we’re just going to give you the big picture. That said, we will of course includes links to loads of extra resources so that you can learn more about whichever analysis method interests you.
Without further delay, let’s get into it.
The “Big 6” Qualitative Analysis Methods
There are many different types of qualitative data analysis, all of which serve different purposes and have unique strengths and weaknesses . We’ll start by outlining the analysis methods and then we’ll dive into the details for each.
The 6 most popular methods (or at least the ones we see at Grad Coach) are:
- Content analysis
- Narrative analysis
- Discourse analysis
- Thematic analysis
- Grounded theory (GT)
- Interpretive phenomenological analysis (IPA)
Let’s take a look at each of them…
QDA Method #1: Qualitative Content Analysis
Content analysis is possibly the most common and straightforward QDA method. At the simplest level, content analysis is used to evaluate patterns within a piece of content (for example, words, phrases or images) or across multiple pieces of content or sources of communication. For example, a collection of newspaper articles or political speeches.
With content analysis, you could, for instance, identify the frequency with which an idea is shared or spoken about – like the number of times a Kardashian is mentioned on Twitter. Or you could identify patterns of deeper underlying interpretations – for instance, by identifying phrases or words in tourist pamphlets that highlight India as an ancient country.
Because content analysis can be used in such a wide variety of ways, it’s important to go into your analysis with a very specific question and goal, or you’ll get lost in the fog. With content analysis, you’ll group large amounts of text into codes , summarise these into categories, and possibly even tabulate the data to calculate the frequency of certain concepts or variables. Because of this, content analysis provides a small splash of quantitative thinking within a qualitative method.
Naturally, while content analysis is widely useful, it’s not without its drawbacks . One of the main issues with content analysis is that it can be very time-consuming , as it requires lots of reading and re-reading of the texts. Also, because of its multidimensional focus on both qualitative and quantitative aspects, it is sometimes accused of losing important nuances in communication.
Content analysis also tends to concentrate on a very specific timeline and doesn’t take into account what happened before or after that timeline. This isn’t necessarily a bad thing though – just something to be aware of. So, keep these factors in mind if you’re considering content analysis. Every analysis method has its limitations , so don’t be put off by these – just be aware of them ! If you’re interested in learning more about content analysis, the video below provides a good starting point.
QDA Method #2: Narrative Analysis
As the name suggests, narrative analysis is all about listening to people telling stories and analysing what that means . Since stories serve a functional purpose of helping us make sense of the world, we can gain insights into the ways that people deal with and make sense of reality by analysing their stories and the ways they’re told.
You could, for example, use narrative analysis to explore whether how something is being said is important. For instance, the narrative of a prisoner trying to justify their crime could provide insight into their view of the world and the justice system. Similarly, analysing the ways entrepreneurs talk about the struggles in their careers or cancer patients telling stories of hope could provide powerful insights into their mindsets and perspectives . Simply put, narrative analysis is about paying attention to the stories that people tell – and more importantly, the way they tell them.
Of course, the narrative approach has its weaknesses , too. Sample sizes are generally quite small due to the time-consuming process of capturing narratives. Because of this, along with the multitude of social and lifestyle factors which can influence a subject, narrative analysis can be quite difficult to reproduce in subsequent research. This means that it’s difficult to test the findings of some of this research.
Similarly, researcher bias can have a strong influence on the results here, so you need to be particularly careful about the potential biases you can bring into your analysis when using this method. Nevertheless, narrative analysis is still a very useful qualitative analysis method – just keep these limitations in mind and be careful not to draw broad conclusions . If you’re keen to learn more about narrative analysis, the video below provides a great introduction to this qualitative analysis method.
QDA Method #3: Discourse Analysis
Discourse is simply a fancy word for written or spoken language or debate . So, discourse analysis is all about analysing language within its social context. In other words, analysing language – such as a conversation, a speech, etc – within the culture and society it takes place. For example, you could analyse how a janitor speaks to a CEO, or how politicians speak about terrorism.
To truly understand these conversations or speeches, the culture and history of those involved in the communication are important factors to consider. For example, a janitor might speak more casually with a CEO in a company that emphasises equality among workers. Similarly, a politician might speak more about terrorism if there was a recent terrorist incident in the country.
So, as you can see, by using discourse analysis, you can identify how culture , history or power dynamics (to name a few) have an effect on the way concepts are spoken about. So, if your research aims and objectives involve understanding culture or power dynamics, discourse analysis can be a powerful method.
Because there are many social influences in terms of how we speak to each other, the potential use of discourse analysis is vast . Of course, this also means it’s important to have a very specific research question (or questions) in mind when analysing your data and looking for patterns and themes, or you might land up going down a winding rabbit hole.
Discourse analysis can also be very time-consuming as you need to sample the data to the point of saturation – in other words, until no new information and insights emerge. But this is, of course, part of what makes discourse analysis such a powerful technique. So, keep these factors in mind when considering this QDA method. Again, if you’re keen to learn more, the video below presents a good starting point.
QDA Method #4: Thematic Analysis
Thematic analysis looks at patterns of meaning in a data set – for example, a set of interviews or focus group transcripts. But what exactly does that… mean? Well, a thematic analysis takes bodies of data (which are often quite large) and groups them according to similarities – in other words, themes . These themes help us make sense of the content and derive meaning from it.
Let’s take a look at an example.
With thematic analysis, you could analyse 100 online reviews of a popular sushi restaurant to find out what patrons think about the place. By reviewing the data, you would then identify the themes that crop up repeatedly within the data – for example, “fresh ingredients” or “friendly wait staff”.
So, as you can see, thematic analysis can be pretty useful for finding out about people’s experiences , views, and opinions . Therefore, if your research aims and objectives involve understanding people’s experience or view of something, thematic analysis can be a great choice.
Since thematic analysis is a bit of an exploratory process, it’s not unusual for your research questions to develop , or even change as you progress through the analysis. While this is somewhat natural in exploratory research, it can also be seen as a disadvantage as it means that data needs to be re-reviewed each time a research question is adjusted. In other words, thematic analysis can be quite time-consuming – but for a good reason. So, keep this in mind if you choose to use thematic analysis for your project and budget extra time for unexpected adjustments.

QDA Method #5: Grounded theory (GT)
Grounded theory is a powerful qualitative analysis method where the intention is to create a new theory (or theories) using the data at hand, through a series of “ tests ” and “ revisions ”. Strictly speaking, GT is more a research design type than an analysis method, but we’ve included it here as it’s often referred to as a method.
What’s most important with grounded theory is that you go into the analysis with an open mind and let the data speak for itself – rather than dragging existing hypotheses or theories into your analysis. In other words, your analysis must develop from the ground up (hence the name).
Let’s look at an example of GT in action.
Assume you’re interested in developing a theory about what factors influence students to watch a YouTube video about qualitative analysis. Using Grounded theory , you’d start with this general overarching question about the given population (i.e., graduate students). First, you’d approach a small sample – for example, five graduate students in a department at a university. Ideally, this sample would be reasonably representative of the broader population. You’d interview these students to identify what factors lead them to watch the video.
After analysing the interview data, a general pattern could emerge. For example, you might notice that graduate students are more likely to read a post about qualitative methods if they are just starting on their dissertation journey, or if they have an upcoming test about research methods.
From here, you’ll look for another small sample – for example, five more graduate students in a different department – and see whether this pattern holds true for them. If not, you’ll look for commonalities and adapt your theory accordingly. As this process continues, the theory would develop . As we mentioned earlier, what’s important with grounded theory is that the theory develops from the data – not from some preconceived idea.
So, what are the drawbacks of grounded theory? Well, some argue that there’s a tricky circularity to grounded theory. For it to work, in principle, you should know as little as possible regarding the research question and population, so that you reduce the bias in your interpretation. However, in many circumstances, it’s also thought to be unwise to approach a research question without knowledge of the current literature . In other words, it’s a bit of a “chicken or the egg” situation.
Regardless, grounded theory remains a popular (and powerful) option. Naturally, it’s a very useful method when you’re researching a topic that is completely new or has very little existing research about it, as it allows you to start from scratch and work your way from the ground up .

QDA Method #6: Interpretive Phenomenological Analysis (IPA)
Interpretive. Phenomenological. Analysis. IPA . Try saying that three times fast…
Let’s just stick with IPA, okay?
IPA is designed to help you understand the personal experiences of a subject (for example, a person or group of people) concerning a major life event, an experience or a situation . This event or experience is the “phenomenon” that makes up the “P” in IPA. Such phenomena may range from relatively common events – such as motherhood, or being involved in a car accident – to those which are extremely rare – for example, someone’s personal experience in a refugee camp. So, IPA is a great choice if your research involves analysing people’s personal experiences of something that happened to them.
It’s important to remember that IPA is subject – centred . In other words, it’s focused on the experiencer . This means that, while you’ll likely use a coding system to identify commonalities, it’s important not to lose the depth of experience or meaning by trying to reduce everything to codes. Also, keep in mind that since your sample size will generally be very small with IPA, you often won’t be able to draw broad conclusions about the generalisability of your findings. But that’s okay as long as it aligns with your research aims and objectives.
Another thing to be aware of with IPA is personal bias . While researcher bias can creep into all forms of research, self-awareness is critically important with IPA, as it can have a major impact on the results. For example, a researcher who was a victim of a crime himself could insert his own feelings of frustration and anger into the way he interprets the experience of someone who was kidnapped. So, if you’re going to undertake IPA, you need to be very self-aware or you could muddy the analysis.

How to choose the right analysis method
In light of all of the qualitative analysis methods we’ve covered so far, you’re probably asking yourself the question, “ How do I choose the right one? ”
Much like all the other methodological decisions you’ll need to make, selecting the right qualitative analysis method largely depends on your research aims, objectives and questions . In other words, the best tool for the job depends on what you’re trying to build. For example:
- Perhaps your research aims to analyse the use of words and what they reveal about the intention of the storyteller and the cultural context of the time.
- Perhaps your research aims to develop an understanding of the unique personal experiences of people that have experienced a certain event, or
- Perhaps your research aims to develop insight regarding the influence of a certain culture on its members.
As you can probably see, each of these research aims are distinctly different , and therefore different analysis methods would be suitable for each one. For example, narrative analysis would likely be a good option for the first aim, while grounded theory wouldn’t be as relevant.
It’s also important to remember that each method has its own set of strengths, weaknesses and general limitations. No single analysis method is perfect . So, depending on the nature of your research, it may make sense to adopt more than one method (this is called triangulation ). Keep in mind though that this will of course be quite time-consuming.
As we’ve seen, all of the qualitative analysis methods we’ve discussed make use of coding and theme-generating techniques, but the intent and approach of each analysis method differ quite substantially. So, it’s very important to come into your research with a clear intention before you decide which analysis method (or methods) to use.
Start by reviewing your research aims , objectives and research questions to assess what exactly you’re trying to find out – then select a qualitative analysis method that fits. Never pick a method just because you like it or have experience using it – your analysis method (or methods) must align with your broader research aims and objectives.

Let’s recap on QDA methods…
In this post, we looked at six popular qualitative data analysis methods:
- First, we looked at content analysis , a straightforward method that blends a little bit of quant into a primarily qualitative analysis.
- Then we looked at narrative analysis , which is about analysing how stories are told.
- Next up was discourse analysis – which is about analysing conversations and interactions.
- Then we moved on to thematic analysis – which is about identifying themes and patterns.
- From there, we went south with grounded theory – which is about starting from scratch with a specific question and using the data alone to build a theory in response to that question.
- And finally, we looked at IPA – which is about understanding people’s unique experiences of a phenomenon.
Of course, these aren’t the only options when it comes to qualitative data analysis, but they’re a great starting point if you’re dipping your toes into qualitative research for the first time.
If you’re still feeling a bit confused, consider our private coaching service , where we hold your hand through the research process to help you develop your best work.

Psst... there’s more!
This post was based on one of our popular Research Bootcamps . If you're working on a research project, you'll definitely want to check this out ...
87 Comments
This has been very helpful. Thank you.
Thank you madam,
Thank you so much for this information
I wonder it so clear for understand and good for me. can I ask additional query?
Very insightful and useful
Good work done with clear explanations. Thank you.
Thanks so much for the write-up, it’s really good.
Thanks madam . It is very important .
thank you very good
Great presentation
This has been very well explained in simple language . It is useful even for a new researcher.
Great to hear that. Good luck with your qualitative data analysis, Pramod!
This is very useful information. And it was very a clear language structured presentation. Thanks a lot.
Thank you so much.
very informative sequential presentation
Precise explanation of method.
Hi, may we use 2 data analysis methods in our qualitative research?
Thanks for your comment. Most commonly, one would use one type of analysis method, but it depends on your research aims and objectives.
You explained it in very simple language, everyone can understand it. Thanks so much.
Thank you very much, this is very helpful. It has been explained in a very simple manner that even a layman understands
Thank nicely explained can I ask is Qualitative content analysis the same as thematic analysis?
Thanks for your comment. No, QCA and thematic are two different types of analysis. This article might help clarify – https://onlinelibrary.wiley.com/doi/10.1111/nhs.12048
This is my first time to come across a well explained data analysis. so helpful.
I have thoroughly enjoyed your explanation of the six qualitative analysis methods. This is very helpful. Thank you!
Thank you very much, this is well explained and useful
i need a citation of your book.
Thanks a lot , remarkable indeed, enlighting to the best
Hi Derek, What other theories/methods would you recommend when the data is a whole speech?
Keep writing useful artikel.
It is important concept about QDA and also the way to express is easily understandable, so thanks for all.
Thank you, this is well explained and very useful.
Very helpful .Thanks.
Hi there! Very well explained. Simple but very useful style of writing. Please provide the citation of the text. warm regards
The session was very helpful and insightful. Thank you
This was very helpful and insightful. Easy to read and understand
As a professional academic writer, this has been so informative and educative. Keep up the good work Grad Coach you are unmatched with quality content for sure.
Keep up the good work Grad Coach you are unmatched with quality content for sure.
Its Great and help me the most. A Million Thanks you Dr.
It is a very nice work
Very insightful. Please, which of this approach could be used for a research that one is trying to elicit students’ misconceptions in a particular concept ?
This is Amazing and well explained, thanks
great overview
What do we call a research data analysis method that one use to advise or determining the best accounting tool or techniques that should be adopted in a company.
Informative video, explained in a clear and simple way. Kudos
Waoo! I have chosen method wrong for my data analysis. But I can revise my work according to this guide. Thank you so much for this helpful lecture.
This has been very helpful. It gave me a good view of my research objectives and how to choose the best method. Thematic analysis it is.
Very helpful indeed. Thanku so much for the insight.
This was incredibly helpful.
Very helpful.
very educative
Nicely written especially for novice academic researchers like me! Thank you.
choosing a right method for a paper is always a hard job for a student, this is a useful information, but it would be more useful personally for me, if the author provide me with a little bit more information about the data analysis techniques in type of explanatory research. Can we use qualitative content analysis technique for explanatory research ? or what is the suitable data analysis method for explanatory research in social studies?
that was very helpful for me. because these details are so important to my research. thank you very much
I learnt a lot. Thank you
Relevant and Informative, thanks !
Well-planned and organized, thanks much! 🙂
I have reviewed qualitative data analysis in a simplest way possible. The content will highly be useful for developing my book on qualitative data analysis methods. Cheers!
Clear explanation on qualitative and how about Case study
This was helpful. Thank you
This was really of great assistance, it was just the right information needed. Explanation very clear and follow.
Wow, Thanks for making my life easy
This was helpful thanks .
Very helpful…. clear and written in an easily understandable manner. Thank you.
This was so helpful as it was easy to understand. I’m a new to research thank you so much.
so educative…. but Ijust want to know which method is coding of the qualitative or tallying done?
Thank you for the great content, I have learnt a lot. So helpful
precise and clear presentation with simple language and thank you for that.
very informative content, thank you.
You guys are amazing on YouTube on this platform. Your teachings are great, educative, and informative. kudos!
Brilliant Delivery. You made a complex subject seem so easy. Well done.
Beautifully explained.
Thanks a lot
Is there a video the captures the practical process of coding using automated applications?
Thanks for the comment. We don’t recommend using automated applications for coding, as they are not sufficiently accurate in our experience.
content analysis can be qualitative research?
THANK YOU VERY MUCH.
Thank you very much for such a wonderful content
do you have any material on Data collection
What a powerful explanation of the QDA methods. Thank you.
Great explanation both written and Video. i have been using of it on a day to day working of my thesis project in accounting and finance. Thank you very much for your support.
very helpful, thank you so much
The tutorial is useful. I benefited a lot.
This is an eye opener for me and very informative, I have used some of your guidance notes on my Thesis, I wonder if you can assist with your 1. name of your book, year of publication, topic etc., this is for citing in my Bibliography,
I certainly hope to hear from you
Submit a Comment Cancel reply
Your email address will not be published. Required fields are marked *
Save my name, email, and website in this browser for the next time I comment.
- Print Friendly
- Privacy Policy

Home » Qualitative Research – Methods, Analysis Types and Guide
Qualitative Research – Methods, Analysis Types and Guide
Table of Contents

Qualitative Research
Qualitative research is a type of research methodology that focuses on exploring and understanding people’s beliefs, attitudes, behaviors, and experiences through the collection and analysis of non-numerical data. It seeks to answer research questions through the examination of subjective data, such as interviews, focus groups, observations, and textual analysis.
Qualitative research aims to uncover the meaning and significance of social phenomena, and it typically involves a more flexible and iterative approach to data collection and analysis compared to quantitative research. Qualitative research is often used in fields such as sociology, anthropology, psychology, and education.
Qualitative Research Methods

Qualitative Research Methods are as follows:
One-to-One Interview
This method involves conducting an interview with a single participant to gain a detailed understanding of their experiences, attitudes, and beliefs. One-to-one interviews can be conducted in-person, over the phone, or through video conferencing. The interviewer typically uses open-ended questions to encourage the participant to share their thoughts and feelings. One-to-one interviews are useful for gaining detailed insights into individual experiences.
Focus Groups
This method involves bringing together a group of people to discuss a specific topic in a structured setting. The focus group is led by a moderator who guides the discussion and encourages participants to share their thoughts and opinions. Focus groups are useful for generating ideas and insights, exploring social norms and attitudes, and understanding group dynamics.
Ethnographic Studies
This method involves immersing oneself in a culture or community to gain a deep understanding of its norms, beliefs, and practices. Ethnographic studies typically involve long-term fieldwork and observation, as well as interviews and document analysis. Ethnographic studies are useful for understanding the cultural context of social phenomena and for gaining a holistic understanding of complex social processes.
Text Analysis
This method involves analyzing written or spoken language to identify patterns and themes. Text analysis can be quantitative or qualitative. Qualitative text analysis involves close reading and interpretation of texts to identify recurring themes, concepts, and patterns. Text analysis is useful for understanding media messages, public discourse, and cultural trends.
This method involves an in-depth examination of a single person, group, or event to gain an understanding of complex phenomena. Case studies typically involve a combination of data collection methods, such as interviews, observations, and document analysis, to provide a comprehensive understanding of the case. Case studies are useful for exploring unique or rare cases, and for generating hypotheses for further research.
Process of Observation
This method involves systematically observing and recording behaviors and interactions in natural settings. The observer may take notes, use audio or video recordings, or use other methods to document what they see. Process of observation is useful for understanding social interactions, cultural practices, and the context in which behaviors occur.
Record Keeping
This method involves keeping detailed records of observations, interviews, and other data collected during the research process. Record keeping is essential for ensuring the accuracy and reliability of the data, and for providing a basis for analysis and interpretation.
This method involves collecting data from a large sample of participants through a structured questionnaire. Surveys can be conducted in person, over the phone, through mail, or online. Surveys are useful for collecting data on attitudes, beliefs, and behaviors, and for identifying patterns and trends in a population.
Qualitative data analysis is a process of turning unstructured data into meaningful insights. It involves extracting and organizing information from sources like interviews, focus groups, and surveys. The goal is to understand people’s attitudes, behaviors, and motivations
Qualitative Research Analysis Methods
Qualitative Research analysis methods involve a systematic approach to interpreting and making sense of the data collected in qualitative research. Here are some common qualitative data analysis methods:
Thematic Analysis
This method involves identifying patterns or themes in the data that are relevant to the research question. The researcher reviews the data, identifies keywords or phrases, and groups them into categories or themes. Thematic analysis is useful for identifying patterns across multiple data sources and for generating new insights into the research topic.
Content Analysis
This method involves analyzing the content of written or spoken language to identify key themes or concepts. Content analysis can be quantitative or qualitative. Qualitative content analysis involves close reading and interpretation of texts to identify recurring themes, concepts, and patterns. Content analysis is useful for identifying patterns in media messages, public discourse, and cultural trends.
Discourse Analysis
This method involves analyzing language to understand how it constructs meaning and shapes social interactions. Discourse analysis can involve a variety of methods, such as conversation analysis, critical discourse analysis, and narrative analysis. Discourse analysis is useful for understanding how language shapes social interactions, cultural norms, and power relationships.
Grounded Theory Analysis
This method involves developing a theory or explanation based on the data collected. Grounded theory analysis starts with the data and uses an iterative process of coding and analysis to identify patterns and themes in the data. The theory or explanation that emerges is grounded in the data, rather than preconceived hypotheses. Grounded theory analysis is useful for understanding complex social phenomena and for generating new theoretical insights.
Narrative Analysis
This method involves analyzing the stories or narratives that participants share to gain insights into their experiences, attitudes, and beliefs. Narrative analysis can involve a variety of methods, such as structural analysis, thematic analysis, and discourse analysis. Narrative analysis is useful for understanding how individuals construct their identities, make sense of their experiences, and communicate their values and beliefs.
Phenomenological Analysis
This method involves analyzing how individuals make sense of their experiences and the meanings they attach to them. Phenomenological analysis typically involves in-depth interviews with participants to explore their experiences in detail. Phenomenological analysis is useful for understanding subjective experiences and for developing a rich understanding of human consciousness.
Comparative Analysis
This method involves comparing and contrasting data across different cases or groups to identify similarities and differences. Comparative analysis can be used to identify patterns or themes that are common across multiple cases, as well as to identify unique or distinctive features of individual cases. Comparative analysis is useful for understanding how social phenomena vary across different contexts and groups.
Applications of Qualitative Research
Qualitative research has many applications across different fields and industries. Here are some examples of how qualitative research is used:
- Market Research: Qualitative research is often used in market research to understand consumer attitudes, behaviors, and preferences. Researchers conduct focus groups and one-on-one interviews with consumers to gather insights into their experiences and perceptions of products and services.
- Health Care: Qualitative research is used in health care to explore patient experiences and perspectives on health and illness. Researchers conduct in-depth interviews with patients and their families to gather information on their experiences with different health care providers and treatments.
- Education: Qualitative research is used in education to understand student experiences and to develop effective teaching strategies. Researchers conduct classroom observations and interviews with students and teachers to gather insights into classroom dynamics and instructional practices.
- Social Work : Qualitative research is used in social work to explore social problems and to develop interventions to address them. Researchers conduct in-depth interviews with individuals and families to understand their experiences with poverty, discrimination, and other social problems.
- Anthropology : Qualitative research is used in anthropology to understand different cultures and societies. Researchers conduct ethnographic studies and observe and interview members of different cultural groups to gain insights into their beliefs, practices, and social structures.
- Psychology : Qualitative research is used in psychology to understand human behavior and mental processes. Researchers conduct in-depth interviews with individuals to explore their thoughts, feelings, and experiences.
- Public Policy : Qualitative research is used in public policy to explore public attitudes and to inform policy decisions. Researchers conduct focus groups and one-on-one interviews with members of the public to gather insights into their perspectives on different policy issues.
How to Conduct Qualitative Research
Here are some general steps for conducting qualitative research:
- Identify your research question: Qualitative research starts with a research question or set of questions that you want to explore. This question should be focused and specific, but also broad enough to allow for exploration and discovery.
- Select your research design: There are different types of qualitative research designs, including ethnography, case study, grounded theory, and phenomenology. You should select a design that aligns with your research question and that will allow you to gather the data you need to answer your research question.
- Recruit participants: Once you have your research question and design, you need to recruit participants. The number of participants you need will depend on your research design and the scope of your research. You can recruit participants through advertisements, social media, or through personal networks.
- Collect data: There are different methods for collecting qualitative data, including interviews, focus groups, observation, and document analysis. You should select the method or methods that align with your research design and that will allow you to gather the data you need to answer your research question.
- Analyze data: Once you have collected your data, you need to analyze it. This involves reviewing your data, identifying patterns and themes, and developing codes to organize your data. You can use different software programs to help you analyze your data, or you can do it manually.
- Interpret data: Once you have analyzed your data, you need to interpret it. This involves making sense of the patterns and themes you have identified, and developing insights and conclusions that answer your research question. You should be guided by your research question and use your data to support your conclusions.
- Communicate results: Once you have interpreted your data, you need to communicate your results. This can be done through academic papers, presentations, or reports. You should be clear and concise in your communication, and use examples and quotes from your data to support your findings.
Examples of Qualitative Research
Here are some real-time examples of qualitative research:
- Customer Feedback: A company may conduct qualitative research to understand the feedback and experiences of its customers. This may involve conducting focus groups or one-on-one interviews with customers to gather insights into their attitudes, behaviors, and preferences.
- Healthcare : A healthcare provider may conduct qualitative research to explore patient experiences and perspectives on health and illness. This may involve conducting in-depth interviews with patients and their families to gather information on their experiences with different health care providers and treatments.
- Education : An educational institution may conduct qualitative research to understand student experiences and to develop effective teaching strategies. This may involve conducting classroom observations and interviews with students and teachers to gather insights into classroom dynamics and instructional practices.
- Social Work: A social worker may conduct qualitative research to explore social problems and to develop interventions to address them. This may involve conducting in-depth interviews with individuals and families to understand their experiences with poverty, discrimination, and other social problems.
- Anthropology : An anthropologist may conduct qualitative research to understand different cultures and societies. This may involve conducting ethnographic studies and observing and interviewing members of different cultural groups to gain insights into their beliefs, practices, and social structures.
- Psychology : A psychologist may conduct qualitative research to understand human behavior and mental processes. This may involve conducting in-depth interviews with individuals to explore their thoughts, feelings, and experiences.
- Public Policy: A government agency or non-profit organization may conduct qualitative research to explore public attitudes and to inform policy decisions. This may involve conducting focus groups and one-on-one interviews with members of the public to gather insights into their perspectives on different policy issues.
Purpose of Qualitative Research
The purpose of qualitative research is to explore and understand the subjective experiences, behaviors, and perspectives of individuals or groups in a particular context. Unlike quantitative research, which focuses on numerical data and statistical analysis, qualitative research aims to provide in-depth, descriptive information that can help researchers develop insights and theories about complex social phenomena.
Qualitative research can serve multiple purposes, including:
- Exploring new or emerging phenomena : Qualitative research can be useful for exploring new or emerging phenomena, such as new technologies or social trends. This type of research can help researchers develop a deeper understanding of these phenomena and identify potential areas for further study.
- Understanding complex social phenomena : Qualitative research can be useful for exploring complex social phenomena, such as cultural beliefs, social norms, or political processes. This type of research can help researchers develop a more nuanced understanding of these phenomena and identify factors that may influence them.
- Generating new theories or hypotheses: Qualitative research can be useful for generating new theories or hypotheses about social phenomena. By gathering rich, detailed data about individuals’ experiences and perspectives, researchers can develop insights that may challenge existing theories or lead to new lines of inquiry.
- Providing context for quantitative data: Qualitative research can be useful for providing context for quantitative data. By gathering qualitative data alongside quantitative data, researchers can develop a more complete understanding of complex social phenomena and identify potential explanations for quantitative findings.
When to use Qualitative Research
Here are some situations where qualitative research may be appropriate:
- Exploring a new area: If little is known about a particular topic, qualitative research can help to identify key issues, generate hypotheses, and develop new theories.
- Understanding complex phenomena: Qualitative research can be used to investigate complex social, cultural, or organizational phenomena that are difficult to measure quantitatively.
- Investigating subjective experiences: Qualitative research is particularly useful for investigating the subjective experiences of individuals or groups, such as their attitudes, beliefs, values, or emotions.
- Conducting formative research: Qualitative research can be used in the early stages of a research project to develop research questions, identify potential research participants, and refine research methods.
- Evaluating interventions or programs: Qualitative research can be used to evaluate the effectiveness of interventions or programs by collecting data on participants’ experiences, attitudes, and behaviors.
Characteristics of Qualitative Research
Qualitative research is characterized by several key features, including:
- Focus on subjective experience: Qualitative research is concerned with understanding the subjective experiences, beliefs, and perspectives of individuals or groups in a particular context. Researchers aim to explore the meanings that people attach to their experiences and to understand the social and cultural factors that shape these meanings.
- Use of open-ended questions: Qualitative research relies on open-ended questions that allow participants to provide detailed, in-depth responses. Researchers seek to elicit rich, descriptive data that can provide insights into participants’ experiences and perspectives.
- Sampling-based on purpose and diversity: Qualitative research often involves purposive sampling, in which participants are selected based on specific criteria related to the research question. Researchers may also seek to include participants with diverse experiences and perspectives to capture a range of viewpoints.
- Data collection through multiple methods: Qualitative research typically involves the use of multiple data collection methods, such as in-depth interviews, focus groups, and observation. This allows researchers to gather rich, detailed data from multiple sources, which can provide a more complete picture of participants’ experiences and perspectives.
- Inductive data analysis: Qualitative research relies on inductive data analysis, in which researchers develop theories and insights based on the data rather than testing pre-existing hypotheses. Researchers use coding and thematic analysis to identify patterns and themes in the data and to develop theories and explanations based on these patterns.
- Emphasis on researcher reflexivity: Qualitative research recognizes the importance of the researcher’s role in shaping the research process and outcomes. Researchers are encouraged to reflect on their own biases and assumptions and to be transparent about their role in the research process.
Advantages of Qualitative Research
Qualitative research offers several advantages over other research methods, including:
- Depth and detail: Qualitative research allows researchers to gather rich, detailed data that provides a deeper understanding of complex social phenomena. Through in-depth interviews, focus groups, and observation, researchers can gather detailed information about participants’ experiences and perspectives that may be missed by other research methods.
- Flexibility : Qualitative research is a flexible approach that allows researchers to adapt their methods to the research question and context. Researchers can adjust their research methods in real-time to gather more information or explore unexpected findings.
- Contextual understanding: Qualitative research is well-suited to exploring the social and cultural context in which individuals or groups are situated. Researchers can gather information about cultural norms, social structures, and historical events that may influence participants’ experiences and perspectives.
- Participant perspective : Qualitative research prioritizes the perspective of participants, allowing researchers to explore subjective experiences and understand the meanings that participants attach to their experiences.
- Theory development: Qualitative research can contribute to the development of new theories and insights about complex social phenomena. By gathering rich, detailed data and using inductive data analysis, researchers can develop new theories and explanations that may challenge existing understandings.
- Validity : Qualitative research can offer high validity by using multiple data collection methods, purposive and diverse sampling, and researcher reflexivity. This can help ensure that findings are credible and trustworthy.
Limitations of Qualitative Research
Qualitative research also has some limitations, including:
- Subjectivity : Qualitative research relies on the subjective interpretation of researchers, which can introduce bias into the research process. The researcher’s perspective, beliefs, and experiences can influence the way data is collected, analyzed, and interpreted.
- Limited generalizability: Qualitative research typically involves small, purposive samples that may not be representative of larger populations. This limits the generalizability of findings to other contexts or populations.
- Time-consuming: Qualitative research can be a time-consuming process, requiring significant resources for data collection, analysis, and interpretation.
- Resource-intensive: Qualitative research may require more resources than other research methods, including specialized training for researchers, specialized software for data analysis, and transcription services.
- Limited reliability: Qualitative research may be less reliable than quantitative research, as it relies on the subjective interpretation of researchers. This can make it difficult to replicate findings or compare results across different studies.
- Ethics and confidentiality: Qualitative research involves collecting sensitive information from participants, which raises ethical concerns about confidentiality and informed consent. Researchers must take care to protect the privacy and confidentiality of participants and obtain informed consent.
Also see Research Methods
About the author
Muhammad Hassan
Researcher, Academic Writer, Web developer
You may also like

Ethnographic Research -Types, Methods and Guide

Explanatory Research – Types, Methods, Guide

Experimental Design – Types, Methods, Guide

Questionnaire – Definition, Types, and Examples

Observational Research – Methods and Guide

Descriptive Research Design – Types, Methods and...
- Skip to main content
- Skip to primary sidebar
- Skip to footer
- QuestionPro

- Solutions Industries Gaming Automotive Sports and events Education Government Travel & Hospitality Financial Services Healthcare Cannabis Technology Use Case AskWhy Communities Audience Contactless surveys Mobile LivePolls Member Experience GDPR Positive People Science 360 Feedback Surveys
- Resources Blog eBooks Survey Templates Case Studies Training Help center
Home Market Research
Qualitative Data Analysis: What is it, Methods + Examples

In a world rich with information and narrative, understanding the deeper layers of human experiences requires a unique vision that goes beyond numbers and figures. This is where the power of qualitative data analysis comes to light.
In this blog, we’ll learn about qualitative data analysis, explore its methods, and provide real-life examples showcasing its power in uncovering insights.
What is Qualitative Data Analysis?
Qualitative data analysis is a systematic process of examining non-numerical data to extract meaning, patterns, and insights.
In contrast to quantitative analysis, which focuses on numbers and statistical metrics, the qualitative study focuses on the qualitative aspects of data, such as text, images, audio, and videos. It seeks to understand every aspect of human experiences, perceptions, and behaviors by examining the data’s richness.
Companies frequently conduct this analysis on customer feedback. You can collect qualitative data from reviews, complaints, chat messages, interactions with support centers, customer interviews, case notes, or even social media comments. This kind of data holds the key to understanding customer sentiments and preferences in a way that goes beyond mere numbers.
Importance of Qualitative Data Analysis
Qualitative data analysis plays a crucial role in your research and decision-making process across various disciplines. Let’s explore some key reasons that underline the significance of this analysis:
In-Depth Understanding
It enables you to explore complex and nuanced aspects of a phenomenon, delving into the ‘how’ and ‘why’ questions. This method provides you with a deeper understanding of human behavior, experiences, and contexts that quantitative approaches might not capture fully.
Contextual Insight
You can use this analysis to give context to numerical data. It will help you understand the circumstances and conditions that influence participants’ thoughts, feelings, and actions. This contextual insight becomes essential for generating comprehensive explanations.
Theory Development
You can generate or refine hypotheses via qualitative data analysis. As you analyze the data attentively, you can form hypotheses, concepts, and frameworks that will drive your future research and contribute to theoretical advances.
Participant Perspectives
When performing qualitative research, you can highlight participant voices and opinions. This approach is especially useful for understanding marginalized or underrepresented people, as it allows them to communicate their experiences and points of view.
Exploratory Research
The analysis is frequently used at the exploratory stage of your project. It assists you in identifying important variables, developing research questions, and designing quantitative studies that will follow.
Types of Qualitative Data
When conducting qualitative research, you can use several qualitative data collection methods , and here you will come across many sorts of qualitative data that can provide you with unique insights into your study topic. These data kinds add new views and angles to your understanding and analysis.
Interviews and Focus Groups
Interviews and focus groups will be among your key methods for gathering qualitative data. Interviews are one-on-one talks in which participants can freely share their thoughts, experiences, and opinions.
Focus groups, on the other hand, are discussions in which members interact with one another, resulting in dynamic exchanges of ideas. Both methods provide rich qualitative data and direct access to participant perspectives.
Observations and Field Notes
Observations and field notes are another useful sort of qualitative data. You can immerse yourself in the research environment through direct observation, carefully documenting behaviors, interactions, and contextual factors.
These observations will be recorded in your field notes, providing a complete picture of the environment and the behaviors you’re researching. This data type is especially important for comprehending behavior in their natural setting.
Textual and Visual Data
Textual and visual data include a wide range of resources that can be qualitatively analyzed. Documents, written narratives, and transcripts from various sources, such as interviews or speeches, are examples of textual data.
Photographs, films, and even artwork provide a visual layer to your research. These forms of data allow you to investigate what is spoken and the underlying emotions, details, and symbols expressed by language or pictures.
When to Choose Qualitative Data Analysis over Quantitative Data Analysis
As you begin your research journey, understanding why the analysis of qualitative data is important will guide your approach to understanding complex events. If you analyze qualitative data, it will provide new insights that complement quantitative methodologies, which will give you a broader understanding of your study topic.
It is critical to know when to use qualitative analysis over quantitative procedures. You can prefer qualitative data analysis when:
- Complexity Reigns: When your research questions involve deep human experiences, motivations, or emotions, qualitative research excels at revealing these complexities.
- Exploration is Key: Qualitative analysis is ideal for exploratory research. It will assist you in understanding a new or poorly understood topic before formulating quantitative hypotheses.
- Context Matters: If you want to understand how context affects behaviors or results, qualitative data analysis provides the depth needed to grasp these relationships.
- Unanticipated Findings: When your study provides surprising new viewpoints or ideas, qualitative analysis helps you to delve deeply into these emerging themes.
- Subjective Interpretation is Vital: When it comes to understanding people’s subjective experiences and interpretations, qualitative data analysis is the way to go.
You can make informed decisions regarding the right approach for your research objectives if you understand the importance of qualitative analysis and recognize the situations where it shines.
Qualitative Data Analysis Methods and Examples
Exploring various qualitative data analysis methods will provide you with a wide collection for making sense of your research findings. Once the data has been collected, you can choose from several analysis methods based on your research objectives and the data type you’ve collected.
There are five main methods for analyzing qualitative data. Each method takes a distinct approach to identifying patterns, themes, and insights within your qualitative data. They are:
Method 1: Content Analysis
Content analysis is a methodical technique for analyzing textual or visual data in a structured manner. In this method, you will categorize qualitative data by splitting it into manageable pieces and assigning the manual coding process to these units.
As you go, you’ll notice ongoing codes and designs that will allow you to conclude the content. This method is very beneficial for detecting common ideas, concepts, or themes in your data without losing the context.
Steps to Do Content Analysis
Follow these steps when conducting content analysis:
- Collect and Immerse: Begin by collecting the necessary textual or visual data. Immerse yourself in this data to fully understand its content, context, and complexities.
- Assign Codes and Categories: Assign codes to relevant data sections that systematically represent major ideas or themes. Arrange comparable codes into groups that cover the major themes.
- Analyze and Interpret: Develop a structured framework from the categories and codes. Then, evaluate the data in the context of your research question, investigate relationships between categories, discover patterns, and draw meaning from these connections.
Benefits & Challenges
There are various advantages to using content analysis:
- Structured Approach: It offers a systematic approach to dealing with large data sets and ensures consistency throughout the research.
- Objective Insights: This method promotes objectivity, which helps to reduce potential biases in your study.
- Pattern Discovery: Content analysis can help uncover hidden trends, themes, and patterns that are not always obvious.
- Versatility: You can apply content analysis to various data formats, including text, internet content, images, etc.
However, keep in mind the challenges that arise:
- Subjectivity: Even with the best attempts, a certain bias may remain in coding and interpretation.
- Complexity: Analyzing huge data sets requires time and great attention to detail.
- Contextual Nuances: Content analysis may not capture all of the contextual richness that qualitative data analysis highlights.
Example of Content Analysis
Suppose you’re conducting market research and looking at customer feedback on a product. As you collect relevant data and analyze feedback, you’ll see repeating codes like “price,” “quality,” “customer service,” and “features.” These codes are organized into categories such as “positive reviews,” “negative reviews,” and “suggestions for improvement.”
According to your findings, themes such as “price” and “customer service” stand out and show that pricing and customer service greatly impact customer satisfaction. This example highlights the power of content analysis for obtaining significant insights from large textual data collections.
Method 2: Thematic Analysis
Thematic analysis is a well-structured procedure for identifying and analyzing recurring themes in your data. As you become more engaged in the data, you’ll generate codes or short labels representing key concepts. These codes are then organized into themes, providing a consistent framework for organizing and comprehending the substance of the data.
The analysis allows you to organize complex narratives and perspectives into meaningful categories, which will allow you to identify connections and patterns that may not be visible at first.
Steps to Do Thematic Analysis
Follow these steps when conducting a thematic analysis:
- Code and Group: Start by thoroughly examining the data and giving initial codes that identify the segments. To create initial themes, combine relevant codes.
- Code and Group: Begin by engaging yourself in the data, assigning first codes to notable segments. To construct basic themes, group comparable codes together.
- Analyze and Report: Analyze the data within each theme to derive relevant insights. Organize the topics into a consistent structure and explain your findings, along with data extracts that represent each theme.
Thematic analysis has various benefits:
- Structured Exploration: It is a method for identifying patterns and themes in complex qualitative data.
- Comprehensive knowledge: Thematic analysis promotes an in-depth understanding of the complications and meanings of the data.
- Application Flexibility: This method can be customized to various research situations and data kinds.
However, challenges may arise, such as:
- Interpretive Nature: Interpreting qualitative data in thematic analysis is vital, and it is critical to manage researcher bias.
- Time-consuming: The study can be time-consuming, especially with large data sets.
- Subjectivity: The selection of codes and topics might be subjective.
Example of Thematic Analysis
Assume you’re conducting a thematic analysis on job satisfaction interviews. Following your immersion in the data, you assign initial codes such as “work-life balance,” “career growth,” and “colleague relationships.” As you organize these codes, you’ll notice themes develop, such as “Factors Influencing Job Satisfaction” and “Impact on Work Engagement.”
Further investigation reveals the tales and experiences included within these themes and provides insights into how various elements influence job satisfaction. This example demonstrates how thematic analysis can reveal meaningful patterns and insights in qualitative data.
Method 3: Narrative Analysis
The narrative analysis involves the narratives that people share. You’ll investigate the histories in your data, looking at how stories are created and the meanings they express. This method is excellent for learning how people make sense of their experiences through narrative.
Steps to Do Narrative Analysis
The following steps are involved in narrative analysis:
- Gather and Analyze: Start by collecting narratives, such as first-person tales, interviews, or written accounts. Analyze the stories, focusing on the plot, feelings, and characters.
- Find Themes: Look for recurring themes or patterns in various narratives. Think about the similarities and differences between these topics and personal experiences.
- Interpret and Extract Insights: Contextualize the narratives within their larger context. Accept the subjective nature of each narrative and analyze the narrator’s voice and style. Extract insights from the tales by diving into the emotions, motivations, and implications communicated by the stories.
There are various advantages to narrative analysis:
- Deep Exploration: It lets you look deeply into people’s personal experiences and perspectives.
- Human-Centered: This method prioritizes the human perspective, allowing individuals to express themselves.
However, difficulties may arise, such as:
- Interpretive Complexity: Analyzing narratives requires dealing with the complexities of meaning and interpretation.
- Time-consuming: Because of the richness and complexities of tales, working with them can be time-consuming.
Example of Narrative Analysis
Assume you’re conducting narrative analysis on refugee interviews. As you read the stories, you’ll notice common themes of toughness, loss, and hope. The narratives provide insight into the obstacles that refugees face, their strengths, and the dreams that guide them.
The analysis can provide a deeper insight into the refugees’ experiences and the broader social context they navigate by examining the narratives’ emotional subtleties and underlying meanings. This example highlights how narrative analysis can reveal important insights into human stories.
Method 4: Grounded Theory Analysis
Grounded theory analysis is an iterative and systematic approach that allows you to create theories directly from data without being limited by pre-existing hypotheses. With an open mind, you collect data and generate early codes and labels that capture essential ideas or concepts within the data.
As you progress, you refine these codes and increasingly connect them, eventually developing a theory based on the data. Grounded theory analysis is a dynamic process for developing new insights and hypotheses based on details in your data.
Steps to Do Grounded Theory Analysis
Grounded theory analysis requires the following steps:
- Initial Coding: First, immerse yourself in the data, producing initial codes that represent major concepts or patterns.
- Categorize and Connect: Using axial coding, organize the initial codes, which establish relationships and connections between topics.
- Build the Theory: Focus on creating a core category that connects the codes and themes. Regularly refine the theory by comparing and integrating new data, ensuring that it evolves organically from the data.
Grounded theory analysis has various benefits:
- Theory Generation: It provides a one-of-a-kind opportunity to generate hypotheses straight from data and promotes new insights.
- In-depth Understanding: The analysis allows you to deeply analyze the data and reveal complex relationships and patterns.
- Flexible Process: This method is customizable and ongoing, which allows you to enhance your research as you collect additional data.
However, challenges might arise with:
- Time and Resources: Because grounded theory analysis is a continuous process, it requires a large commitment of time and resources.
- Theoretical Development: Creating a grounded theory involves a thorough understanding of qualitative data analysis software and theoretical concepts.
- Interpretation of Complexity: Interpreting and incorporating a newly developed theory into existing literature can be intellectually hard.
Example of Grounded Theory Analysis
Assume you’re performing a grounded theory analysis on workplace collaboration interviews. As you open code the data, you will discover notions such as “communication barriers,” “team dynamics,” and “leadership roles.” Axial coding demonstrates links between these notions, emphasizing the significance of efficient communication in developing collaboration.
You create the core “Integrated Communication Strategies” category through selective coding, which unifies new topics.
This theory-driven category serves as the framework for understanding how numerous aspects contribute to effective team collaboration. This example shows how grounded theory analysis allows you to generate a theory directly from the inherent nature of the data.
Method 5: Discourse Analysis
Discourse analysis focuses on language and communication. You’ll look at how language produces meaning and how it reflects power relations, identities, and cultural influences. This strategy examines what is said and how it is said; the words, phrasing, and larger context of communication.
The analysis is precious when investigating power dynamics, identities, and cultural influences encoded in language. By evaluating the language used in your data, you can identify underlying assumptions, cultural standards, and how individuals negotiate meaning through communication.
Steps to Do Discourse Analysis
Conducting discourse analysis entails the following steps:
- Select Discourse: For analysis, choose language-based data such as texts, speeches, or media content.
- Analyze Language: Immerse yourself in the conversation, examining language choices, metaphors, and underlying assumptions.
- Discover Patterns: Recognize the dialogue’s reoccurring themes, ideologies, and power dynamics. To fully understand the effects of these patterns, put them in their larger context.
There are various advantages of using discourse analysis:
- Understanding Language: It provides an extensive understanding of how language builds meaning and influences perceptions.
- Uncovering Power Dynamics: The analysis reveals how power dynamics appear via language.
- Cultural Insights: This method identifies cultural norms, beliefs, and ideologies stored in communication.
However, the following challenges may arise:
- Complexity of Interpretation: Language analysis involves navigating multiple levels of nuance and interpretation.
- Subjectivity: Interpretation can be subjective, so controlling researcher bias is important.
- Time-Intensive: Discourse analysis can take a lot of time because careful linguistic study is required in this analysis.
Example of Discourse Analysis
Consider doing discourse analysis on media coverage of a political event. You notice repeating linguistic patterns in news articles that depict the event as a conflict between opposing parties. Through deconstruction, you can expose how this framing supports particular ideologies and power relations.
You can illustrate how language choices influence public perceptions and contribute to building the narrative around the event by analyzing the speech within the broader political and social context. This example shows how discourse analysis can reveal hidden power dynamics and cultural influences on communication.
How to do Qualitative Data Analysis with the QuestionPro Research suite?
QuestionPro is a popular survey and research platform that offers tools for collecting and analyzing qualitative and quantitative data. Follow these general steps for conducting qualitative data analysis using the QuestionPro Research Suite:
- Collect Qualitative Data: Set up your survey to capture qualitative responses. It might involve open-ended questions, text boxes, or comment sections where participants can provide detailed responses.
- Export Qualitative Responses: Export the responses once you’ve collected qualitative data through your survey. QuestionPro typically allows you to export survey data in various formats, such as Excel or CSV.
- Prepare Data for Analysis: Review the exported data and clean it if necessary. Remove irrelevant or duplicate entries to ensure your data is ready for analysis.
- Code and Categorize Responses: Segment and label data, letting new patterns emerge naturally, then develop categories through axial coding to structure the analysis.
- Identify Themes: Analyze the coded responses to identify recurring themes, patterns, and insights. Look for similarities and differences in participants’ responses.
- Generate Reports and Visualizations: Utilize the reporting features of QuestionPro to create visualizations, charts, and graphs that help communicate the themes and findings from your qualitative research.
- Interpret and Draw Conclusions: Interpret the themes and patterns you’ve identified in the qualitative data. Consider how these findings answer your research questions or provide insights into your study topic.
- Integrate with Quantitative Data (if applicable): If you’re also conducting quantitative research using QuestionPro, consider integrating your qualitative findings with quantitative results to provide a more comprehensive understanding.
Qualitative data analysis is vital in uncovering various human experiences, views, and stories. If you’re ready to transform your research journey and apply the power of qualitative analysis, now is the moment to do it. Book a demo with QuestionPro today and begin your journey of exploration.
LEARN MORE FREE TRIAL
MORE LIKE THIS

Age Gating: Effective Strategies for Online Content Control
Aug 23, 2024

Customer Experience Lessons from 13,000 Feet — Tuesday CX Thoughts
Aug 20, 2024

Insight: Definition & meaning, types and examples
Aug 19, 2024

Employee Loyalty: Strategies for Long-Term Business Success
Other categories.
- Academic Research
- Artificial Intelligence
- Assessments
- Brand Awareness
- Case Studies
- Communities
- Consumer Insights
- Customer effort score
- Customer Engagement
- Customer Experience
- Customer Loyalty
- Customer Research
- Customer Satisfaction
- Employee Benefits
- Employee Engagement
- Employee Retention
- Friday Five
- General Data Protection Regulation
- Insights Hub
- Life@QuestionPro
- Market Research
- Mobile diaries
- Mobile Surveys
- New Features
- Online Communities
- Question Types
- Questionnaire
- QuestionPro Products
- Release Notes
- Research Tools and Apps
- Revenue at Risk
- Survey Templates
- Training Tips
- Tuesday CX Thoughts (TCXT)
- Uncategorized
- What’s Coming Up
- Workforce Intelligence
Have a language expert improve your writing
Run a free plagiarism check in 10 minutes, generate accurate citations for free.
- Knowledge Base
Methodology
- What Is Qualitative Research? | Methods & Examples
What Is Qualitative Research? | Methods & Examples
Published on June 19, 2020 by Pritha Bhandari . Revised on June 22, 2023.
Qualitative research involves collecting and analyzing non-numerical data (e.g., text, video, or audio) to understand concepts, opinions, or experiences. It can be used to gather in-depth insights into a problem or generate new ideas for research.
Qualitative research is the opposite of quantitative research , which involves collecting and analyzing numerical data for statistical analysis.
Qualitative research is commonly used in the humanities and social sciences, in subjects such as anthropology, sociology, education, health sciences, history, etc.
- How does social media shape body image in teenagers?
- How do children and adults interpret healthy eating in the UK?
- What factors influence employee retention in a large organization?
- How is anxiety experienced around the world?
- How can teachers integrate social issues into science curriculums?
Table of contents
Approaches to qualitative research, qualitative research methods, qualitative data analysis, advantages of qualitative research, disadvantages of qualitative research, other interesting articles, frequently asked questions about qualitative research.
Qualitative research is used to understand how people experience the world. While there are many approaches to qualitative research, they tend to be flexible and focus on retaining rich meaning when interpreting data.
Common approaches include grounded theory, ethnography , action research , phenomenological research, and narrative research. They share some similarities, but emphasize different aims and perspectives.
| Approach | What does it involve? |
|---|---|
| Grounded theory | Researchers collect rich data on a topic of interest and develop theories . |
| Researchers immerse themselves in groups or organizations to understand their cultures. | |
| Action research | Researchers and participants collaboratively link theory to practice to drive social change. |
| Phenomenological research | Researchers investigate a phenomenon or event by describing and interpreting participants’ lived experiences. |
| Narrative research | Researchers examine how stories are told to understand how participants perceive and make sense of their experiences. |
Note that qualitative research is at risk for certain research biases including the Hawthorne effect , observer bias , recall bias , and social desirability bias . While not always totally avoidable, awareness of potential biases as you collect and analyze your data can prevent them from impacting your work too much.
Prevent plagiarism. Run a free check.
Each of the research approaches involve using one or more data collection methods . These are some of the most common qualitative methods:
- Observations: recording what you have seen, heard, or encountered in detailed field notes.
- Interviews: personally asking people questions in one-on-one conversations.
- Focus groups: asking questions and generating discussion among a group of people.
- Surveys : distributing questionnaires with open-ended questions.
- Secondary research: collecting existing data in the form of texts, images, audio or video recordings, etc.
- You take field notes with observations and reflect on your own experiences of the company culture.
- You distribute open-ended surveys to employees across all the company’s offices by email to find out if the culture varies across locations.
- You conduct in-depth interviews with employees in your office to learn about their experiences and perspectives in greater detail.
Qualitative researchers often consider themselves “instruments” in research because all observations, interpretations and analyses are filtered through their own personal lens.
For this reason, when writing up your methodology for qualitative research, it’s important to reflect on your approach and to thoroughly explain the choices you made in collecting and analyzing the data.
Qualitative data can take the form of texts, photos, videos and audio. For example, you might be working with interview transcripts, survey responses, fieldnotes, or recordings from natural settings.
Most types of qualitative data analysis share the same five steps:
- Prepare and organize your data. This may mean transcribing interviews or typing up fieldnotes.
- Review and explore your data. Examine the data for patterns or repeated ideas that emerge.
- Develop a data coding system. Based on your initial ideas, establish a set of codes that you can apply to categorize your data.
- Assign codes to the data. For example, in qualitative survey analysis, this may mean going through each participant’s responses and tagging them with codes in a spreadsheet. As you go through your data, you can create new codes to add to your system if necessary.
- Identify recurring themes. Link codes together into cohesive, overarching themes.
There are several specific approaches to analyzing qualitative data. Although these methods share similar processes, they emphasize different concepts.
| Approach | When to use | Example |
|---|---|---|
| To describe and categorize common words, phrases, and ideas in qualitative data. | A market researcher could perform content analysis to find out what kind of language is used in descriptions of therapeutic apps. | |
| To identify and interpret patterns and themes in qualitative data. | A psychologist could apply thematic analysis to travel blogs to explore how tourism shapes self-identity. | |
| To examine the content, structure, and design of texts. | A media researcher could use textual analysis to understand how news coverage of celebrities has changed in the past decade. | |
| To study communication and how language is used to achieve effects in specific contexts. | A political scientist could use discourse analysis to study how politicians generate trust in election campaigns. |
Qualitative research often tries to preserve the voice and perspective of participants and can be adjusted as new research questions arise. Qualitative research is good for:
- Flexibility
The data collection and analysis process can be adapted as new ideas or patterns emerge. They are not rigidly decided beforehand.
- Natural settings
Data collection occurs in real-world contexts or in naturalistic ways.
- Meaningful insights
Detailed descriptions of people’s experiences, feelings and perceptions can be used in designing, testing or improving systems or products.
- Generation of new ideas
Open-ended responses mean that researchers can uncover novel problems or opportunities that they wouldn’t have thought of otherwise.
Researchers must consider practical and theoretical limitations in analyzing and interpreting their data. Qualitative research suffers from:
- Unreliability
The real-world setting often makes qualitative research unreliable because of uncontrolled factors that affect the data.
- Subjectivity
Due to the researcher’s primary role in analyzing and interpreting data, qualitative research cannot be replicated . The researcher decides what is important and what is irrelevant in data analysis, so interpretations of the same data can vary greatly.
- Limited generalizability
Small samples are often used to gather detailed data about specific contexts. Despite rigorous analysis procedures, it is difficult to draw generalizable conclusions because the data may be biased and unrepresentative of the wider population .
- Labor-intensive
Although software can be used to manage and record large amounts of text, data analysis often has to be checked or performed manually.
If you want to know more about statistics , methodology , or research bias , make sure to check out some of our other articles with explanations and examples.
- Chi square goodness of fit test
- Degrees of freedom
- Null hypothesis
- Discourse analysis
- Control groups
- Mixed methods research
- Non-probability sampling
- Quantitative research
- Inclusion and exclusion criteria
Research bias
- Rosenthal effect
- Implicit bias
- Cognitive bias
- Selection bias
- Negativity bias
- Status quo bias
Quantitative research deals with numbers and statistics, while qualitative research deals with words and meanings.
Quantitative methods allow you to systematically measure variables and test hypotheses . Qualitative methods allow you to explore concepts and experiences in more detail.
There are five common approaches to qualitative research :
- Grounded theory involves collecting data in order to develop new theories.
- Ethnography involves immersing yourself in a group or organization to understand its culture.
- Narrative research involves interpreting stories to understand how people make sense of their experiences and perceptions.
- Phenomenological research involves investigating phenomena through people’s lived experiences.
- Action research links theory and practice in several cycles to drive innovative changes.
Data collection is the systematic process by which observations or measurements are gathered in research. It is used in many different contexts by academics, governments, businesses, and other organizations.
There are various approaches to qualitative data analysis , but they all share five steps in common:
- Prepare and organize your data.
- Review and explore your data.
- Develop a data coding system.
- Assign codes to the data.
- Identify recurring themes.
The specifics of each step depend on the focus of the analysis. Some common approaches include textual analysis , thematic analysis , and discourse analysis .
Cite this Scribbr article
If you want to cite this source, you can copy and paste the citation or click the “Cite this Scribbr article” button to automatically add the citation to our free Citation Generator.
Bhandari, P. (2023, June 22). What Is Qualitative Research? | Methods & Examples. Scribbr. Retrieved August 24, 2024, from https://www.scribbr.com/methodology/qualitative-research/
Is this article helpful?

Pritha Bhandari
Other students also liked, qualitative vs. quantitative research | differences, examples & methods, how to do thematic analysis | step-by-step guide & examples, "i thought ai proofreading was useless but..".
I've been using Scribbr for years now and I know it's a service that won't disappoint. It does a good job spotting mistakes”
- Open access
- Published: 27 May 2020
How to use and assess qualitative research methods
- Loraine Busetto ORCID: orcid.org/0000-0002-9228-7875 1 ,
- Wolfgang Wick 1 , 2 &
- Christoph Gumbinger 1
Neurological Research and Practice volume 2 , Article number: 14 ( 2020 ) Cite this article
772k Accesses
352 Citations
90 Altmetric
Metrics details
This paper aims to provide an overview of the use and assessment of qualitative research methods in the health sciences. Qualitative research can be defined as the study of the nature of phenomena and is especially appropriate for answering questions of why something is (not) observed, assessing complex multi-component interventions, and focussing on intervention improvement. The most common methods of data collection are document study, (non-) participant observations, semi-structured interviews and focus groups. For data analysis, field-notes and audio-recordings are transcribed into protocols and transcripts, and coded using qualitative data management software. Criteria such as checklists, reflexivity, sampling strategies, piloting, co-coding, member-checking and stakeholder involvement can be used to enhance and assess the quality of the research conducted. Using qualitative in addition to quantitative designs will equip us with better tools to address a greater range of research problems, and to fill in blind spots in current neurological research and practice.
The aim of this paper is to provide an overview of qualitative research methods, including hands-on information on how they can be used, reported and assessed. This article is intended for beginning qualitative researchers in the health sciences as well as experienced quantitative researchers who wish to broaden their understanding of qualitative research.
What is qualitative research?
Qualitative research is defined as “the study of the nature of phenomena”, including “their quality, different manifestations, the context in which they appear or the perspectives from which they can be perceived” , but excluding “their range, frequency and place in an objectively determined chain of cause and effect” [ 1 ]. This formal definition can be complemented with a more pragmatic rule of thumb: qualitative research generally includes data in form of words rather than numbers [ 2 ].
Why conduct qualitative research?
Because some research questions cannot be answered using (only) quantitative methods. For example, one Australian study addressed the issue of why patients from Aboriginal communities often present late or not at all to specialist services offered by tertiary care hospitals. Using qualitative interviews with patients and staff, it found one of the most significant access barriers to be transportation problems, including some towns and communities simply not having a bus service to the hospital [ 3 ]. A quantitative study could have measured the number of patients over time or even looked at possible explanatory factors – but only those previously known or suspected to be of relevance. To discover reasons for observed patterns, especially the invisible or surprising ones, qualitative designs are needed.
While qualitative research is common in other fields, it is still relatively underrepresented in health services research. The latter field is more traditionally rooted in the evidence-based-medicine paradigm, as seen in " research that involves testing the effectiveness of various strategies to achieve changes in clinical practice, preferably applying randomised controlled trial study designs (...) " [ 4 ]. This focus on quantitative research and specifically randomised controlled trials (RCT) is visible in the idea of a hierarchy of research evidence which assumes that some research designs are objectively better than others, and that choosing a "lesser" design is only acceptable when the better ones are not practically or ethically feasible [ 5 , 6 ]. Others, however, argue that an objective hierarchy does not exist, and that, instead, the research design and methods should be chosen to fit the specific research question at hand – "questions before methods" [ 2 , 7 , 8 , 9 ]. This means that even when an RCT is possible, some research problems require a different design that is better suited to addressing them. Arguing in JAMA, Berwick uses the example of rapid response teams in hospitals, which he describes as " a complex, multicomponent intervention – essentially a process of social change" susceptible to a range of different context factors including leadership or organisation history. According to him, "[in] such complex terrain, the RCT is an impoverished way to learn. Critics who use it as a truth standard in this context are incorrect" [ 8 ] . Instead of limiting oneself to RCTs, Berwick recommends embracing a wider range of methods , including qualitative ones, which for "these specific applications, (...) are not compromises in learning how to improve; they are superior" [ 8 ].
Research problems that can be approached particularly well using qualitative methods include assessing complex multi-component interventions or systems (of change), addressing questions beyond “what works”, towards “what works for whom when, how and why”, and focussing on intervention improvement rather than accreditation [ 7 , 9 , 10 , 11 , 12 ]. Using qualitative methods can also help shed light on the “softer” side of medical treatment. For example, while quantitative trials can measure the costs and benefits of neuro-oncological treatment in terms of survival rates or adverse effects, qualitative research can help provide a better understanding of patient or caregiver stress, visibility of illness or out-of-pocket expenses.
How to conduct qualitative research?
Given that qualitative research is characterised by flexibility, openness and responsivity to context, the steps of data collection and analysis are not as separate and consecutive as they tend to be in quantitative research [ 13 , 14 ]. As Fossey puts it : “sampling, data collection, analysis and interpretation are related to each other in a cyclical (iterative) manner, rather than following one after another in a stepwise approach” [ 15 ]. The researcher can make educated decisions with regard to the choice of method, how they are implemented, and to which and how many units they are applied [ 13 ]. As shown in Fig. 1 , this can involve several back-and-forth steps between data collection and analysis where new insights and experiences can lead to adaption and expansion of the original plan. Some insights may also necessitate a revision of the research question and/or the research design as a whole. The process ends when saturation is achieved, i.e. when no relevant new information can be found (see also below: sampling and saturation). For reasons of transparency, it is essential for all decisions as well as the underlying reasoning to be well-documented.
Iterative research process
While it is not always explicitly addressed, qualitative methods reflect a different underlying research paradigm than quantitative research (e.g. constructivism or interpretivism as opposed to positivism). The choice of methods can be based on the respective underlying substantive theory or theoretical framework used by the researcher [ 2 ].
Data collection
The methods of qualitative data collection most commonly used in health research are document study, observations, semi-structured interviews and focus groups [ 1 , 14 , 16 , 17 ].
Document study
Document study (also called document analysis) refers to the review by the researcher of written materials [ 14 ]. These can include personal and non-personal documents such as archives, annual reports, guidelines, policy documents, diaries or letters.
Observations
Observations are particularly useful to gain insights into a certain setting and actual behaviour – as opposed to reported behaviour or opinions [ 13 ]. Qualitative observations can be either participant or non-participant in nature. In participant observations, the observer is part of the observed setting, for example a nurse working in an intensive care unit [ 18 ]. In non-participant observations, the observer is “on the outside looking in”, i.e. present in but not part of the situation, trying not to influence the setting by their presence. Observations can be planned (e.g. for 3 h during the day or night shift) or ad hoc (e.g. as soon as a stroke patient arrives at the emergency room). During the observation, the observer takes notes on everything or certain pre-determined parts of what is happening around them, for example focusing on physician-patient interactions or communication between different professional groups. Written notes can be taken during or after the observations, depending on feasibility (which is usually lower during participant observations) and acceptability (e.g. when the observer is perceived to be judging the observed). Afterwards, these field notes are transcribed into observation protocols. If more than one observer was involved, field notes are taken independently, but notes can be consolidated into one protocol after discussions. Advantages of conducting observations include minimising the distance between the researcher and the researched, the potential discovery of topics that the researcher did not realise were relevant and gaining deeper insights into the real-world dimensions of the research problem at hand [ 18 ].
Semi-structured interviews
Hijmans & Kuyper describe qualitative interviews as “an exchange with an informal character, a conversation with a goal” [ 19 ]. Interviews are used to gain insights into a person’s subjective experiences, opinions and motivations – as opposed to facts or behaviours [ 13 ]. Interviews can be distinguished by the degree to which they are structured (i.e. a questionnaire), open (e.g. free conversation or autobiographical interviews) or semi-structured [ 2 , 13 ]. Semi-structured interviews are characterized by open-ended questions and the use of an interview guide (or topic guide/list) in which the broad areas of interest, sometimes including sub-questions, are defined [ 19 ]. The pre-defined topics in the interview guide can be derived from the literature, previous research or a preliminary method of data collection, e.g. document study or observations. The topic list is usually adapted and improved at the start of the data collection process as the interviewer learns more about the field [ 20 ]. Across interviews the focus on the different (blocks of) questions may differ and some questions may be skipped altogether (e.g. if the interviewee is not able or willing to answer the questions or for concerns about the total length of the interview) [ 20 ]. Qualitative interviews are usually not conducted in written format as it impedes on the interactive component of the method [ 20 ]. In comparison to written surveys, qualitative interviews have the advantage of being interactive and allowing for unexpected topics to emerge and to be taken up by the researcher. This can also help overcome a provider or researcher-centred bias often found in written surveys, which by nature, can only measure what is already known or expected to be of relevance to the researcher. Interviews can be audio- or video-taped; but sometimes it is only feasible or acceptable for the interviewer to take written notes [ 14 , 16 , 20 ].
Focus groups
Focus groups are group interviews to explore participants’ expertise and experiences, including explorations of how and why people behave in certain ways [ 1 ]. Focus groups usually consist of 6–8 people and are led by an experienced moderator following a topic guide or “script” [ 21 ]. They can involve an observer who takes note of the non-verbal aspects of the situation, possibly using an observation guide [ 21 ]. Depending on researchers’ and participants’ preferences, the discussions can be audio- or video-taped and transcribed afterwards [ 21 ]. Focus groups are useful for bringing together homogeneous (to a lesser extent heterogeneous) groups of participants with relevant expertise and experience on a given topic on which they can share detailed information [ 21 ]. Focus groups are a relatively easy, fast and inexpensive method to gain access to information on interactions in a given group, i.e. “the sharing and comparing” among participants [ 21 ]. Disadvantages include less control over the process and a lesser extent to which each individual may participate. Moreover, focus group moderators need experience, as do those tasked with the analysis of the resulting data. Focus groups can be less appropriate for discussing sensitive topics that participants might be reluctant to disclose in a group setting [ 13 ]. Moreover, attention must be paid to the emergence of “groupthink” as well as possible power dynamics within the group, e.g. when patients are awed or intimidated by health professionals.
Choosing the “right” method
As explained above, the school of thought underlying qualitative research assumes no objective hierarchy of evidence and methods. This means that each choice of single or combined methods has to be based on the research question that needs to be answered and a critical assessment with regard to whether or to what extent the chosen method can accomplish this – i.e. the “fit” between question and method [ 14 ]. It is necessary for these decisions to be documented when they are being made, and to be critically discussed when reporting methods and results.
Let us assume that our research aim is to examine the (clinical) processes around acute endovascular treatment (EVT), from the patient’s arrival at the emergency room to recanalization, with the aim to identify possible causes for delay and/or other causes for sub-optimal treatment outcome. As a first step, we could conduct a document study of the relevant standard operating procedures (SOPs) for this phase of care – are they up-to-date and in line with current guidelines? Do they contain any mistakes, irregularities or uncertainties that could cause delays or other problems? Regardless of the answers to these questions, the results have to be interpreted based on what they are: a written outline of what care processes in this hospital should look like. If we want to know what they actually look like in practice, we can conduct observations of the processes described in the SOPs. These results can (and should) be analysed in themselves, but also in comparison to the results of the document analysis, especially as regards relevant discrepancies. Do the SOPs outline specific tests for which no equipment can be observed or tasks to be performed by specialized nurses who are not present during the observation? It might also be possible that the written SOP is outdated, but the actual care provided is in line with current best practice. In order to find out why these discrepancies exist, it can be useful to conduct interviews. Are the physicians simply not aware of the SOPs (because their existence is limited to the hospital’s intranet) or do they actively disagree with them or does the infrastructure make it impossible to provide the care as described? Another rationale for adding interviews is that some situations (or all of their possible variations for different patient groups or the day, night or weekend shift) cannot practically or ethically be observed. In this case, it is possible to ask those involved to report on their actions – being aware that this is not the same as the actual observation. A senior physician’s or hospital manager’s description of certain situations might differ from a nurse’s or junior physician’s one, maybe because they intentionally misrepresent facts or maybe because different aspects of the process are visible or important to them. In some cases, it can also be relevant to consider to whom the interviewee is disclosing this information – someone they trust, someone they are otherwise not connected to, or someone they suspect or are aware of being in a potentially “dangerous” power relationship to them. Lastly, a focus group could be conducted with representatives of the relevant professional groups to explore how and why exactly they provide care around EVT. The discussion might reveal discrepancies (between SOPs and actual care or between different physicians) and motivations to the researchers as well as to the focus group members that they might not have been aware of themselves. For the focus group to deliver relevant information, attention has to be paid to its composition and conduct, for example, to make sure that all participants feel safe to disclose sensitive or potentially problematic information or that the discussion is not dominated by (senior) physicians only. The resulting combination of data collection methods is shown in Fig. 2 .
Possible combination of data collection methods
Attributions for icons: “Book” by Serhii Smirnov, “Interview” by Adrien Coquet, FR, “Magnifying Glass” by anggun, ID, “Business communication” by Vectors Market; all from the Noun Project
The combination of multiple data source as described for this example can be referred to as “triangulation”, in which multiple measurements are carried out from different angles to achieve a more comprehensive understanding of the phenomenon under study [ 22 , 23 ].
Data analysis
To analyse the data collected through observations, interviews and focus groups these need to be transcribed into protocols and transcripts (see Fig. 3 ). Interviews and focus groups can be transcribed verbatim , with or without annotations for behaviour (e.g. laughing, crying, pausing) and with or without phonetic transcription of dialects and filler words, depending on what is expected or known to be relevant for the analysis. In the next step, the protocols and transcripts are coded , that is, marked (or tagged, labelled) with one or more short descriptors of the content of a sentence or paragraph [ 2 , 15 , 23 ]. Jansen describes coding as “connecting the raw data with “theoretical” terms” [ 20 ]. In a more practical sense, coding makes raw data sortable. This makes it possible to extract and examine all segments describing, say, a tele-neurology consultation from multiple data sources (e.g. SOPs, emergency room observations, staff and patient interview). In a process of synthesis and abstraction, the codes are then grouped, summarised and/or categorised [ 15 , 20 ]. The end product of the coding or analysis process is a descriptive theory of the behavioural pattern under investigation [ 20 ]. The coding process is performed using qualitative data management software, the most common ones being InVivo, MaxQDA and Atlas.ti. It should be noted that these are data management tools which support the analysis performed by the researcher(s) [ 14 ].
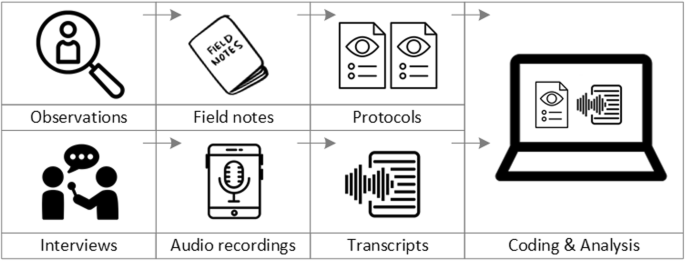
From data collection to data analysis
Attributions for icons: see Fig. 2 , also “Speech to text” by Trevor Dsouza, “Field Notes” by Mike O’Brien, US, “Voice Record” by ProSymbols, US, “Inspection” by Made, AU, and “Cloud” by Graphic Tigers; all from the Noun Project
How to report qualitative research?
Protocols of qualitative research can be published separately and in advance of the study results. However, the aim is not the same as in RCT protocols, i.e. to pre-define and set in stone the research questions and primary or secondary endpoints. Rather, it is a way to describe the research methods in detail, which might not be possible in the results paper given journals’ word limits. Qualitative research papers are usually longer than their quantitative counterparts to allow for deep understanding and so-called “thick description”. In the methods section, the focus is on transparency of the methods used, including why, how and by whom they were implemented in the specific study setting, so as to enable a discussion of whether and how this may have influenced data collection, analysis and interpretation. The results section usually starts with a paragraph outlining the main findings, followed by more detailed descriptions of, for example, the commonalities, discrepancies or exceptions per category [ 20 ]. Here it is important to support main findings by relevant quotations, which may add information, context, emphasis or real-life examples [ 20 , 23 ]. It is subject to debate in the field whether it is relevant to state the exact number or percentage of respondents supporting a certain statement (e.g. “Five interviewees expressed negative feelings towards XYZ”) [ 21 ].
How to combine qualitative with quantitative research?
Qualitative methods can be combined with other methods in multi- or mixed methods designs, which “[employ] two or more different methods [ …] within the same study or research program rather than confining the research to one single method” [ 24 ]. Reasons for combining methods can be diverse, including triangulation for corroboration of findings, complementarity for illustration and clarification of results, expansion to extend the breadth and range of the study, explanation of (unexpected) results generated with one method with the help of another, or offsetting the weakness of one method with the strength of another [ 1 , 17 , 24 , 25 , 26 ]. The resulting designs can be classified according to when, why and how the different quantitative and/or qualitative data strands are combined. The three most common types of mixed method designs are the convergent parallel design , the explanatory sequential design and the exploratory sequential design. The designs with examples are shown in Fig. 4 .
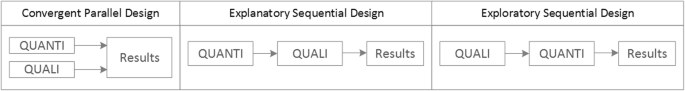
Three common mixed methods designs
In the convergent parallel design, a qualitative study is conducted in parallel to and independently of a quantitative study, and the results of both studies are compared and combined at the stage of interpretation of results. Using the above example of EVT provision, this could entail setting up a quantitative EVT registry to measure process times and patient outcomes in parallel to conducting the qualitative research outlined above, and then comparing results. Amongst other things, this would make it possible to assess whether interview respondents’ subjective impressions of patients receiving good care match modified Rankin Scores at follow-up, or whether observed delays in care provision are exceptions or the rule when compared to door-to-needle times as documented in the registry. In the explanatory sequential design, a quantitative study is carried out first, followed by a qualitative study to help explain the results from the quantitative study. This would be an appropriate design if the registry alone had revealed relevant delays in door-to-needle times and the qualitative study would be used to understand where and why these occurred, and how they could be improved. In the exploratory design, the qualitative study is carried out first and its results help informing and building the quantitative study in the next step [ 26 ]. If the qualitative study around EVT provision had shown a high level of dissatisfaction among the staff members involved, a quantitative questionnaire investigating staff satisfaction could be set up in the next step, informed by the qualitative study on which topics dissatisfaction had been expressed. Amongst other things, the questionnaire design would make it possible to widen the reach of the research to more respondents from different (types of) hospitals, regions, countries or settings, and to conduct sub-group analyses for different professional groups.
How to assess qualitative research?
A variety of assessment criteria and lists have been developed for qualitative research, ranging in their focus and comprehensiveness [ 14 , 17 , 27 ]. However, none of these has been elevated to the “gold standard” in the field. In the following, we therefore focus on a set of commonly used assessment criteria that, from a practical standpoint, a researcher can look for when assessing a qualitative research report or paper.
Assessors should check the authors’ use of and adherence to the relevant reporting checklists (e.g. Standards for Reporting Qualitative Research (SRQR)) to make sure all items that are relevant for this type of research are addressed [ 23 , 28 ]. Discussions of quantitative measures in addition to or instead of these qualitative measures can be a sign of lower quality of the research (paper). Providing and adhering to a checklist for qualitative research contributes to an important quality criterion for qualitative research, namely transparency [ 15 , 17 , 23 ].
Reflexivity
While methodological transparency and complete reporting is relevant for all types of research, some additional criteria must be taken into account for qualitative research. This includes what is called reflexivity, i.e. sensitivity to the relationship between the researcher and the researched, including how contact was established and maintained, or the background and experience of the researcher(s) involved in data collection and analysis. Depending on the research question and population to be researched this can be limited to professional experience, but it may also include gender, age or ethnicity [ 17 , 27 ]. These details are relevant because in qualitative research, as opposed to quantitative research, the researcher as a person cannot be isolated from the research process [ 23 ]. It may influence the conversation when an interviewed patient speaks to an interviewer who is a physician, or when an interviewee is asked to discuss a gynaecological procedure with a male interviewer, and therefore the reader must be made aware of these details [ 19 ].
Sampling and saturation
The aim of qualitative sampling is for all variants of the objects of observation that are deemed relevant for the study to be present in the sample “ to see the issue and its meanings from as many angles as possible” [ 1 , 16 , 19 , 20 , 27 ] , and to ensure “information-richness [ 15 ]. An iterative sampling approach is advised, in which data collection (e.g. five interviews) is followed by data analysis, followed by more data collection to find variants that are lacking in the current sample. This process continues until no new (relevant) information can be found and further sampling becomes redundant – which is called saturation [ 1 , 15 ] . In other words: qualitative data collection finds its end point not a priori , but when the research team determines that saturation has been reached [ 29 , 30 ].
This is also the reason why most qualitative studies use deliberate instead of random sampling strategies. This is generally referred to as “ purposive sampling” , in which researchers pre-define which types of participants or cases they need to include so as to cover all variations that are expected to be of relevance, based on the literature, previous experience or theory (i.e. theoretical sampling) [ 14 , 20 ]. Other types of purposive sampling include (but are not limited to) maximum variation sampling, critical case sampling or extreme or deviant case sampling [ 2 ]. In the above EVT example, a purposive sample could include all relevant professional groups and/or all relevant stakeholders (patients, relatives) and/or all relevant times of observation (day, night and weekend shift).
Assessors of qualitative research should check whether the considerations underlying the sampling strategy were sound and whether or how researchers tried to adapt and improve their strategies in stepwise or cyclical approaches between data collection and analysis to achieve saturation [ 14 ].
Good qualitative research is iterative in nature, i.e. it goes back and forth between data collection and analysis, revising and improving the approach where necessary. One example of this are pilot interviews, where different aspects of the interview (especially the interview guide, but also, for example, the site of the interview or whether the interview can be audio-recorded) are tested with a small number of respondents, evaluated and revised [ 19 ]. In doing so, the interviewer learns which wording or types of questions work best, or which is the best length of an interview with patients who have trouble concentrating for an extended time. Of course, the same reasoning applies to observations or focus groups which can also be piloted.
Ideally, coding should be performed by at least two researchers, especially at the beginning of the coding process when a common approach must be defined, including the establishment of a useful coding list (or tree), and when a common meaning of individual codes must be established [ 23 ]. An initial sub-set or all transcripts can be coded independently by the coders and then compared and consolidated after regular discussions in the research team. This is to make sure that codes are applied consistently to the research data.
Member checking
Member checking, also called respondent validation , refers to the practice of checking back with study respondents to see if the research is in line with their views [ 14 , 27 ]. This can happen after data collection or analysis or when first results are available [ 23 ]. For example, interviewees can be provided with (summaries of) their transcripts and asked whether they believe this to be a complete representation of their views or whether they would like to clarify or elaborate on their responses [ 17 ]. Respondents’ feedback on these issues then becomes part of the data collection and analysis [ 27 ].
Stakeholder involvement
In those niches where qualitative approaches have been able to evolve and grow, a new trend has seen the inclusion of patients and their representatives not only as study participants (i.e. “members”, see above) but as consultants to and active participants in the broader research process [ 31 , 32 , 33 ]. The underlying assumption is that patients and other stakeholders hold unique perspectives and experiences that add value beyond their own single story, making the research more relevant and beneficial to researchers, study participants and (future) patients alike [ 34 , 35 ]. Using the example of patients on or nearing dialysis, a recent scoping review found that 80% of clinical research did not address the top 10 research priorities identified by patients and caregivers [ 32 , 36 ]. In this sense, the involvement of the relevant stakeholders, especially patients and relatives, is increasingly being seen as a quality indicator in and of itself.
How not to assess qualitative research
The above overview does not include certain items that are routine in assessments of quantitative research. What follows is a non-exhaustive, non-representative, experience-based list of the quantitative criteria often applied to the assessment of qualitative research, as well as an explanation of the limited usefulness of these endeavours.
Protocol adherence
Given the openness and flexibility of qualitative research, it should not be assessed by how well it adheres to pre-determined and fixed strategies – in other words: its rigidity. Instead, the assessor should look for signs of adaptation and refinement based on lessons learned from earlier steps in the research process.
Sample size
For the reasons explained above, qualitative research does not require specific sample sizes, nor does it require that the sample size be determined a priori [ 1 , 14 , 27 , 37 , 38 , 39 ]. Sample size can only be a useful quality indicator when related to the research purpose, the chosen methodology and the composition of the sample, i.e. who was included and why.
Randomisation
While some authors argue that randomisation can be used in qualitative research, this is not commonly the case, as neither its feasibility nor its necessity or usefulness has been convincingly established for qualitative research [ 13 , 27 ]. Relevant disadvantages include the negative impact of a too large sample size as well as the possibility (or probability) of selecting “ quiet, uncooperative or inarticulate individuals ” [ 17 ]. Qualitative studies do not use control groups, either.
Interrater reliability, variability and other “objectivity checks”
The concept of “interrater reliability” is sometimes used in qualitative research to assess to which extent the coding approach overlaps between the two co-coders. However, it is not clear what this measure tells us about the quality of the analysis [ 23 ]. This means that these scores can be included in qualitative research reports, preferably with some additional information on what the score means for the analysis, but it is not a requirement. Relatedly, it is not relevant for the quality or “objectivity” of qualitative research to separate those who recruited the study participants and collected and analysed the data. Experiences even show that it might be better to have the same person or team perform all of these tasks [ 20 ]. First, when researchers introduce themselves during recruitment this can enhance trust when the interview takes place days or weeks later with the same researcher. Second, when the audio-recording is transcribed for analysis, the researcher conducting the interviews will usually remember the interviewee and the specific interview situation during data analysis. This might be helpful in providing additional context information for interpretation of data, e.g. on whether something might have been meant as a joke [ 18 ].
Not being quantitative research
Being qualitative research instead of quantitative research should not be used as an assessment criterion if it is used irrespectively of the research problem at hand. Similarly, qualitative research should not be required to be combined with quantitative research per se – unless mixed methods research is judged as inherently better than single-method research. In this case, the same criterion should be applied for quantitative studies without a qualitative component.
The main take-away points of this paper are summarised in Table 1 . We aimed to show that, if conducted well, qualitative research can answer specific research questions that cannot to be adequately answered using (only) quantitative designs. Seeing qualitative and quantitative methods as equal will help us become more aware and critical of the “fit” between the research problem and our chosen methods: I can conduct an RCT to determine the reasons for transportation delays of acute stroke patients – but should I? It also provides us with a greater range of tools to tackle a greater range of research problems more appropriately and successfully, filling in the blind spots on one half of the methodological spectrum to better address the whole complexity of neurological research and practice.
Availability of data and materials
Not applicable.
Abbreviations
Endovascular treatment
Randomised Controlled Trial
Standard Operating Procedure
Standards for Reporting Qualitative Research
Philipsen, H., & Vernooij-Dassen, M. (2007). Kwalitatief onderzoek: nuttig, onmisbaar en uitdagend. In L. PLBJ & H. TCo (Eds.), Kwalitatief onderzoek: Praktische methoden voor de medische praktijk . [Qualitative research: useful, indispensable and challenging. In: Qualitative research: Practical methods for medical practice (pp. 5–12). Houten: Bohn Stafleu van Loghum.
Chapter Google Scholar
Punch, K. F. (2013). Introduction to social research: Quantitative and qualitative approaches . London: Sage.
Kelly, J., Dwyer, J., Willis, E., & Pekarsky, B. (2014). Travelling to the city for hospital care: Access factors in country aboriginal patient journeys. Australian Journal of Rural Health, 22 (3), 109–113.
Article Google Scholar
Nilsen, P., Ståhl, C., Roback, K., & Cairney, P. (2013). Never the twain shall meet? - a comparison of implementation science and policy implementation research. Implementation Science, 8 (1), 1–12.
Howick J, Chalmers I, Glasziou, P., Greenhalgh, T., Heneghan, C., Liberati, A., Moschetti, I., Phillips, B., & Thornton, H. (2011). The 2011 Oxford CEBM evidence levels of evidence (introductory document) . Oxford Center for Evidence Based Medicine. https://www.cebm.net/2011/06/2011-oxford-cebm-levels-evidence-introductory-document/ .
Eakin, J. M. (2016). Educating critical qualitative health researchers in the land of the randomized controlled trial. Qualitative Inquiry, 22 (2), 107–118.
May, A., & Mathijssen, J. (2015). Alternatieven voor RCT bij de evaluatie van effectiviteit van interventies!? Eindrapportage. In Alternatives for RCTs in the evaluation of effectiveness of interventions!? Final report .
Google Scholar
Berwick, D. M. (2008). The science of improvement. Journal of the American Medical Association, 299 (10), 1182–1184.
Article CAS Google Scholar
Christ, T. W. (2014). Scientific-based research and randomized controlled trials, the “gold” standard? Alternative paradigms and mixed methodologies. Qualitative Inquiry, 20 (1), 72–80.
Lamont, T., Barber, N., Jd, P., Fulop, N., Garfield-Birkbeck, S., Lilford, R., Mear, L., Raine, R., & Fitzpatrick, R. (2016). New approaches to evaluating complex health and care systems. BMJ, 352:i154.
Drabble, S. J., & O’Cathain, A. (2015). Moving from Randomized Controlled Trials to Mixed Methods Intervention Evaluation. In S. Hesse-Biber & R. B. Johnson (Eds.), The Oxford Handbook of Multimethod and Mixed Methods Research Inquiry (pp. 406–425). London: Oxford University Press.
Chambers, D. A., Glasgow, R. E., & Stange, K. C. (2013). The dynamic sustainability framework: Addressing the paradox of sustainment amid ongoing change. Implementation Science : IS, 8 , 117.
Hak, T. (2007). Waarnemingsmethoden in kwalitatief onderzoek. In L. PLBJ & H. TCo (Eds.), Kwalitatief onderzoek: Praktische methoden voor de medische praktijk . [Observation methods in qualitative research] (pp. 13–25). Houten: Bohn Stafleu van Loghum.
Russell, C. K., & Gregory, D. M. (2003). Evaluation of qualitative research studies. Evidence Based Nursing, 6 (2), 36–40.
Fossey, E., Harvey, C., McDermott, F., & Davidson, L. (2002). Understanding and evaluating qualitative research. Australian and New Zealand Journal of Psychiatry, 36 , 717–732.
Yanow, D. (2000). Conducting interpretive policy analysis (Vol. 47). Thousand Oaks: Sage University Papers Series on Qualitative Research Methods.
Shenton, A. K. (2004). Strategies for ensuring trustworthiness in qualitative research projects. Education for Information, 22 , 63–75.
van der Geest, S. (2006). Participeren in ziekte en zorg: meer over kwalitatief onderzoek. Huisarts en Wetenschap, 49 (4), 283–287.
Hijmans, E., & Kuyper, M. (2007). Het halfopen interview als onderzoeksmethode. In L. PLBJ & H. TCo (Eds.), Kwalitatief onderzoek: Praktische methoden voor de medische praktijk . [The half-open interview as research method (pp. 43–51). Houten: Bohn Stafleu van Loghum.
Jansen, H. (2007). Systematiek en toepassing van de kwalitatieve survey. In L. PLBJ & H. TCo (Eds.), Kwalitatief onderzoek: Praktische methoden voor de medische praktijk . [Systematics and implementation of the qualitative survey (pp. 27–41). Houten: Bohn Stafleu van Loghum.
Pv, R., & Peremans, L. (2007). Exploreren met focusgroepgesprekken: de ‘stem’ van de groep onder de loep. In L. PLBJ & H. TCo (Eds.), Kwalitatief onderzoek: Praktische methoden voor de medische praktijk . [Exploring with focus group conversations: the “voice” of the group under the magnifying glass (pp. 53–64). Houten: Bohn Stafleu van Loghum.
Carter, N., Bryant-Lukosius, D., DiCenso, A., Blythe, J., & Neville, A. J. (2014). The use of triangulation in qualitative research. Oncology Nursing Forum, 41 (5), 545–547.
Boeije H: Analyseren in kwalitatief onderzoek: Denken en doen, [Analysis in qualitative research: Thinking and doing] vol. Den Haag Boom Lemma uitgevers; 2012.
Hunter, A., & Brewer, J. (2015). Designing Multimethod Research. In S. Hesse-Biber & R. B. Johnson (Eds.), The Oxford Handbook of Multimethod and Mixed Methods Research Inquiry (pp. 185–205). London: Oxford University Press.
Archibald, M. M., Radil, A. I., Zhang, X., & Hanson, W. E. (2015). Current mixed methods practices in qualitative research: A content analysis of leading journals. International Journal of Qualitative Methods, 14 (2), 5–33.
Creswell, J. W., & Plano Clark, V. L. (2011). Choosing a Mixed Methods Design. In Designing and Conducting Mixed Methods Research . Thousand Oaks: SAGE Publications.
Mays, N., & Pope, C. (2000). Assessing quality in qualitative research. BMJ, 320 (7226), 50–52.
O'Brien, B. C., Harris, I. B., Beckman, T. J., Reed, D. A., & Cook, D. A. (2014). Standards for reporting qualitative research: A synthesis of recommendations. Academic Medicine : Journal of the Association of American Medical Colleges, 89 (9), 1245–1251.
Saunders, B., Sim, J., Kingstone, T., Baker, S., Waterfield, J., Bartlam, B., Burroughs, H., & Jinks, C. (2018). Saturation in qualitative research: Exploring its conceptualization and operationalization. Quality and Quantity, 52 (4), 1893–1907.
Moser, A., & Korstjens, I. (2018). Series: Practical guidance to qualitative research. Part 3: Sampling, data collection and analysis. European Journal of General Practice, 24 (1), 9–18.
Marlett, N., Shklarov, S., Marshall, D., Santana, M. J., & Wasylak, T. (2015). Building new roles and relationships in research: A model of patient engagement research. Quality of Life Research : an international journal of quality of life aspects of treatment, care and rehabilitation, 24 (5), 1057–1067.
Demian, M. N., Lam, N. N., Mac-Way, F., Sapir-Pichhadze, R., & Fernandez, N. (2017). Opportunities for engaging patients in kidney research. Canadian Journal of Kidney Health and Disease, 4 , 2054358117703070–2054358117703070.
Noyes, J., McLaughlin, L., Morgan, K., Roberts, A., Stephens, M., Bourne, J., Houlston, M., Houlston, J., Thomas, S., Rhys, R. G., et al. (2019). Designing a co-productive study to overcome known methodological challenges in organ donation research with bereaved family members. Health Expectations . 22(4):824–35.
Piil, K., Jarden, M., & Pii, K. H. (2019). Research agenda for life-threatening cancer. European Journal Cancer Care (Engl), 28 (1), e12935.
Hofmann, D., Ibrahim, F., Rose, D., Scott, D. L., Cope, A., Wykes, T., & Lempp, H. (2015). Expectations of new treatment in rheumatoid arthritis: Developing a patient-generated questionnaire. Health Expectations : an international journal of public participation in health care and health policy, 18 (5), 995–1008.
Jun, M., Manns, B., Laupacis, A., Manns, L., Rehal, B., Crowe, S., & Hemmelgarn, B. R. (2015). Assessing the extent to which current clinical research is consistent with patient priorities: A scoping review using a case study in patients on or nearing dialysis. Canadian Journal of Kidney Health and Disease, 2 , 35.
Elsie Baker, S., & Edwards, R. (2012). How many qualitative interviews is enough? In National Centre for Research Methods Review Paper . National Centre for Research Methods. http://eprints.ncrm.ac.uk/2273/4/how_many_interviews.pdf .
Sandelowski, M. (1995). Sample size in qualitative research. Research in Nursing & Health, 18 (2), 179–183.
Sim, J., Saunders, B., Waterfield, J., & Kingstone, T. (2018). Can sample size in qualitative research be determined a priori? International Journal of Social Research Methodology, 21 (5), 619–634.
Download references
Acknowledgements
no external funding.
Author information
Authors and affiliations.
Department of Neurology, Heidelberg University Hospital, Im Neuenheimer Feld 400, 69120, Heidelberg, Germany
Loraine Busetto, Wolfgang Wick & Christoph Gumbinger
Clinical Cooperation Unit Neuro-Oncology, German Cancer Research Center, Heidelberg, Germany
Wolfgang Wick
You can also search for this author in PubMed Google Scholar
Contributions
LB drafted the manuscript; WW and CG revised the manuscript; all authors approved the final versions.
Corresponding author
Correspondence to Loraine Busetto .
Ethics declarations
Ethics approval and consent to participate, consent for publication, competing interests.
The authors declare no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ .
Reprints and permissions
About this article
Cite this article.
Busetto, L., Wick, W. & Gumbinger, C. How to use and assess qualitative research methods. Neurol. Res. Pract. 2 , 14 (2020). https://doi.org/10.1186/s42466-020-00059-z
Download citation
Received : 30 January 2020
Accepted : 22 April 2020
Published : 27 May 2020
DOI : https://doi.org/10.1186/s42466-020-00059-z
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Qualitative research
- Mixed methods
- Quality assessment
Neurological Research and Practice
ISSN: 2524-3489
- Submission enquiries: Access here and click Contact Us
- General enquiries: [email protected]
Qualitative Text Analysis: A Systematic Approach
- Open Access
- First Online: 27 April 2019
Cite this chapter
You have full access to this open access chapter

- Udo Kuckartz 4
Part of the book series: ICME-13 Monographs ((ICME13Mo))
121k Accesses
211 Citations
3 Altmetric
Thematic analysis, often called Qualitative Content Analysis (QCA) in Europe, is one of the most commonly used methods for analyzing qualitative data. This paper presents the basics of this systematic method of qualitative data analysis, highlights its key characteristics, and describes a typical workflow. The aim is to present the main characteristics and to give a simple example of the process so that readers can assess whether this method might be useful for their own research. Special attention is paid to the formation of categories, since all scholars agree that categories are at the heart of the method.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others

Qualitative Content Analysis: Theoretical Background and Procedures

Qualitative Methodology

Analysis and Synthesis
- Qualitative data analysis
- Text analysis
- Qualitative methods
- Qualitative content analysis
- MAXQDA software
1 Introduction: Qualitative and Quantitative Data
Thematic analysis, often called Qualitative Content Analysis (QCA) in Europe, is one of the most commonly used methods for analyzing qualitative data (Guest et al. 2012 ; Kuckartz 2014 ; Mayring 2014 , 2015 ; Schreier 2012 ). This chapter presents the basics of this systematic method of qualitative data analysis, highlights its key characteristics, and describes a typical workflow.
Working with codes and categories is a proven method in qualitative research. QCA is a method that is reliable, easy to learn, transparent, and it is a method that is easily understood by other researchers. In short, it is a method that enjoys a high level of recognition and is to be highly recommended, especially in the context of dissertations.
The aim of this paper is to present the main characteristics and to give a simple example of the process so that readers can assess whether this method might be useful for their own research. Special attention is paid to the formation of categories, since all scholars agree that categories are at the heart of the method.
Let’s start with some of the basics of data analysis in empirical research: What does ‘qualitative data’ mean, and what do we mean by ‘quantitative data’? Quantitative data entail numerical information that results, for example, from the collection of data from a standardized interview. In a quantitative data matrix, each row corresponds to a case, namely, an interview with a respondent. The columns of the matrix are formed by the variables. Table 8.1 therefore shows the data of four cases, here the respondents 1–4. Six variables were collected for these individuals, on a scale of 1–6, concerning how often they perform certain household activities (laundry, small repairs etc.). Typically, these kinds of data sets are available in social research in the form of a rectangular matrix, for instance as shown in Table 8.1 .
A matrix like this that consists of numbers can be analyzed using statistical methods. For example, you can calculate univariate statistics such as mean values, variance, and standard deviations. You can also generate graphical displays such as box plots or bar charts. In addition, variables can be related to each other, for example by using methods of correlation and regression statistics. Another form of analysis tests groups for differences. In the above study, for example, the questions ‘Are women more frequently engaged in laundry than men in the household?’ and ‘Are men more frequently engaged in minor repairs than women in the household?’ can be calculated using an analysis of variance.
Qualitative data are far more diverse and complex than quantitative data. These data may comprise transcripts of face-to-face interviews or focus group discussions, documents, Twitter tweets, YouTube comments, or videos of the teacher-student interactions in the classroom.
In this chapter, I restrict the presentation of the QCA method to a specific type of data, namely qualitative interviews. This collective term can be used to describe very different forms of interviews, such as guideline-assisted interviews or narrative interviews on critical life events conducted in the context of biographical research. The latter can last several hours and comprise more than 30 pages as a transcription. A qualitative interview may also consist of a short online survey, like the one I conducted in preparation for my workshop at the International Congress on Mathematical Education (ICME-13).
Obviously, the different types of qualitative data are not as easy to analyze as the numbers in a quantitative data matrix. Numerous analytical methods have been developed in qualitative research, among them the well-proven method of qualitative content analysis.
2 Key Points of Qualitative Content Analysis
What are the key points of the qualitative content analysis method? Regardless of which variant of QCA is used, the focus will always be on working with categories (codes) and developing a category system (coding frame). What Berelson formulated in 1952 for quantitative content analysis still applies today, both to quantitative and qualitative content analysis:
Content analysis stands or falls by its categories (…) since the categories contain the substance of the investigation, a content analysis can be no better than its system of categories. (Berelson 1952 , p. 147)
Categories are therefore of crucial importance for effective research, not only in their role as analysis tools, but also insofar as they form the substance of the research and the building blocks of the theory the researchers want to develop. That raises the question ‘What are categories?’—or more precisely, ‘What are categories in the context of empirical social research?’ Answering this question is by no means easy and there are at least two ways of doing so. The first way can be described as phenomenological : Kuckartz ( 2016 , pp. 31–39) focuses on the use of this term in the practice of empirical social research, i.e., drawing attention to what is called a category in empirical social research. The result of this analysis is a very diverse spectrum, whereby several different types of categories can be distinguished in social science research literature (ibid., pp. 34–35):
Factual categories denote actual or supposed objective circumstances such as ‘length of training’ or ‘occupation’.
Thematic categories refer to certain topics, arguments, schools of thought etc. such as ‘inclusion’, ‘environmental justice’ or ‘Ukrainian conflict’.
Evaluative categories are related to an evaluation scale—usually ordinal types, for example the category ‘helper syndrome’ with the characteristics ‘not pronounced’, ‘somewhat pronounced’ and ‘pronounced’. For evaluative categories, it is the researchers who classify the data according to predefined criteria.
Analytical categories are the result of intensive analysis of the data, i.e., these categories move away from the description of the data, for example by means of thematic categories.
Theoretical categories are subspecies of analytical categories that refer to an existing theory, such as Ajzen’s theory of planned behavior, Ainsworth’s attachment theory, or Foucault’s analysis of power.
Natural categories , also called “in vivo codes” (Charmaz 2006 , p. 56; Kuckartz 2014 , p. 23), are terms used by the actors in the field.
Formal categories denote formal characteristics of an analysis unit, e.g., the length of time in an interview.
The above list is not complete; there are many more types of categories and corresponding methods of coding (Saldana 2015 ).
A second way of answering the question ‘What is a category?’ can be described as conceptual and historical; this way leads us far back into the history of philosophy. The conceptual historical view of the term, originating from ancient Greece, starts with Greek philosophy more than 2000 years ago. Plato and Aristotle already dealt with categories—Aristotle even in an elaboration of the same term (“categories”). The study of categories runs through Western philosophy from Plato and Kant to Peirce and analytical philosophy. The philosophers are by no means in agreement on the concept of categories, but a discussion of the differences between the different schools would far exceed the scope of this paper; Instead, reading the mostly very extensive contributions on the terms ‘category’ and ‘category theory’ in the various lexicons of philosophy is recommended. Categories are basic concepts of cognition; they are—generally speaking—a commonality between certain things: a term, a heading, a label that designates something similar under certain aspects. Categories also play this role in content analysis, as the following quote from the Content Analysis textbook of Früh ( 2004 ) demonstrates:
The pragmatic sense of any content analysis is ultimately to reduce complexity from a certain research-led perspective. Text sets are described in a classifying manner with regard to characteristics of theoretical interest. In this reduction of complexity, information is necessarily lost: On the one hand, information is lost due to the suppression of message characteristics that are present in the examined texts but are not of interest in connection with the present research question; on the other hand, information is lost due to the classification of the analyzed message characteristics. According to specified criteria, some of them are each considered similar to one another and assigned to a certain characteristic class or a characteristic type, which is called ‘category’ in the content analysis. The original differences in meaning of the message characteristics uniformly grouped in a category shall not be taken into account. (p. 42, translated by the author)
But how does qualitative content analysis arrive at its categories, the basic building blocks for forming theory? There are three principal ways to develop categories:
Concept-driven (‘deductive’) development of categories; in this case the categories
are derived from a theory or
derived from the literature (the current state of research) or
derived from the research question (e.g. directly related to an interview guide)
Data-driven (‘inductive’) development of categories; the characteristics here are
the step-by-step procedure,
the method of open coding until saturation occurs,
the continuous organization and systematization of the formed codes, and
the development of top-level codes and subcodes at different levels.
Mixing a concept-driven and data-driven development of codes:
The starting point here is usually a coding frame with deductively formed codes and
the subsequent inductive coding of all data coded with a specific main category.
The terms deductive and inductive are often used for the concept-driven and data-driven approaches, respectively. However, the use of the term ‘deductive’ is rather problematic in this context: In scientific logic, the term ‘inductive’ refers to the abstract conclusion from what has been observed empirically to a general rule or a law; this has little to do with the formation of categories based on empirical data. The situation is similar with the term ‘deductive’: In scientific logic, the deductive conclusion is a logical consequence of its premises; the formation of categories based on the state of research, a theory, or an advanced hypothesis is very different. Categories do not necessarily emerge from a systematic literature review or from a research question. Due to its skid resistance, however, the word pair ‘inductive-deductive’ will probably remain in the language theorem of empirical social research or the formation of categories for a long time to come. Nevertheless, I try to avoid the terms inductive and deductive, and—like Schreier ( 2012 , p. 84)—prefer the terms ‘data-driven’ and ‘concept-driven’ for these different approaches to the formation of categories.
The decisive action in QCA is the coding of the data, i.e. a precisely defined part of the material is selected, and a category is assigned. As shown in the following figure, this may be a passage from an interview. Here, paragraph 15 of the text was coded with the code Simultaneousness (Fig. 8.1 ).

Text passage with a coded text segment
The individuals who perform this segmentation and coding of the data are referred to as coders. In this context, we also speak of “inter- and intracoder agreement” (reliability) (Krippendorff 2012 ; Kuckartz 2016 ; Schreier 2012 ). In quantitative content analysis, the units to be coded are usually defined in advance and referred to as coding units. In qualitative content analysis, on the other hand, coding units are not usually defined in advance; they are created by the coding process.
The general workflow of a qualitative content analysis is in Fig. 8.2 . In all variants the research question plays the central role in this method: It provides the perspective for the textual work necessary at the beginning, that is, the intensive reading and study of the texts (Kuckartz 2016 , p. 45). For qualitative methods, it is common for the individual analysis phases to be carried out on a circular basis. This also applies to QCA: The creation of categories and subcategories and the coding of the data can take place in several cycles. Saldana ( 2015 ) speaks of first cycle coding and second cycle coding, for example. The number of cycles is not fixed, and only in rare cases would one get by with just a single cycle.
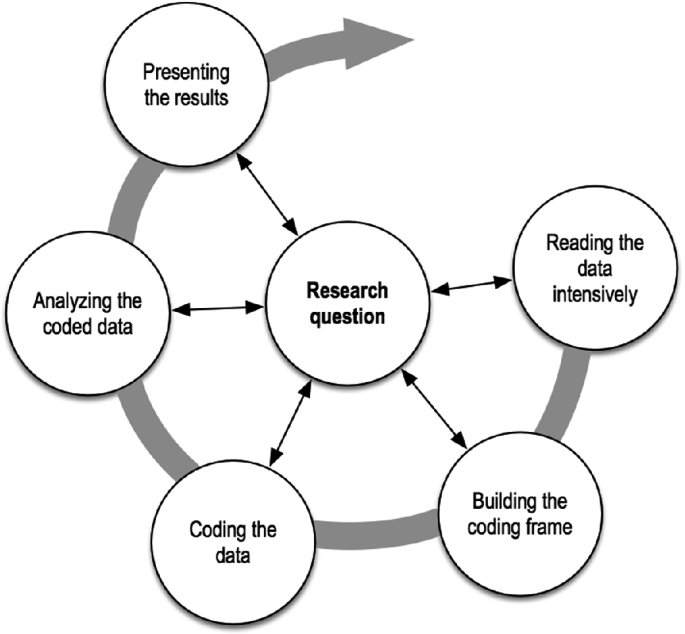
The five phases of qualitative content analysis
Once all the data have been coded with the final category frame, a systematization and structuring of all the relevant data in view of the research questions at hand will have been achieved. Table 8.2 illustrates a model of such a thematic matrix. It is similar to the quantitative data matrix shown in Fig. 8.2 , but instead of containing numbers, the cells of the matrix now contain text excerpts coded with the respective corresponding category.
The further analysis of the matrix can now take two directions: If you look at columns, you can examine certain topics. These forms of analysis can be described as ‘category-based’. Looking at the rows, you can focus on cases (people) and carry out a ‘case-oriented analysis’.
Category-based analyses can focus on a specific category or even consider several categories simultaneously. For example, the statements made by the research participants can be contrasted between two or across several topics. Such complex analyses can lead to very rich descriptions or to the determination of influencing factors and effects, which can then be displayed in a concept map. Case-oriented analyses allow you to identify similarities between cases, identify extreme cases, and form types. Methods of consistently comparing and contrasting cases can be used to this end. For example, if you have determined a typology, you can then visualize it as a constellation of clusters and cases.
3 The Analysis Process in Detail
The example used in the following is a short online survey conducted in preparation for the ‘Workshop on qualitative text analysis’ as part of the ICME 13. The aim of the survey was to provide an overview of the research needs of the participants and their level of knowledge. In other words, its aim was descriptive and not about the development of hypotheses or a theory. In this online interview, I asked the following five questions and asked the participants to write their responses directly below the questions. Table 8.3 contains the resulting qualitative data.
Typically, QCA consists of six steps
Step 1: Preparing the data, initiating text work
Step 2: Forming main categories corresponding to the questions asked in the interview
Step 3: Coding data with the main categories
Step 4: Compiling text passages of the main categories and forming subcategories inductively on the material; assigning text passages to subcategories
Step 5: Category-based analyses and presenting results
Step 6: Reporting and documentation.
Since the purpose of the survey in this case was to get an overview of the relevant interests of the workshop participants and to tailor the workshop to their needs, the last step was omitted. There was no need for reporting and documentation.
The first phase consists of preparing of the data and conducting an initial read-through the responses; the analysis of this short survey did not require extensive interpretation of the responses. Since respondents used different fonts and font sizes in their e-mails, these had to be standardized first when preparing the data. In addition, the overall formatting was also adjusted to render it more uniform across responses. This would not have been absolutely necessary for the analysis, but without this preparation, later compilations of coded text passages might have looked rather chaotic.
In the second phase of QCA, categories are formed. When analyzing data obtained through an online survey, it is best to create a set of main categories based on the questions asked. In this analysis, the following five categories were formed for the first coding cycle:
Motives and goals
Experience with QCA
Specific questions about QCA
Experience with QDAS ( Q ualitative d ata a nalysis s oftware)
Academic discipline.
Since the questions in the online survey were numbered, the numbers were retained for better orientation, but they could have been dispensed with without any problems.
According to the differentiation of categories laid out earlier in this paper, the categories Motives and goals and Specific questions about QCA are thematic categories. Category 5 Academic Discipline is a factual code. The other two categories Experience with QCA and Experience with QDAS are about the experiences with the method and with QDA software. If the researcher is interested in the extent of participants’ experience, both categories are evaluative categories; alternatively, if the specific type of experience is the primary point of interest, the categories are thematic. Since the aim of this survey was to get an overview of the level of knowledge and practical experience of the respondents, an overview was sufficient; detailed knowledge of the types of experience the participants had gained was not absolutely necessary. Reading the responses also demonstrated that the respondents understood the question in this sense and that in most cases no specific details were provided. In any case, working with software like MAXQDA guarantees that you can always return to the original texts should this be useful or necessary during the course of the analysis.
In the third phase of the analysis, the corresponding text segments are coded with the five main categories. Figure 8.3 shows a screenshot of the software MAXQDA after this first cycle of coding was performed on the survey responses. The assignments of the codes are displayed to the left of the corresponding text sections.

Display of a text with code assignments after the first cycle of coding
In the following fourth phase of the analysis, the coding frame is developed further. To do this, all the text passages coded with one of the main categories are first compiled, a procedure which is also referred to as retrieval . Subcodes are then developed directly in the relation to this data—in other words, the creation of categories is data-driven. This process is described in the following with regard to the first main category Motives and goals :
The category Motives and goals coded the responses to the question regarding what the participants wanted to learn in the workshop. First, all text passages to which this category was assigned were compiled. Then each of these text passages was coded a second time. This was done with a procedure similar to that of open coding in Grounded Theory (Strauss and Corbin 1990 ). In this case, the codes were short sequences of words that described what the participants wanted to learn:
analyze mathematics textbook curricula
learn type-building analysis
analyze e-portfolios and group discussions
analyze responses to open-ended questions
learn more about different research methods
how to establish credibility in practice
learn more about rigor within the process and how to ensure its validity
the role of reliability coefficients
insight into conducting qualitative research
learn about the QCA method
how to code video transcripts
how to take the richness of data into account (not only numbers)
analyze large numbers of open questions
learn more about a few different approaches to choose from
searching for a suitable method to analyze the interviews
interesting for me to see how colleagues are working.
As part of the software MAXQDA there is a module called “Creative Coding” that allows you to visually group codes obtained through the open coding method. After arranging the open codes, seven subcategories were created for the category “Motives and Objectives”, namely
Getting an overview of qualitative research
Getting an overview of QCA
Learning basic techniques
Learning about special type of analysis
Reliability and validity
Learning to analyze special types of data
Interesting for me to see how colleagues are working.
Figure 8.4 shows a visual display of the category formation; the original statements are assigned to the respective category. It turns out that many participants in the workshop were mainly interested in obtaining an overview of qualitative content analysis and qualitative research in general. The graph also implicitly illustrates the differences between a quantitative and qualitative analysis of the responses: Four participants (a comparatively large proportion) wanted to learn how to analyze specific types of data, but a closer look at the details, that is, the qualitative dimension, reveals that the types of data the respondents had in mind were completely different.
Visualization of the motives grouped into subcategories
Once the subcategories have been created, all the data coded with the main category Motives and goals must be coded a second time. This is also known as the second coding cycle. In this sample survey, all the coded text passages were included in the formation of the subcategories due to the relatively small sample. In the case of small sample sizes like this, the Creative Coding module automatically reassigns the subcategories. In the case of larger samples, however, category formation will usually be carried out only with a subsample and not with all the data, or the process of open coding will be performed only until the system of subcategories appears saturated and no further subcategories need to be redefined. Then, of course, the data that have not been considered up to this point must still be coded in line with the final category system.
The two categories Experience with QCA and Experience with QDAS were used to code the text passages in which the respondents reported on their experience with the QCA method and the use of QDA software. For the purposes of preparing the workshop as described above, the analysis should address only whether participants had prior experience and how extensive this experience was. An evaluative category with the values ‘yes’, ‘partial’, ‘no’ was therefore defined.
For the third main category, Specific questions about QCA , no subcategories were formed, since the questions formulated by the participants had to be retained in their wording to answer them in the workshop. However, the questions asked were sorted by topic, and essentially identical questions were summarized.
For category 5, Academic discipline , subcategories were initially formed according to the disciplines mentioned by the respondents. However, it quickly transpired that almost all participants came from the field of mathematics education and that there were only a few individual cases from other fields such as development psychology or primary school teacher (see Fig. 8.5 ). These individual cases were combined into the subcategory others for the final category system, so that ultimately only two subcategories were formed.

Main category “Academic discipline and status” with subcategories
After the main categories have been processed in this way—five in the case of this survey—the fifth phase ‘Category-based analyses and presenting results’ can begin. However, it should be clear that in the fourth phase of the development of the category system, an extensive amount of analytical work has already being carried out. The identification of the different motive types represents an analytical achievement in itself and is, at the same time, the foundation of the corresponding category-based analysis in phase 5. The category Motives and goals was of central importance in this survey. In addition to identifying the various motives, both quantitative and qualitative analyses can now be carried out. Quantitatively, we can determine how many people expressed which motives in their statement. Of course, it is quite possible for someone to have expressed several motives. In terms of a qualitative analysis, we can ask what is behind these categories in greater detail. In relation to the subcategory Learning to analyze special types of data , for example, we could ask which special data types the respondents had in mind here.
The category-based analysis always offers the option of focusing on qualitative and/or quantitative aspects. A frequency analysis of the category Experience with QDAS shows that the vast majority of participants have not yet had any practical experience with QDA software (see Fig. 8.6 ).
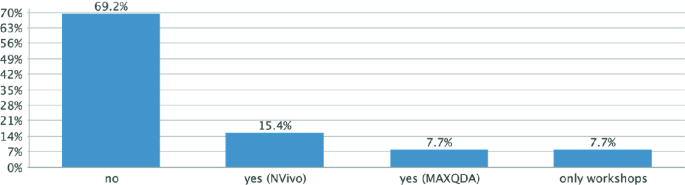
Bar chart of the category “Experiences with QDA software”
The question concerning their experience with text analysis methods presents a somewhat different picture. Quantitatively, we can see that more people are experienced in this regard, while the more detailed qualitative view reveals that this experience mainly involved the Grounded Theory method. It is interesting to compare the two categories that deal with experience. Table 8.4 contains an excerpt from such a comparison between five people.
There are also many further possibilities regarding the analysis of interrelationships that can be carried out in this fifth phase. For example, the connection between motives and goals, and previous knowledge and experience, can be examined. In relation to the specific questions asked by respondents in the survey, one could create a cross table (or “crosstab”) in which the questions asked by the experienced group are compared with the questions asked by those with no experience.
There are many other analysis options for larger studies than those presented for the small online survey. Qualitative content analysis is not a method that is always applied in the same way regardless of the data or research questions at hand. Although it is a systematic procedure, it nonetheless offers a flexibility that allows you to adapt it to the respective requirements of a project. There are other analytical possibilities in this regard, which were not mentioned in the above description. Among these, two should be highlighted in particular, namely, the possibility of paraphrasing text passages and the possibility of creating thematic summaries.
Paraphrasing passages of text can be understood in its everyday sense, namely, that researchers reformulate these text passages in their own words. This can be a very useful tool for category development. This technique is especially recommended for beginners, as it forces them to read the text line by line, interpret it to gain a thorough understanding, and then record it in their own words. It is certainly too time-consuming in most cases to edit all texts in this way but paraphrasing a selected subset of texts can sharpen your analytical view and be a valuable intermediate step in the development of a meaningful category system. Moreover, these paraphrases can then be sorted, particularly significant paraphrases can be combined, and gradually more abstract and theoretically rich categories can be formed.
In contrast to paraphrasing texts, formulating thematic summaries assumes that the texts have already been coded. In this approach, all the text passages coded in regard to a specific topic are read for each case and a thematic summary is written for each person. Usually, there is a huge gap between a category and the amount of original text assigned to it in the case of longer qualitative interviews, such as narrative interviews. On the one hand there is a relatively short code, such as ‘Environmental behavior in relation to nutrition’, and on the other there are numerous passages of varying length in which a respondent says something on this subject. A thematic summary summarizes all these passages as said by a certain person from the perspective of the research question. This means that the text is not repeated, but rather edited conceptually. Summaries thus create a second level between the original text and the categories and concepts. They also enable complex analyses to be carried out in which several categories are compared or the statements of different groups (women/men, different age groups, different schooling, etc.) are contrasted. This would be nigh impossible if the original quotations were always used since the amount of text would simply be too large, and it would consequently not be possible to create case overviews. A thematic summary, on the other hand, compresses what one person has said in such a way that it can easily be included in further analyses.
A third possibility the QCA method offers is the visualization of relationships between categories. Diagrams, in the form of concept maps, can be generated in which the influencing factors, effects, and relations are visualized.
Phase 6, ‘Reporting and documentation’, is about putting the results of your analyses on paper. The research report of a project working with the QCA method is usually divided into a descriptive and an analytical section. Depending on the method and the significance of the categories, category-based analyses will be the center of attention. The case dimension, however, which is all too often neglected, should also be taken into account in the report. It is often very valuable for the recipients of the research not only to learn something about the connections between the categories, but also something about the participants, that is, the cases that are consciously selected for such a presentation. It is particularly interesting if the cases are grouped into types and the report presents cases that are representative of these types.
The category-based presentation should be illustrated with quotes from the original material. However, you should also be aware of the danger of selective plausibility, i.e., that one mainly selects quotations that clarify the alleged connections between categories, while contradictory examples are not considered. For this reason, counterexamples should always be sought and included in the report.
Category-based analysis should not be limited to a description of the results per category but should also look at the relationships between two or more categories. In other words, you should move from the initial description to the development of a theory.
4 Summary and Conclusions
This chapter presents a method for the methodically controlled analysis of texts in empirical research. To conclude, therefore, the characteristics of the QCA method are concisely summarized:
The focus of the QCA method is on the categories with which the data are coded.
The categories of the final coding frame are described as precisely as possible and it is ensured that the coding procedure itself is reliable, i.e., that different coders concur in their coding.
The data must be coded completely. Complete in this sense means that all passages in the texts that are relevant to the research question are coded. It does, however, make sense to leave those parts of the data uncoded, which are outside the focus of the research question.
The codes and categories can be formed in different ways: empirically, i.e., based directly on the material, or conceptually, i.e., based on the current state of research or on a theory/hypothesis or, rather, as an implementation of the guidelines used in an interview or focus group.
The QCA method is carried out in several phases, ranging from data preparation, category building and coding—which may run in several cycles—to analysis, report writing and presenting the results. QCA therefore means more than just coding the data. Coding is an important step in the analysis, but it is ultimately a preparation for the subsequent analytical steps.
The actual analysis phase consists of summarizing the data, and constantly comparing and contrasting the data. The analysis techniques can be qualitative as well as quantitative. The qualitative analysis may, for example, consist of comparing the statements of certain groups (for instance according to their characteristics, e.g., socio-demographic characteristics) on certain topics. Differences and similarities are identified and summarized in a report. Quantitative analyses may, on the other hand, consist of comparing the frequency of certain categories and/or subcategories for certain groups.
Summary tables and diagrams (e.g., concept maps) can play an important role in the analysis. A good example of a presentation in table form would be a case overview of selected research participants (or groups), in which their statements on certain topics, their judgements and variable values are displayed. An example of a concept map would be a diagram of the determined causal effects of different categories.
Visualizations can also have a diagnostic function in QCA—similarly to imaging procedures in medicine. For example, a ‘cases by categories’ or ‘categories by categories’ display can help identify patterns in the data and indicate which categories are particularly frequently or particularly rarely associated with certain other categories.
When analyzing texts, you should keep in mind that you are working in the field of interpretation. It can be assumed that texts or statements could be interpreted differently. Instead of adopting a constructivist ‘anything goes’ approach, the QCA method tries to reach a consensus—as far as this is possible—on the subjective meaning of statements and tries to define the categories formed or used by it so precisely that an intersubjective agreement can be achieved in the application of the categories.
Group processes play an important role in this process of achieving the necessary level of agreement. Divergent assignments to categories are discussed as a team and should result in an improvement of the category definitions. Categories for which no agreement can be reached in the coding of relevant points in the data must be excluded from the analysis. Content analysis stands and falls by its categories. An analysis with the help of categories that are interpreted and applied differently in the research team, does not make sense.
QCA does not claim to be the best method but recognizes that it has its limits (the interpretation barrier) and that its results have to face comparison with those of competing methods.
The systematic approach of QCA is multidisciplinary and can be applied in many disciplines, including mathematics education (Schwarz 2015 ). This method is particularly appropriate when working with clearly formulated research questions, because these questions play the central role in this method. Indeed, in every phase of the analysis there is a strong reference to the questions leading the research. One strength of QCA is that it can be used both to describe social phenomena and to develop theories or test hypotheses (Hopf 2016 , pp. 155–166).
Berelson, B. (1952). Content analysis in communication research . Glencoe: Free Press.
Google Scholar
Charmaz, K. (2006). Constructing grounded theory . Thousand Oaks: SAGE.
Früh, W. (2004). Inhaltsanalyse. Theorie und Praxis (5th ed.). Konstanz: UVK.
Guest, G., MacQueen, K. M., & Namey, E. E. (2012). Applied thematic analysis . Thousand Oaks: SAGE.
Book Google Scholar
Hopf, C. (2016). In W. Hopf & U. Kuckartz (Eds.), Schriften zu Methodologie und Methoden qualitativer Sozialforschung . Wiesbaden: Springer.
Krippendorff, K. H. (2012). Content analysis: An introduction to its methodology (3rd ed.). Thousand Oaks: SAGE.
Kuckartz, U. (2014). Qualitative text analysis: A guide to methods, practice and using software . Los Angeles: SAGE.
Kuckartz, U. (2016). Qualitative Inhaltsanalyse. Methoden, Praxis, Computerunterstützung (3rd ed.). Weinheim: Beltz Juventa.
Mayring, P. (2014). Qualitative content analysis: Theoretical foundation, basic procedures and software solution. Klagenfurt. http://nbn-resolving.de/urn:nbn:de:0168-ssoar-395173 .
Mayring, P. (2015). Qualitative content analysis: Theoretical background and procedures. In A. Bikner-Ahsbahs, C. Knipping, & N. Presmeg (Eds.), Approaches to qualitative research in mathematics education. Examples of methodology and methods (pp. 365–380). Dordrecht: Springer.
Saldana, J. (2015). The coding manual for qualitative researchers (3rd ed.). Thousand Oaks: SAGE.
Schreier, M. (2012). Qualitative content analysis in practice . Thousand Oaks: SAGE.
Schwarz, B. (2015). A study on professional competence of future teacher students as an example of a study using qualitative content analysis. In A. Bikner-Ahsbahs, C. Knipping, & N. Presmeg (Eds.), Approaches to qualitative research in mathematics education. Examples of methodology and methods (pp. 381–399). Dordrecht: Springer.
Strauss, A. L., & Corbin, J. M. (1990). Basics of qualitative research: Grounded theory procedures and techniques. Thousand Oaks: Sage Publications, Inc.
Download references
Author information
Authors and affiliations.
Philipps-University Marburg, Marburg, Germany
Udo Kuckartz
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Udo Kuckartz .
Editor information
Editors and affiliations.
Faculty of Education, Universität Hamburg, Hamburg, Germany
Gabriele Kaiser
Mathematics Department, Illinois State University, Normal, IL, USA
Norma Presmeg
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Reprints and permissions
Copyright information
© 2019 The Author(s)
About this chapter
Kuckartz, U. (2019). Qualitative Text Analysis: A Systematic Approach. In: Kaiser, G., Presmeg, N. (eds) Compendium for Early Career Researchers in Mathematics Education . ICME-13 Monographs. Springer, Cham. https://doi.org/10.1007/978-3-030-15636-7_8
Download citation
DOI : https://doi.org/10.1007/978-3-030-15636-7_8
Published : 27 April 2019
Publisher Name : Springer, Cham
Print ISBN : 978-3-030-15635-0
Online ISBN : 978-3-030-15636-7
eBook Packages : Education Education (R0)
Share this chapter
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Publish with us
Policies and ethics
- Find a journal
- Track your research

The Ultimate Guide to Qualitative Research - Part 2: Handling Qualitative Data

- Handling qualitative data
- Transcripts
- Field notes
- Survey data and responses
- Visual and audio data
- Data organization
- Data coding
- Coding frame
- Auto and smart coding
- Organizing codes
- Introduction
What is qualitative data analysis?
Qualitative data analysis methods, how do you analyze qualitative data, content analysis, thematic analysis.
- Thematic analysis vs. content analysis
- Narrative research
Phenomenological research
Discourse analysis, grounded theory.
- Deductive reasoning
- Inductive reasoning
- Inductive vs. deductive reasoning
- Qualitative data interpretation
- Qualitative data analysis software
Qualitative data analysis
Analyzing qualitative data is the next step after you have completed the use of qualitative data collection methods . The qualitative analysis process aims to identify themes and patterns that emerge across the data.

In simplified terms, qualitative research methods involve non-numerical data collection followed by an explanation based on the attributes of the data . For example, if you are asked to explain in qualitative terms a thermal image displayed in multiple colors, then you would explain the color differences rather than the heat's numerical value. If you have a large amount of data (e.g., of group discussions or observations of real-life situations), the next step is to transcribe and prepare the raw data for subsequent analysis.
Researchers can conduct studies fully based on qualitative methodology, or researchers can preface a quantitative research study with a qualitative study to identify issues that were not originally envisioned but are important to the study. Quantitative researchers may also collect and analyze qualitative data following their quantitative analyses to better understand the meanings behind their statistical results.
Conducting qualitative research can especially help build an understanding of how and why certain outcomes were achieved (in addition to what was achieved). For example, qualitative data analysis is often used for policy and program evaluation research since it can answer certain important questions more efficiently and effectively than quantitative approaches.

Qualitative data analysis can also answer important questions about the relevance, unintended effects, and impact of programs, such as:
- Were expectations reasonable?
- Did processes operate as expected?
- Were key players able to carry out their duties?
- Were there any unintended effects of the program?
The importance of qualitative data analysis
Qualitative approaches have the advantage of allowing for more diversity in responses and the capacity to adapt to new developments or issues during the research process itself. While qualitative data analysis can be demanding and time-consuming to conduct, many fields of research utilize qualitative software tools that have been specifically developed to provide more succinct, cost-efficient, and timely results.
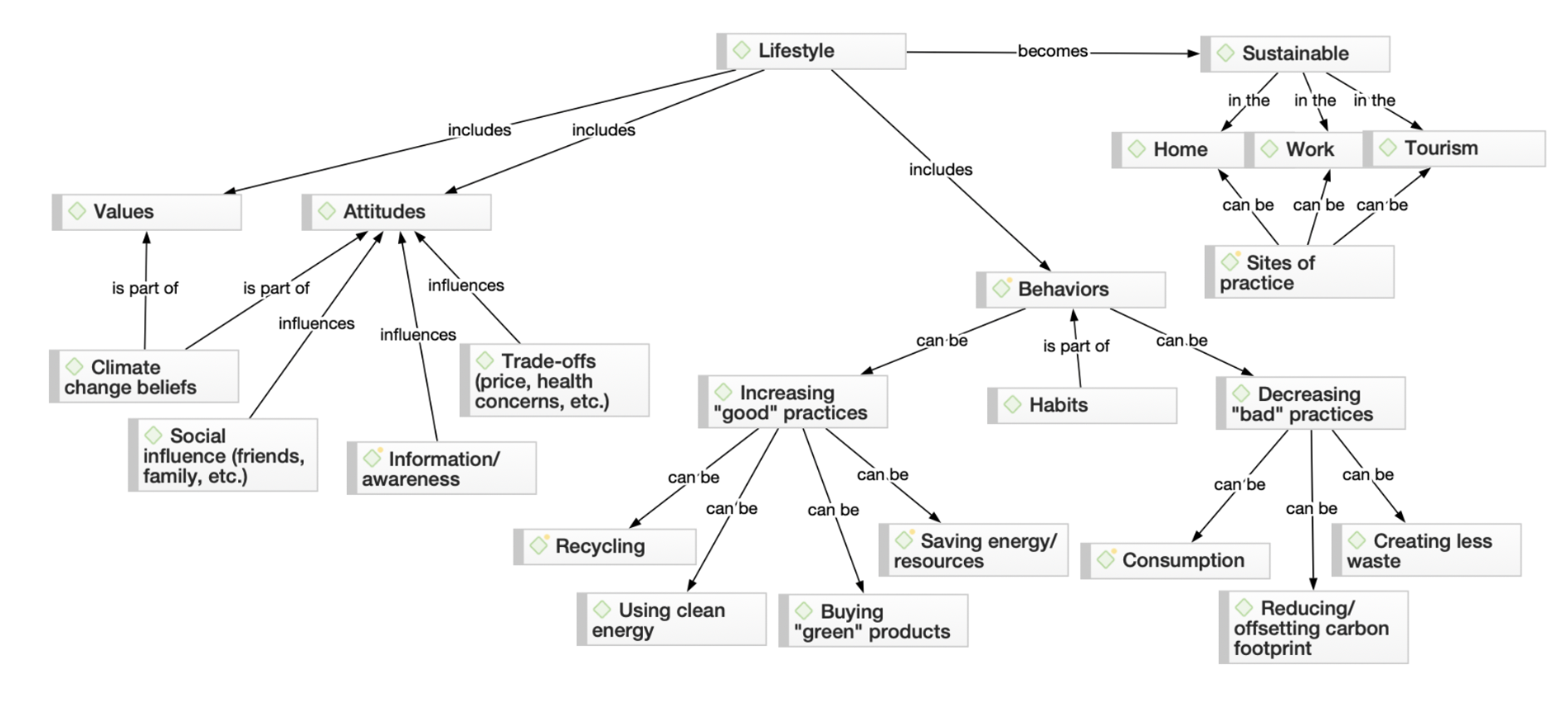
Qualitative data analysis is an important part of research and building greater understanding across fields for a number of reasons. First, cases for qualitative data analysis can be selected purposefully according to whether they typify certain characteristics or contextual locations. In other words, qualitative data permits deep immersion into a topic, phenomenon, or area of interest. Rather than seeking generalizability to the population the sample of participants represent, qualitative research aims to construct an in-depth and nuanced understanding of the research topic.
Secondly, the role or position of the researcher in qualitative data analysis is given greater critical attention. This is because, in qualitative data analysis, the possibility of the researcher taking a ‘neutral' or transcendent position is seen as more problematic in practical and/or philosophical terms. Hence, qualitative researchers are often exhorted to reflect on their role in the research process and make this clear in the analysis.

Thirdly, while qualitative data analysis can take a wide variety of forms, it largely differs from quantitative research in the focus on language, signs, experiences, and meaning. In addition, qualitative approaches to analysis are often holistic and contextual rather than analyzing the data in a piecemeal fashion or removing the data from its context. Qualitative approaches thus allow researchers to explore inquiries from directions that could not be accessed with only numerical quantitative data.
Establishing research rigor
Systematic and transparent approaches to the analysis of qualitative data are essential for rigor . For example, many qualitative research methods require researchers to carefully code data and discern and document themes in a consistent and credible way.
Perhaps the most traditional division in the way qualitative and quantitative research have been used in the social sciences is for qualitative methods to be used for exploratory purposes (e.g., to generate new theory or propositions) or to explain puzzling quantitative results, while quantitative methods are used to test hypotheses .

After you’ve collected relevant data , what is the best way to look at your data ? As always, it will depend on your research question . For instance, if you employed an observational research method to learn about a group’s shared practices, an ethnographic approach could be appropriate to explain the various dimensions of culture. If you collected textual data to understand how people talk about something, then a discourse analysis approach might help you generate key insights about language and communication.

The qualitative data coding process involves iterative categorization and recategorization, ensuring the evolution of the analysis to best represent the data. The procedure typically concludes with the interpretation of patterns and trends identified through the coding process.
To start off, let’s look at two broad approaches to data analysis.
Deductive analysis
Deductive analysis is guided by pre-existing theories or ideas. It starts with a theoretical framework , which is then used to code the data. The researcher can thus use this theoretical framework to interpret their data and answer their research question .
The key steps include coding the data based on the predetermined concepts or categories and using the theory to guide the interpretation of patterns among the codings. Deductive analysis is particularly useful when researchers aim to verify or extend an existing theory within a new context.
Inductive analysis
Inductive analysis involves the generation of new theories or ideas based on the data. The process starts without any preconceived theories or codes, and patterns, themes, and categories emerge out of the data.

The researcher codes the data to capture any concepts or patterns that seem interesting or important to the research question . These codes are then compared and linked, leading to the formation of broader categories or themes. The main goal of inductive analysis is to allow the data to 'speak for itself' rather than imposing pre-existing expectations or ideas onto the data.
Deductive and inductive approaches can be seen as sitting on opposite poles, and all research falls somewhere within that spectrum. Most often, qualitative data analysis approaches blend both deductive and inductive elements to contribute to the existing conversation around a topic while remaining open to potential unexpected findings. To help you make informed decisions about which qualitative data analysis approach fits with your research objectives, let's look at some of the common approaches for qualitative data analysis.
Content analysis is a research method used to identify patterns and themes within qualitative data. This approach involves systematically coding and categorizing specific aspects of the content in the data to uncover trends and patterns. An often important part of content analysis is quantifying frequencies and patterns of words or characteristics present in the data .
It is a highly flexible technique that can be adapted to various data types , including text, images, and audiovisual content . While content analysis can be exploratory in nature, it is also common to use pre-established theories and follow a more deductive approach to categorizing and quantifying the qualitative data.

Thematic analysis is a method used to identify, analyze, and report patterns or themes within the data. This approach moves beyond counting explicit words or phrases and focuses on also identifying implicit concepts and themes within the data.

Researchers conduct detailed coding of the data to ascertain repeated themes or patterns of meaning. Codes can be categorized into themes, and the researcher can analyze how the themes relate to one another. Thematic analysis is flexible in terms of the research framework, allowing for both inductive (data-driven) and deductive (theory-driven) approaches. The outcome is a rich, detailed, and complex account of the data.
Grounded theory is a systematic qualitative research methodology that is used to inductively generate theory that is 'grounded' in the data itself. Analysis takes place simultaneously with data collection , and researchers iterate between data collection and analysis until a comprehensive theory is developed.
Grounded theory is characterized by simultaneous data collection and analysis, the development of theoretical codes from the data, purposeful sampling of participants, and the constant comparison of data with emerging categories and concepts. The ultimate goal is to create a theoretical explanation that fits the data and answers the research question .
Discourse analysis is a qualitative research approach that emphasizes the role of language in social contexts. It involves examining communication and language use beyond the level of the sentence, considering larger units of language such as texts or conversations.

Discourse analysts typically investigate how social meanings and understandings are constructed in different contexts, emphasizing the connection between language and power. It can be applied to texts of all kinds, including interviews , documents, case studies , and social media posts.
Phenomenological research focuses on exploring how human beings make sense of an experience and delves into the essence of this experience. It strives to understand people's perceptions, perspectives, and understandings of a particular situation or phenomenon.

It involves in-depth engagement with participants, often through interviews or conversations, to explore their lived experiences. The goal is to derive detailed descriptions of the essence of the experience and to interpret what insights or implications this may bear on our understanding of this phenomenon.

Whatever your data analysis approach, start with ATLAS.ti
Qualitative data analysis done quickly and intuitively with ATLAS.ti. Download a free trial today.
Now that we've summarized the major approaches to data analysis, let's look at the broader process of research and data analysis. Suppose you need to do some research to find answers to any kind of research question, be it an academic inquiry, business problem, or policy decision. In that case, you need to collect some data. There are many methods of collecting data: you can collect primary data yourself by conducting interviews, focus groups , or a survey , for instance. Another option is to use secondary data sources. These are data previously collected for other projects, historical records, reports, statistics – basically everything that exists already and can be relevant to your research.

The data you collect should always be a good fit for your research question . For example, if you are interested in how many people in your target population like your brand compared to others, it is no use to conduct interviews or a few focus groups . The sample will be too small to get a representative picture of the population. If your questions are about "how many….", "what is the spread…" etc., you need to conduct quantitative research . If you are interested in why people like different brands, their motives, and their experiences, then conducting qualitative research can provide you with the answers you are looking for.
Let's describe the important steps involved in conducting research.
Step 1: Planning the research
As the saying goes: "Garbage in, garbage out." Suppose you find out after you have collected data that
- you talked to the wrong people
- asked the wrong questions
- a couple of focus groups sessions would have yielded better results because of the group interaction, or
- a survey including a few open-ended questions sent to a larger group of people would have been sufficient and required less effort.
Think thoroughly about sampling, the questions you will be asking, and in which form. If you conduct a focus group or an interview, you are the research instrument, and your data collection will only be as good as you are. If you have never done it before, seek some training and practice. If you have other people do it, make sure they have the skills.
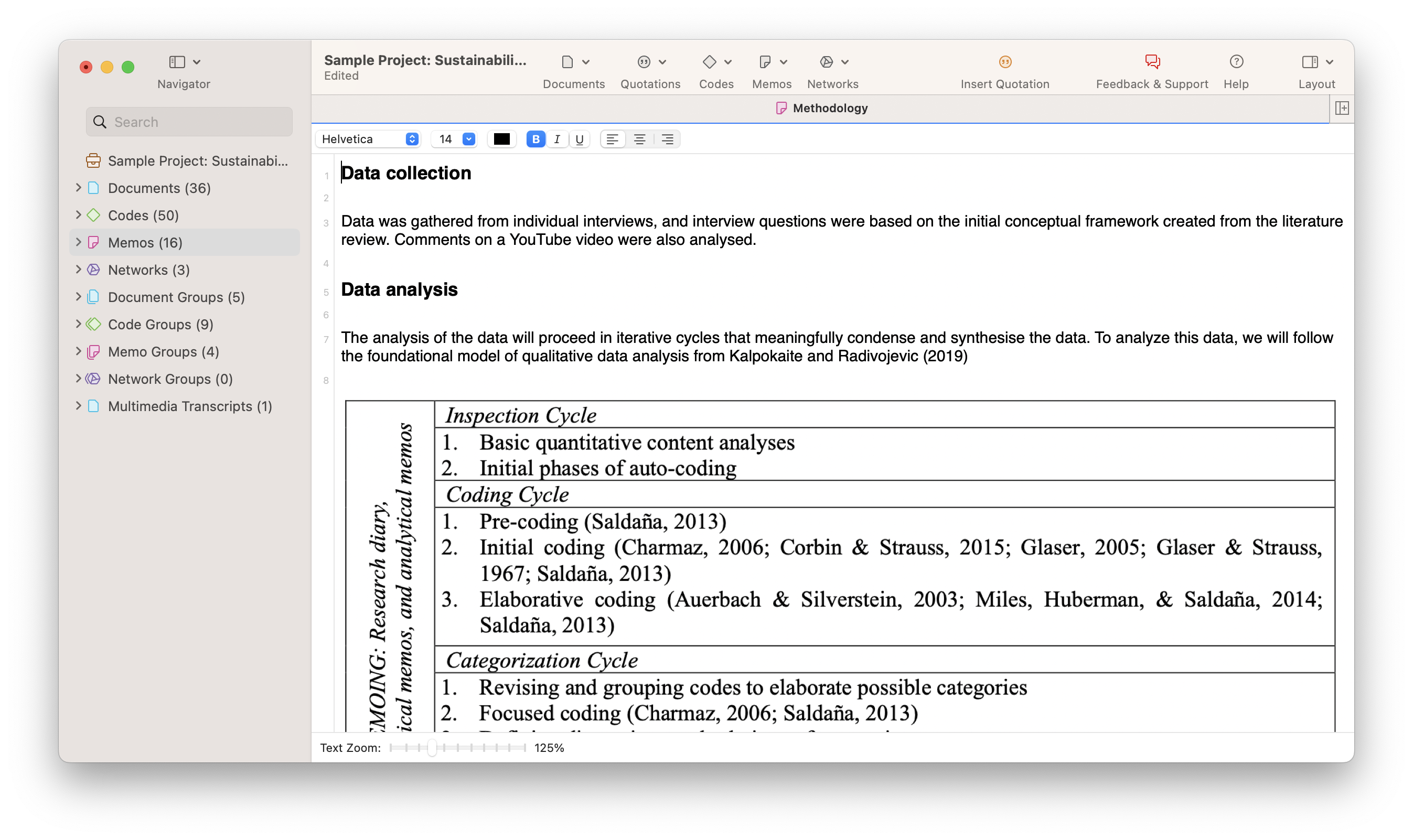
Step 2: Preparing the data
When you conduct focus groups or interviews, think about how to transcribe them. Do you want to run them online or offline? If online, check out which tools can serve your needs, both in terms of functionality and cost. For any audio or video recordings , you can consider using automatic transcription software or services. Automatically generated transcripts can save you time and money, but they still need to be checked. If you don't do this yourself, make sure that you instruct the person doing it on how to prepare the data.
- How should the final transcript be formatted for later analysis?
- Which names and locations should be anonymized?
- What kind of speaker IDs to use?
What about survey data ? Some survey data programs will immediately provide basic descriptive-level analysis of the responses. ATLAS.ti will support you with the analysis of the open-ended questions. For this, you need to export your data as an Excel file. ATLAS.ti's survey import wizard will guide you through the process.
Other kinds of data such as images, videos, audio recordings, text, and more can be imported to ATLAS.ti. You can organize all your data into groups and write comments on each source of data to maintain a systematic organization and documentation of your data.

Step 3: Exploratory data analysis
You can run a few simple exploratory analyses to get to know your data. For instance, you can create a word list or word cloud of all your text data or compare and contrast the words in different documents. You can also let ATLAS.ti find relevant concepts for you. There are many tools available that can automatically code your text data, so you can also use these codings to explore your data and refine your coding.

For instance, you can get a feeling for the sentiments expressed in the data. Who is more optimistic, pessimistic, or neutral in their responses? ATLAS.ti can auto-code the positive, negative, and neutral sentiments in your data. Naturally, you can also simply browse through your data and highlight relevant segments that catch your attention or attach codes to begin condensing the data.

Step 4: Build a code system
Whether you start with auto-coding or manual coding, after having generated some first codes, you need to get some order in your code system to develop a cohesive understanding. You can build your code system by sorting codes into groups and creating categories and subcodes. As this process requires reading and re-reading your data, you will become very familiar with your data. Counting on a tool like ATLAS.ti qualitative data analysis software will support you in the process and make it easier to review your data, modify codings if necessary, change code labels, and write operational definitions to explain what each code means.

Step 5: Query your coded data and write up the analysis
Once you have coded your data, it is time to take the analysis a step further. When using software for qualitative data analysis , it is easy to compare and contrast subsets in your data, such as groups of participants or sets of themes.

For instance, you can query the various opinions of female vs. male respondents. Is there a difference between consumers from rural or urban areas or among different age groups or educational levels? Which codes occur together throughout the data set? Are there relationships between various concepts, and if so, why?
Step 6: Data visualization
Data visualization brings your data to life. It is a powerful way of seeing patterns and relationships in your data. For instance, diagrams allow you to see how your codes are distributed across documents or specific subpopulations in your data.

Exploring coded data on a canvas, moving around code labels in a virtual space, linking codes and other elements of your data set, and thinking about how they are related and why – all of these will advance your analysis and spur further insights. Visuals are also great for communicating results to others.
Step 7: Data presentation
The final step is to summarize the analysis in a written report . You can now put together the memos you have written about the various topics, select some salient quotes that illustrate your writing, and add visuals such as tables and diagrams. If you follow the steps above, you will already have all the building blocks, and you just have to put them together in a report or presentation.
When preparing a report or a presentation, keep your audience in mind. Does your audience better understand numbers than long sections of detailed interpretations? If so, add more tables, charts, and short supportive data quotes to your report or presentation. If your audience loves a good interpretation, add your full-length memos and walk your audience through your conceptual networks and illustrative data quotes.

Qualitative data analysis begins with ATLAS.ti
For tools that can make the most out of your data, check out ATLAS.ti with a free trial.
HOTSAUCE is back 🔥 We’re bringing the heat to CX Circle NYC on Sep 10 🗽 Get your ticket for the #1 DX event
Learn / Guides / Qualitative data analysis guide
Back to guides
5 qualitative data analysis methods
Qualitative data uncovers valuable insights that help you improve the user and customer experience. But how exactly do you measure and analyze data that isn't quantifiable?
There are different qualitative data analysis methods to help you make sense of qualitative feedback and customer insights, depending on your business goals and the type of data you've collected.
Before you choose a qualitative data analysis method for your team, you need to consider the available techniques and explore their use cases to understand how each process might help you better understand your users.
This guide covers five qualitative analysis methods to choose from, and will help you pick the right one(s) based on your goals.
Content analysis
Thematic analysis
Narrative analysis
Grounded theory analysis
Discourse analysis
5 qualitative data analysis methods explained
Qualitative data analysis ( QDA ) is the process of organizing, analyzing, and interpreting qualitative research data—non-numeric, conceptual information, and user feedback—to capture themes and patterns, answer research questions, and identify actions to improve your product or website.
Step 1 in the research process (after planning ) is qualitative data collection. You can use behavior analytics software—like Hotjar —to capture qualitative data with context, and learn the real motivation behind user behavior, by collecting written customer feedback with Surveys or scheduling an in-depth user interview with Engage .
Use Hotjar’s tools to collect feedback, uncover behavior trends, and understand the ‘why’ behind user actions.
1. Content analysis
Content analysis is a qualitative research method that examines and quantifies the presence of certain words, subjects, and concepts in text, image, video, or audio messages. The method transforms qualitative input into quantitative data to help you make reliable conclusions about what customers think of your brand, and how you can improve their experience and opinion.
Conduct content analysis manually (which can be time-consuming) or use analysis tools like Lexalytics to reveal communication patterns, uncover differences in individual or group communication trends, and make broader connections between concepts.

How content analysis can help your team
Content analysis is often used by marketers and customer service specialists, helping them understand customer behavior and measure brand reputation.
For example, you may run a customer survey with open-ended questions to discover users’ concerns—in their own words—about their experience with your product. Instead of having to process hundreds of answers manually, a content analysis tool helps you analyze and group results based on the emotion expressed in texts.
Some other examples of content analysis include:
Analyzing brand mentions on social media to understand your brand's reputation
Reviewing customer feedback to evaluate (and then improve) the customer and user experience (UX)
Researching competitors’ website pages to identify their competitive advantages and value propositions
Interpreting customer interviews and survey results to determine user preferences, and setting the direction for new product or feature developments
Content analysis was a major part of our growth during my time at Hypercontext.
[It gave us] a better understanding of the [blog] topics that performed best for signing new users up. We were also able to go deeper within those blog posts to better understand the formats [that worked].
2. Thematic analysis
Thematic analysis helps you identify, categorize, analyze, and interpret patterns in qualitative study data , and can be done with tools like Dovetail and Thematic .
While content analysis and thematic analysis seem similar, they're different in concept:
Content analysis can be applied to both qualitative and quantitative data , and focuses on identifying frequencies and recurring words and subjects
Thematic analysis can only be applied to qualitative data, and focuses on identifying patterns and themes
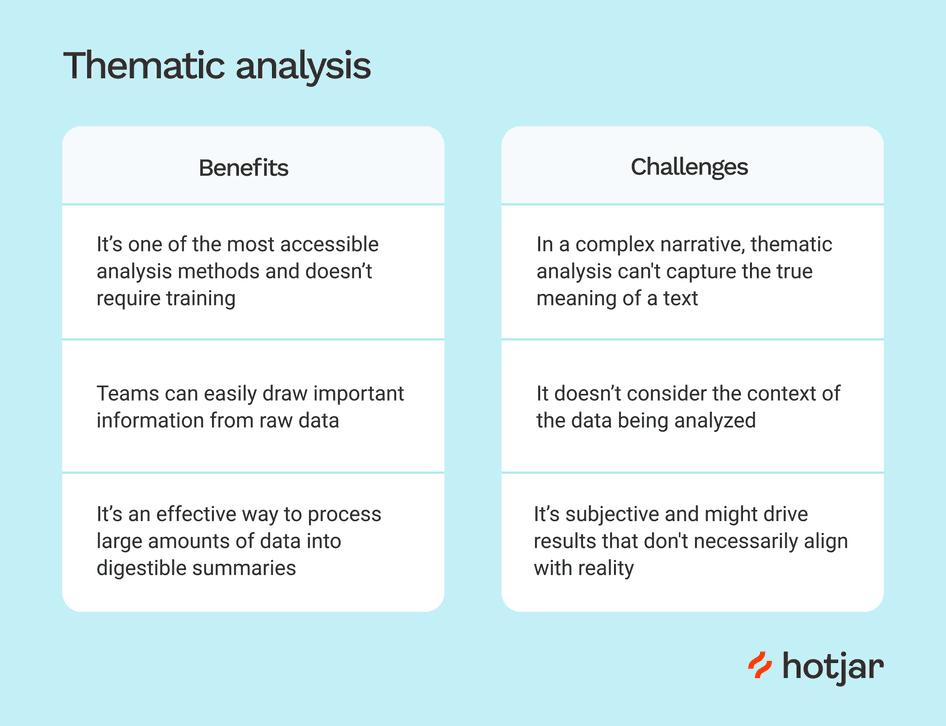
How thematic analysis can help your team
Thematic analysis can be used by pretty much anyone: from product marketers, to customer relationship managers, to UX researchers.
For example, product teams use thematic analysis to better understand user behaviors and needs and improve UX . Analyzing customer feedback lets you identify themes (e.g. poor navigation or a buggy mobile interface) highlighted by users and get actionable insight into what they really expect from the product.
💡 Pro tip: looking for a way to expedite the data analysis process for large amounts of data you collected with a survey? Try Hotjar’s AI for Surveys : along with generating a survey based on your goal in seconds, our AI will analyze the raw data and prepare an automated summary report that presents key thematic findings, respondent quotes, and actionable steps to take, making the analysis of qualitative data a breeze.
3. Narrative analysis
Narrative analysis is a method used to interpret research participants’ stories —things like testimonials , case studies, focus groups, interviews, and other text or visual data—with tools like Delve and AI-powered ATLAS.ti .
Some formats don’t work well with narrative analysis, including heavily structured interviews and written surveys, which don’t give participants as much opportunity to tell their stories in their own words.
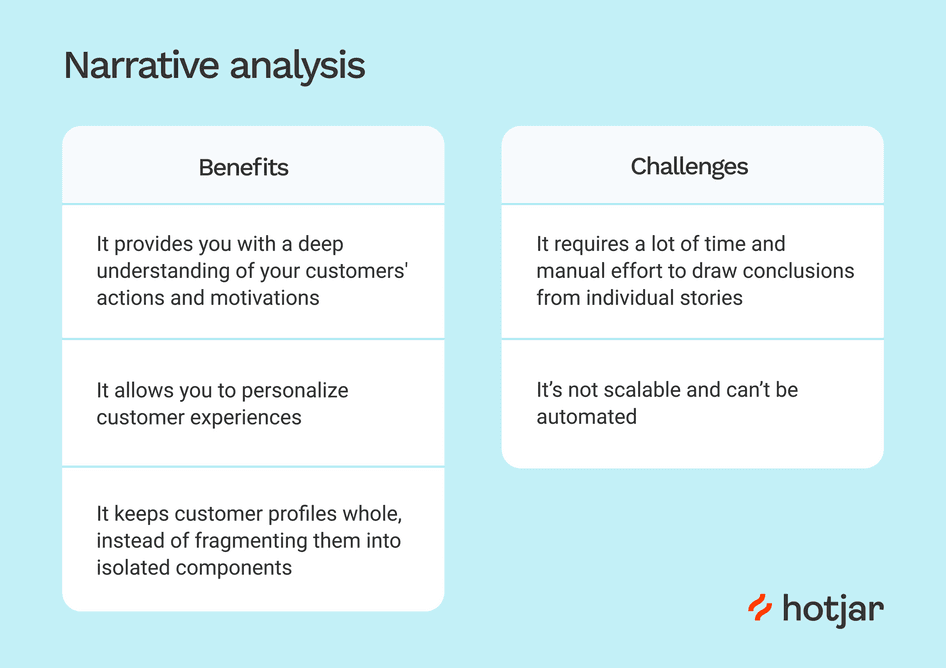
How narrative analysis can help your team
Narrative analysis provides product teams with valuable insight into the complexity of customers’ lives, feelings, and behaviors.
In a marketing research context, narrative analysis involves capturing and reviewing customer stories—on social media, for example—to get in-depth insight into their lives, priorities, and challenges.
This might look like analyzing daily content shared by your audiences’ favorite influencers on Instagram, or analyzing customer reviews on sites like G2 or Capterra to gain a deep understanding of individual customer experiences. The results of this analysis also contribute to developing corresponding customer personas .
💡 Pro tip: conducting user interviews is an excellent way to collect data for narrative analysis. Though interviews can be time-intensive, there are tools out there that streamline the workload.
Hotjar Engage automates the entire process, from recruiting to scheduling to generating the all-important interview transcripts you’ll need for the analysis phase of your research project.
4. Grounded theory analysis
Grounded theory analysis is a method of conducting qualitative research to develop theories by examining real-world data. This technique involves the creation of hypotheses and theories through qualitative data collection and evaluation, and can be performed with qualitative data analysis software tools like MAXQDA and NVivo .
Unlike other qualitative data analysis techniques, this method is inductive rather than deductive: it develops theories from data, not the other way around.

How grounded theory analysis can help your team
Grounded theory analysis is used by software engineers, product marketers, managers, and other specialists who deal with data sets to make informed business decisions.
For example, product marketing teams may turn to customer surveys to understand the reasons behind high churn rates , then use grounded theory to analyze responses and develop hypotheses about why users churn, and how you can get them to stay.
Grounded theory can also be helpful in the talent management process. For example, HR representatives may use it to develop theories about low employee engagement, and come up with solutions based on their research findings.
5. Discourse analysis
Discourse analysis is the act of researching the underlying meaning of qualitative data. It involves the observation of texts, audio, and videos to study the relationships between information and its social context.
In contrast to content analysis, this method focuses on the contextual meaning of language: discourse analysis sheds light on what audiences think of a topic, and why they feel the way they do about it.

How discourse analysis can help your team
In a business context, this method is primarily used by marketing teams. Discourse analysis helps marketers understand the norms and ideas in their market , and reveals why they play such a significant role for their customers.
Once the origins of trends are uncovered, it’s easier to develop a company mission, create a unique tone of voice, and craft effective marketing messages.
Which qualitative data analysis method should you choose?
While the five qualitative data analysis methods we list above are all aimed at processing data and answering research questions, these techniques differ in their intent and the approaches applied.
Choosing the right analysis method for your team isn't a matter of preference—selecting a method that fits is only possible once you define your research goals and have a clear intention. When you know what you need (and why you need it), you can identify an analysis method that aligns with your research objectives.
Gather qualitative data with Hotjar
Use Hotjar’s product experience insights in your qualitative research. Collect feedback, uncover behavior trends, and understand the ‘why’ behind user actions.
FAQs about qualitative data analysis methods
What is the qualitative data analysis approach.
The qualitative data analysis approach refers to the process of systematizing descriptive data collected through interviews, focus groups, surveys, and observations and then interpreting it. The methodology aims to identify patterns and themes behind textual data, and other unquantifiable data, as opposed to numerical data.
What are qualitative data analysis methods?
Five popular qualitative data analysis methods are:
What is the process of qualitative data analysis?
The process of qualitative data analysis includes six steps:
Define your research question
Prepare the data
Choose the method of qualitative analysis
Code the data
Identify themes, patterns, and relationships
Make hypotheses and act
Qualitative data analysis guide
Previous chapter
QDA challenges
Next chapter

Qualitative Data Analysis
Qualitative data refers to non-numeric information such as interview transcripts, notes, video and audio recordings, images and text documents. Qualitative data analysis can be divided into the following five categories:
1. Content analysis . This refers to the process of categorizing verbal or behavioural data to classify, summarize and tabulate the data.
2. Narrative analysis . This method involves the reformulation of stories presented by respondents taking into account context of each case and different experiences of each respondent. In other words, narrative analysis is the revision of primary qualitative data by researcher.
3. Discourse analysis . A method of analysis of naturally occurring talk and all types of written text.
4. Framework analysis . This is more advanced method that consists of several stages such as familiarization, identifying a thematic framework, coding, charting, mapping and interpretation.
5. Grounded theory . This method of qualitative data analysis starts with an analysis of a single case to formulate a theory. Then, additional cases are examined to see if they contribute to the theory.
Qualitative data analysis can be conducted through the following three steps:
Step 1: Developing and Applying Codes . Coding can be explained as categorization of data. A ‘code’ can be a word or a short phrase that represents a theme or an idea. All codes need to be assigned meaningful titles. A wide range of non-quantifiable elements such as events, behaviours, activities, meanings etc. can be coded.
There are three types of coding:
- Open coding . The initial organization of raw data to try to make sense of it.
- Axial coding . Interconnecting and linking the categories of codes.
- Selective coding . Formulating the story through connecting the categories.
Coding can be done manually or using qualitative data analysis software such as
NVivo, Atlas ti 6.0, HyperRESEARCH 2.8, Max QDA and others.
When using manual coding you can use folders, filing cabinets, wallets etc. to gather together materials that are examples of similar themes or analytic ideas. Manual method of coding in qualitative data analysis is rightly considered as labour-intensive, time-consuming and outdated.
In computer-based coding, on the other hand, physical files and cabinets are replaced with computer based directories and files. When choosing software for qualitative data analysis you need to consider a wide range of factors such as the type and amount of data you need to analyse, time required to master the software and cost considerations.
Moreover, it is important to get confirmation from your dissertation supervisor prior to application of any specific qualitative data analysis software.
The following table contains examples of research titles, elements to be coded and identification of relevant codes:
| Born or bred: revising The Great Man theory of leadership in the 21 century | Leadership practice | Born leaders Made leaders Leadership effectiveness |
| A study into advantages and disadvantages of various entry strategies to Chinese market
|
Market entry strategies | Wholly-owned subsidiaries Joint-ventures Franchising Exporting Licensing |
| Impacts of CSR programs and initiative on brand image: a case study of Coca-Cola Company UK. |
Activities, phenomenon | Philanthropy Supporting charitable courses Ethical behaviour Brand awareness Brand value |
| An investigation into the ways of customer relationship management in mobile marketing environment |
Tactics | Viral messages Customer retention Popularity of social networking sites |
Qualitative data coding
Step 2: Identifying themes, patterns and relationships . Unlike quantitative methods , in qualitative data analysis there are no universally applicable techniques that can be applied to generate findings. Analytical and critical thinking skills of researcher plays significant role in data analysis in qualitative studies. Therefore, no qualitative study can be repeated to generate the same results.
Nevertheless, there is a set of techniques that you can use to identify common themes, patterns and relationships within responses of sample group members in relation to codes that have been specified in the previous stage.
Specifically, the most popular and effective methods of qualitative data interpretation include the following:
- Word and phrase repetitions – scanning primary data for words and phrases most commonly used by respondents, as well as, words and phrases used with unusual emotions;
- Primary and secondary data comparisons – comparing the findings of interview/focus group/observation/any other qualitative data collection method with the findings of literature review and discussing differences between them;
- Search for missing information – discussions about which aspects of the issue was not mentioned by respondents, although you expected them to be mentioned;
- Metaphors and analogues – comparing primary research findings to phenomena from a different area and discussing similarities and differences.
Step 3: Summarizing the data . At this last stage you need to link research findings to hypotheses or research aim and objectives. When writing data analysis chapter, you can use noteworthy quotations from the transcript in order to highlight major themes within findings and possible contradictions.
It is important to note that the process of qualitative data analysis described above is general and different types of qualitative studies may require slightly different methods of data analysis.
My e-book, The Ultimate Guide to Writing a Dissertation in Business Studies: a step by step approach contains a detailed, yet simple explanation of qualitative data analysis methods . The e-book explains all stages of the research process starting from the selection of the research area to writing personal reflection. Important elements of dissertations such as research philosophy, research approach, research design, methods of data collection and data analysis are explained in simple words. John Dudovskiy

Qualitative Research: Characteristics, Design, Methods & Examples
Lauren McCall
MSc Health Psychology Graduate
MSc, Health Psychology, University of Nottingham
Lauren obtained an MSc in Health Psychology from The University of Nottingham with a distinction classification.
Learn about our Editorial Process
Saul McLeod, PhD
Editor-in-Chief for Simply Psychology
BSc (Hons) Psychology, MRes, PhD, University of Manchester
Saul McLeod, PhD., is a qualified psychology teacher with over 18 years of experience in further and higher education. He has been published in peer-reviewed journals, including the Journal of Clinical Psychology.
Olivia Guy-Evans, MSc
Associate Editor for Simply Psychology
BSc (Hons) Psychology, MSc Psychology of Education
Olivia Guy-Evans is a writer and associate editor for Simply Psychology. She has previously worked in healthcare and educational sectors.
On This Page:
Qualitative research is a type of research methodology that focuses on gathering and analyzing non-numerical data to gain a deeper understanding of human behavior, experiences, and perspectives.
It aims to explore the “why” and “how” of a phenomenon rather than the “what,” “where,” and “when” typically addressed by quantitative research.
Unlike quantitative research, which focuses on gathering and analyzing numerical data for statistical analysis, qualitative research involves researchers interpreting data to identify themes, patterns, and meanings.
Qualitative research can be used to:
- Gain deep contextual understandings of the subjective social reality of individuals
- To answer questions about experience and meaning from the participant’s perspective
- To design hypotheses, theory must be researched using qualitative methods to determine what is important before research can begin.
Examples of qualitative research questions include:
- How does stress influence young adults’ behavior?
- What factors influence students’ school attendance rates in developed countries?
- How do adults interpret binge drinking in the UK?
- What are the psychological impacts of cervical cancer screening in women?
- How can mental health lessons be integrated into the school curriculum?
Characteristics
Naturalistic setting.
Individuals are studied in their natural setting to gain a deeper understanding of how people experience the world. This enables the researcher to understand a phenomenon close to how participants experience it.
Naturalistic settings provide valuable contextual information to help researchers better understand and interpret the data they collect.
The environment, social interactions, and cultural factors can all influence behavior and experiences, and these elements are more easily observed in real-world settings.
Reality is socially constructed
Qualitative research aims to understand how participants make meaning of their experiences – individually or in social contexts. It assumes there is no objective reality and that the social world is interpreted (Yilmaz, 2013).
The primacy of subject matter
The primary aim of qualitative research is to understand the perspectives, experiences, and beliefs of individuals who have experienced the phenomenon selected for research rather than the average experiences of groups of people (Minichiello, 1990).
An in-depth understanding is attained since qualitative techniques allow participants to freely disclose their experiences, thoughts, and feelings without constraint (Tenny et al., 2022).

Variables are complex, interwoven, and difficult to measure
Factors such as experiences, behaviors, and attitudes are complex and interwoven, so they cannot be reduced to isolated variables , making them difficult to measure quantitatively.
However, a qualitative approach enables participants to describe what, why, or how they were thinking/ feeling during a phenomenon being studied (Yilmaz, 2013).
Emic (insider’s point of view)
The phenomenon being studied is centered on the participants’ point of view (Minichiello, 1990).
Emic is used to describe how participants interact, communicate, and behave in the research setting (Scarduzio, 2017).
Interpretive analysis
In qualitative research, interpretive analysis is crucial in making sense of the collected data.
This process involves examining the raw data, such as interview transcripts, field notes, or documents, and identifying the underlying themes, patterns, and meanings that emerge from the participants’ experiences and perspectives.
Collecting Qualitative Data
There are four main research design methods used to collect qualitative data: observations, interviews, focus groups, and ethnography.
Observations
This method involves watching and recording phenomena as they occur in nature. Observation can be divided into two types: participant and non-participant observation.
In participant observation, the researcher actively participates in the situation/events being observed.
In non-participant observation, the researcher is not an active part of the observation and tries not to influence the behaviors they are observing (Busetto et al., 2020).
Observations can be covert (participants are unaware that a researcher is observing them) or overt (participants are aware of the researcher’s presence and know they are being observed).
However, awareness of an observer’s presence may influence participants’ behavior.
Interviews give researchers a window into the world of a participant by seeking their account of an event, situation, or phenomenon. They are usually conducted on a one-to-one basis and can be distinguished according to the level at which they are structured (Punch, 2013).
Structured interviews involve predetermined questions and sequences to ensure replicability and comparability. However, they are unable to explore emerging issues.
Informal interviews consist of spontaneous, casual conversations which are closer to the truth of a phenomenon. However, information is gathered using quick notes made by the researcher and is therefore subject to recall bias.
Semi-structured interviews have a flexible structure, phrasing, and placement so emerging issues can be explored (Denny & Weckesser, 2022).
The use of probing questions and clarification can lead to a detailed understanding, but semi-structured interviews can be time-consuming and subject to interviewer bias.
Focus groups
Similar to interviews, focus groups elicit a rich and detailed account of an experience. However, focus groups are more dynamic since participants with shared characteristics construct this account together (Denny & Weckesser, 2022).
A shared narrative is built between participants to capture a group experience shaped by a shared context.
The researcher takes on the role of a moderator, who will establish ground rules and guide the discussion by following a topic guide to focus the group discussions.
Typically, focus groups have 4-10 participants as a discussion can be difficult to facilitate with more than this, and this number allows everyone the time to speak.
Ethnography
Ethnography is a methodology used to study a group of people’s behaviors and social interactions in their environment (Reeves et al., 2008).
Data are collected using methods such as observations, field notes, or structured/ unstructured interviews.
The aim of ethnography is to provide detailed, holistic insights into people’s behavior and perspectives within their natural setting. In order to achieve this, researchers immerse themselves in a community or organization.
Due to the flexibility and real-world focus of ethnography, researchers are able to gather an in-depth, nuanced understanding of people’s experiences, knowledge and perspectives that are influenced by culture and society.
In order to develop a representative picture of a particular culture/ context, researchers must conduct extensive field work.
This can be time-consuming as researchers may need to immerse themselves into a community/ culture for a few days, or possibly a few years.
Qualitative Data Analysis Methods
Different methods can be used for analyzing qualitative data. The researcher chooses based on the objectives of their study.
The researcher plays a key role in the interpretation of data, making decisions about the coding, theming, decontextualizing, and recontextualizing of data (Starks & Trinidad, 2007).
Grounded theory
Grounded theory is a qualitative method specifically designed to inductively generate theory from data. It was developed by Glaser and Strauss in 1967 (Glaser & Strauss, 2017).
This methodology aims to develop theories (rather than test hypotheses) that explain a social process, action, or interaction (Petty et al., 2012). To inform the developing theory, data collection and analysis run simultaneously.
There are three key types of coding used in grounded theory: initial (open), intermediate (axial), and advanced (selective) coding.
Throughout the analysis, memos should be created to document methodological and theoretical ideas about the data. Data should be collected and analyzed until data saturation is reached and a theory is developed.
Content analysis
Content analysis was first used in the early twentieth century to analyze textual materials such as newspapers and political speeches.
Content analysis is a research method used to identify and analyze the presence and patterns of themes, concepts, or words in data (Vaismoradi et al., 2013).
This research method can be used to analyze data in different formats, which can be written, oral, or visual.
The goal of content analysis is to develop themes that capture the underlying meanings of data (Schreier, 2012).
Qualitative content analysis can be used to validate existing theories, support the development of new models and theories, and provide in-depth descriptions of particular settings or experiences.
The following six steps provide a guideline for how to conduct qualitative content analysis.
- Define a Research Question : To start content analysis, a clear research question should be developed.
- Identify and Collect Data : Establish the inclusion criteria for your data. Find the relevant sources to analyze.
- Define the Unit or Theme of Analysis : Categorize the content into themes. Themes can be a word, phrase, or sentence.
- Develop Rules for Coding your Data : Define a set of coding rules to ensure that all data are coded consistently.
- Code the Data : Follow the coding rules to categorize data into themes.
- Analyze the Results and Draw Conclusions : Examine the data to identify patterns and draw conclusions in relation to your research question.
Discourse analysis
Discourse analysis is a research method used to study written/ spoken language in relation to its social context (Wood & Kroger, 2000).
In discourse analysis, the researcher interprets details of language materials and the context in which it is situated.
Discourse analysis aims to understand the functions of language (how language is used in real life) and how meaning is conveyed by language in different contexts. Researchers use discourse analysis to investigate social groups and how language is used to achieve specific communication goals.
Different methods of discourse analysis can be used depending on the aims and objectives of a study. However, the following steps provide a guideline on how to conduct discourse analysis.
- Define the Research Question : Develop a relevant research question to frame the analysis.
- Gather Data and Establish the Context : Collect research materials (e.g., interview transcripts, documents). Gather factual details and review the literature to construct a theory about the social and historical context of your study.
- Analyze the Content : Closely examine various components of the text, such as the vocabulary, sentences, paragraphs, and structure of the text. Identify patterns relevant to the research question to create codes, then group these into themes.
- Review the Results : Reflect on the findings to examine the function of the language, and the meaning and context of the discourse.
Thematic analysis
Thematic analysis is a method used to identify, interpret, and report patterns in data, such as commonalities or contrasts.
Although the origin of thematic analysis can be traced back to the early twentieth century, understanding and clarity of thematic analysis is attributed to Braun and Clarke (2006).
Thematic analysis aims to develop themes (patterns of meaning) across a dataset to address a research question.
In thematic analysis, qualitative data is gathered using techniques such as interviews, focus groups, and questionnaires. Audio recordings are transcribed. The dataset is then explored and interpreted by a researcher to identify patterns.
This occurs through the rigorous process of data familiarisation, coding, theme development, and revision. These identified patterns provide a summary of the dataset and can be used to address a research question.
Themes are developed by exploring the implicit and explicit meanings within the data. Two different approaches are used to generate themes: inductive and deductive.
An inductive approach allows themes to emerge from the data. In contrast, a deductive approach uses existing theories or knowledge to apply preconceived ideas to the data.
Phases of Thematic Analysis
Braun and Clarke (2006) provide a guide of the six phases of thematic analysis. These phases can be applied flexibly to fit research questions and data.
| Phase | |
|---|---|
| 1. Gather and transcribe data | Gather raw data, for example interviews or focus groups, and transcribe audio recordings fully |
| 2. Familiarization with data | Read and reread all your data from beginning to end; note down initial ideas |
| 3. Create initial codes | Start identifying preliminary codes which highlight important features of the data and may be relevant to the research question |
| 4. Create new codes which encapsulate potential themes | Review initial codes and explore any similarities, differences, or contradictions to uncover underlying themes; create a map to visualize identified themes |
| 5. Take a break then return to the data | Take a break and then return later to review themes |
| 6. Evaluate themes for good fit | Last opportunity for analysis; check themes are supported and saturated with data |
Template analysis
Template analysis refers to a specific method of thematic analysis which uses hierarchical coding (Brooks et al., 2014).
Template analysis is used to analyze textual data, for example, interview transcripts or open-ended responses on a written questionnaire.
To conduct template analysis, a coding template must be developed (usually from a subset of the data) and subsequently revised and refined. This template represents the themes identified by researchers as important in the dataset.
Codes are ordered hierarchically within the template, with the highest-level codes demonstrating overarching themes in the data and lower-level codes representing constituent themes with a narrower focus.
A guideline for the main procedural steps for conducting template analysis is outlined below.
- Familiarization with the Data : Read (and reread) the dataset in full. Engage, reflect, and take notes on data that may be relevant to the research question.
- Preliminary Coding : Identify initial codes using guidance from the a priori codes, identified before the analysis as likely to be beneficial and relevant to the analysis.
- Organize Themes : Organize themes into meaningful clusters. Consider the relationships between the themes both within and between clusters.
- Produce an Initial Template : Develop an initial template. This may be based on a subset of the data.
- Apply and Develop the Template : Apply the initial template to further data and make any necessary modifications. Refinements of the template may include adding themes, removing themes, or changing the scope/title of themes.
- Finalize Template : Finalize the template, then apply it to the entire dataset.
Frame analysis
Frame analysis is a comparative form of thematic analysis which systematically analyzes data using a matrix output.
Ritchie and Spencer (1994) developed this set of techniques to analyze qualitative data in applied policy research. Frame analysis aims to generate theory from data.
Frame analysis encourages researchers to organize and manage their data using summarization.
This results in a flexible and unique matrix output, in which individual participants (or cases) are represented by rows and themes are represented by columns.
Each intersecting cell is used to summarize findings relating to the corresponding participant and theme.
Frame analysis has five distinct phases which are interrelated, forming a methodical and rigorous framework.
- Familiarization with the Data : Familiarize yourself with all the transcripts. Immerse yourself in the details of each transcript and start to note recurring themes.
- Develop a Theoretical Framework : Identify recurrent/ important themes and add them to a chart. Provide a framework/ structure for the analysis.
- Indexing : Apply the framework systematically to the entire study data.
- Summarize Data in Analytical Framework : Reduce the data into brief summaries of participants’ accounts.
- Mapping and Interpretation : Compare themes and subthemes and check against the original transcripts. Group the data into categories and provide an explanation for them.
Preventing Bias in Qualitative Research
To evaluate qualitative studies, the CASP (Critical Appraisal Skills Programme) checklist for qualitative studies can be used to ensure all aspects of a study have been considered (CASP, 2018).
The quality of research can be enhanced and assessed using criteria such as checklists, reflexivity, co-coding, and member-checking.
Co-coding
Relying on only one researcher to interpret rich and complex data may risk key insights and alternative viewpoints being missed. Therefore, coding is often performed by multiple researchers.
A common strategy must be defined at the beginning of the coding process (Busetto et al., 2020). This includes establishing a useful coding list and finding a common definition of individual codes.
Transcripts are initially coded independently by researchers and then compared and consolidated to minimize error or bias and to bring confirmation of findings.
Member checking
Member checking (or respondent validation) involves checking back with participants to see if the research resonates with their experiences (Russell & Gregory, 2003).
Data can be returned to participants after data collection or when results are first available. For example, participants may be provided with their interview transcript and asked to verify whether this is a complete and accurate representation of their views.
Participants may then clarify or elaborate on their responses to ensure they align with their views (Shenton, 2004).
This feedback becomes part of data collection and ensures accurate descriptions/ interpretations of phenomena (Mays & Pope, 2000).
Reflexivity in qualitative research
Reflexivity typically involves examining your own judgments, practices, and belief systems during data collection and analysis. It aims to identify any personal beliefs which may affect the research.
Reflexivity is essential in qualitative research to ensure methodological transparency and complete reporting. This enables readers to understand how the interaction between the researcher and participant shapes the data.
Depending on the research question and population being researched, factors that need to be considered include the experience of the researcher, how the contact was established and maintained, age, gender, and ethnicity.
These details are important because, in qualitative research, the researcher is a dynamic part of the research process and actively influences the outcome of the research (Boeije, 2014).
Reflexivity Example
Who you are and your characteristics influence how you collect and analyze data. Here is an example of a reflexivity statement for research on smoking. I am a 30-year-old white female from a middle-class background. I live in the southwest of England and have been educated to master’s level. I have been involved in two research projects on oral health. I have never smoked, but I have witnessed how smoking can cause ill health from my volunteering in a smoking cessation clinic. My research aspirations are to help to develop interventions to help smokers quit.
Establishing Trustworthiness in Qualitative Research
Trustworthiness is a concept used to assess the quality and rigor of qualitative research. Four criteria are used to assess a study’s trustworthiness: credibility, transferability, dependability, and confirmability.
1. Credibility in Qualitative Research
Credibility refers to how accurately the results represent the reality and viewpoints of the participants.
To establish credibility in research, participants’ views and the researcher’s representation of their views need to align (Tobin & Begley, 2004).
To increase the credibility of findings, researchers may use data source triangulation, investigator triangulation, peer debriefing, or member checking (Lincoln & Guba, 1985).
2. Transferability in Qualitative Research
Transferability refers to how generalizable the findings are: whether the findings may be applied to another context, setting, or group (Tobin & Begley, 2004).
Transferability can be enhanced by giving thorough and in-depth descriptions of the research setting, sample, and methods (Nowell et al., 2017).
3. Dependability in Qualitative Research
Dependability is the extent to which the study could be replicated under similar conditions and the findings would be consistent.
Researchers can establish dependability using methods such as audit trails so readers can see the research process is logical and traceable (Koch, 1994).
4. Confirmability in Qualitative Research
Confirmability is concerned with establishing that there is a clear link between the researcher’s interpretations/ findings and the data.
Researchers can achieve confirmability by demonstrating how conclusions and interpretations were arrived at (Nowell et al., 2017).
This enables readers to understand the reasoning behind the decisions made.
Audit Trails in Qualitative Research
An audit trail provides evidence of the decisions made by the researcher regarding theory, research design, and data collection, as well as the steps they have chosen to manage, analyze, and report data.
The researcher must provide a clear rationale to demonstrate how conclusions were reached in their study.
A clear description of the research path must be provided to enable readers to trace through the researcher’s logic (Halpren, 1983).
Researchers should maintain records of the raw data, field notes, transcripts, and a reflective journal in order to provide a clear audit trail.
Discovery of unexpected data
Open-ended questions in qualitative research mean the researcher can probe an interview topic and enable the participant to elaborate on responses in an unrestricted manner.
This allows unexpected data to emerge, which can lead to further research into that topic.
The exploratory nature of qualitative research helps generate hypotheses that can be tested quantitatively (Busetto et al., 2020).
Flexibility
Data collection and analysis can be modified and adapted to take the research in a different direction if new ideas or patterns emerge in the data.
This enables researchers to investigate new opportunities while firmly maintaining their research goals.
Naturalistic settings
The behaviors of participants are recorded in real-world settings. Studies that use real-world settings have high ecological validity since participants behave more authentically.
Limitations
Time-consuming .
Qualitative research results in large amounts of data which often need to be transcribed and analyzed manually.
Even when software is used, transcription can be inaccurate, and using software for analysis can result in many codes which need to be condensed into themes.
Subjectivity
The researcher has an integral role in collecting and interpreting qualitative data. Therefore, the conclusions reached are from their perspective and experience.
Consequently, interpretations of data from another researcher may vary greatly.
Limited generalizability
The aim of qualitative research is to provide a detailed, contextualized understanding of an aspect of the human experience from a relatively small sample size.
Despite rigorous analysis procedures, conclusions drawn cannot be generalized to the wider population since data may be biased or unrepresentative.
Therefore, results are only applicable to a small group of the population.
While individual qualitative studies are often limited in their generalizability due to factors such as sample size and context, metasynthesis enables researchers to synthesize findings from multiple studies, potentially leading to more generalizable conclusions.
By integrating findings from studies conducted in diverse settings and with different populations, metasynthesis can provide broader insights into the phenomenon of interest.
Extraneous variables
Qualitative research is often conducted in real-world settings. This may cause results to be unreliable since extraneous variables may affect the data, for example:
- Situational variables : different environmental conditions may influence participants’ behavior in a study. The random variation in factors (such as noise or lighting) may be difficult to control in real-world settings.
- Participant characteristics : this includes any characteristics that may influence how a participant answers/ behaves in a study. This may include a participant’s mood, gender, age, ethnicity, sexual identity, IQ, etc.
- Experimenter effect : experimenter effect refers to how a researcher’s unintentional influence can change the outcome of a study. This occurs when (i) their interactions with participants unintentionally change participants’ behaviors or (ii) due to errors in observation, interpretation, or analysis.
What sample size should qualitative research be?
The sample size for qualitative studies has been recommended to include a minimum of 12 participants to reach data saturation (Braun, 2013).
Are surveys qualitative or quantitative?
Surveys can be used to gather information from a sample qualitatively or quantitatively. Qualitative surveys use open-ended questions to gather detailed information from a large sample using free text responses.
The use of open-ended questions allows for unrestricted responses where participants use their own words, enabling the collection of more in-depth information than closed-ended questions.
In contrast, quantitative surveys consist of closed-ended questions with multiple-choice answer options. Quantitative surveys are ideal to gather a statistical representation of a population.
What are the ethical considerations of qualitative research?
Before conducting a study, you must think about any risks that could occur and take steps to prevent them. Participant Protection : Researchers must protect participants from physical and mental harm. This means you must not embarrass, frighten, offend, or harm participants. Transparency : Researchers are obligated to clearly communicate how they will collect, store, analyze, use, and share the data. Confidentiality : You need to consider how to maintain the confidentiality and anonymity of participants’ data.
What is triangulation in qualitative research?
Triangulation refers to the use of several approaches in a study to comprehensively understand phenomena. This method helps to increase the validity and credibility of research findings.
Types of triangulation include method triangulation (using multiple methods to gather data); investigator triangulation (multiple researchers for collecting/ analyzing data), theory triangulation (comparing several theoretical perspectives to explain a phenomenon), and data source triangulation (using data from various times, locations, and people; Carter et al., 2014).
Why is qualitative research important?
Qualitative research allows researchers to describe and explain the social world. The exploratory nature of qualitative research helps to generate hypotheses that can then be tested quantitatively.
In qualitative research, participants are able to express their thoughts, experiences, and feelings without constraint.
Additionally, researchers are able to follow up on participants’ answers in real-time, generating valuable discussion around a topic. This enables researchers to gain a nuanced understanding of phenomena which is difficult to attain using quantitative methods.
What is coding data in qualitative research?
Coding data is a qualitative data analysis strategy in which a section of text is assigned with a label that describes its content.
These labels may be words or phrases which represent important (and recurring) patterns in the data.
This process enables researchers to identify related content across the dataset. Codes can then be used to group similar types of data to generate themes.
What is the difference between qualitative and quantitative research?
Qualitative research involves the collection and analysis of non-numerical data in order to understand experiences and meanings from the participant’s perspective.
This can provide rich, in-depth insights on complicated phenomena. Qualitative data may be collected using interviews, focus groups, or observations.
In contrast, quantitative research involves the collection and analysis of numerical data to measure the frequency, magnitude, or relationships of variables. This can provide objective and reliable evidence that can be generalized to the wider population.
Quantitative data may be collected using closed-ended questionnaires or experiments.
What is trustworthiness in qualitative research?
Trustworthiness is a concept used to assess the quality and rigor of qualitative research. Four criteria are used to assess a study’s trustworthiness: credibility, transferability, dependability, and confirmability.
Credibility refers to how accurately the results represent the reality and viewpoints of the participants. Transferability refers to whether the findings may be applied to another context, setting, or group.
Dependability is the extent to which the findings are consistent and reliable. Confirmability refers to the objectivity of findings (not influenced by the bias or assumptions of researchers).
What is data saturation in qualitative research?
Data saturation is a methodological principle used to guide the sample size of a qualitative research study.
Data saturation is proposed as a necessary methodological component in qualitative research (Saunders et al., 2018) as it is a vital criterion for discontinuing data collection and/or analysis.
The intention of data saturation is to find “no new data, no new themes, no new coding, and ability to replicate the study” (Guest et al., 2006). Therefore, enough data has been gathered to make conclusions.
Why is sampling in qualitative research important?
In quantitative research, large sample sizes are used to provide statistically significant quantitative estimates.
This is because quantitative research aims to provide generalizable conclusions that represent populations.
However, the aim of sampling in qualitative research is to gather data that will help the researcher understand the depth, complexity, variation, or context of a phenomenon. The small sample sizes in qualitative studies support the depth of case-oriented analysis.
What is narrative analysis?
Narrative analysis is a qualitative research method used to understand how individuals create stories from their personal experiences.
There is an emphasis on understanding the context in which a narrative is constructed, recognizing the influence of historical, cultural, and social factors on storytelling.
Researchers can use different methods together to explore a research question.
Some narrative researchers focus on the content of what is said, using thematic narrative analysis, while others focus on the structure, such as holistic-form or categorical-form structural narrative analysis. Others focus on how the narrative is produced and performed.
Boeije, H. (2014). Analysis in qualitative research. Sage.
Braun, V., & Clarke, V. (2006). Using thematic analysis in psychology. Qualitative research in psychology , 3 (2), 77-101. https://doi.org/10.1191/1478088706qp063oa
Brooks, J., McCluskey, S., Turley, E., & King, N. (2014). The utility of template analysis in qualitative psychology research. Qualitative Research in Psychology , 12 (2), 202–222. https://doi.org/10.1080/14780887.2014.955224
Busetto, L., Wick, W., & Gumbinger, C. (2020). How to use and assess qualitative research methods. Neurological research and practice , 2 (1), 14-14. https://doi.org/10.1186/s42466-020-00059-z
Carter, N., Bryant-Lukosius, D., DiCenso, A., Blythe, J., & Neville, A. J. (2014). The use of triangulation in qualitative research. Oncology nursing forum , 41 (5), 545–547. https://doi.org/10.1188/14.ONF.545-547
Critical Appraisal Skills Programme. (2018). CASP Checklist: 10 questions to help you make sense of a Qualitative research. https://casp-uk.net/images/checklist/documents/CASP-Qualitative-Studies-Checklist/CASP-Qualitative-Checklist-2018_fillable_form.pdf Accessed: March 15 2023
Clarke, V., & Braun, V. (2013). Successful qualitative research: A practical guide for beginners. Successful Qualitative Research , 1-400.
Denny, E., & Weckesser, A. (2022). How to do qualitative research?: Qualitative research methods. BJOG : an international journal of obstetrics and gynaecology , 129 (7), 1166-1167. https://doi.org/10.1111/1471-0528.17150
Glaser, B. G., & Strauss, A. L. (2017). The discovery of grounded theory. The Discovery of Grounded Theory , 1–18. https://doi.org/10.4324/9780203793206-1
Guest, G., Bunce, A., & Johnson, L. (2006). How many interviews are enough? An experiment with data saturation and variability. Field Methods, 18 (1), 59-82. doi:10.1177/1525822X05279903
Halpren, E. S. (1983). Auditing naturalistic inquiries: The development and application of a model (Unpublished doctoral dissertation). Indiana University, Bloomington.
Hammarberg, K., Kirkman, M., & de Lacey, S. (2016). Qualitative research methods: When to use them and how to judge them. Human Reproduction , 31 (3), 498–501. https://doi.org/10.1093/humrep/dev334
Koch, T. (1994). Establishing rigour in qualitative research: The decision trail. Journal of Advanced Nursing, 19, 976–986. doi:10.1111/ j.1365-2648.1994.tb01177.x
Lincoln, Y., & Guba, E. G. (1985). Naturalistic inquiry. Newbury Park, CA: Sage.
Mays, N., & Pope, C. (2000). Assessing quality in qualitative research. BMJ, 320(7226), 50–52.
Minichiello, V. (1990). In-Depth Interviewing: Researching People. Longman Cheshire.
Nowell, L. S., Norris, J. M., White, D. E., & Moules, N. J. (2017). Thematic Analysis: Striving to Meet the Trustworthiness Criteria. International Journal of Qualitative Methods, 16 (1). https://doi.org/10.1177/1609406917733847
Petty, N. J., Thomson, O. P., & Stew, G. (2012). Ready for a paradigm shift? part 2: Introducing qualitative research methodologies and methods. Manual Therapy , 17 (5), 378–384. https://doi.org/10.1016/j.math.2012.03.004
Punch, K. F. (2013). Introduction to social research: Quantitative and qualitative approaches. London: Sage
Reeves, S., Kuper, A., & Hodges, B. D. (2008). Qualitative research methodologies: Ethnography. BMJ , 337 (aug07 3). https://doi.org/10.1136/bmj.a1020
Russell, C. K., & Gregory, D. M. (2003). Evaluation of qualitative research studies. Evidence Based Nursing, 6 (2), 36–40.
Saunders, B., Sim, J., Kingstone, T., Baker, S., Waterfield, J., Bartlam, B., Burroughs, H., & Jinks, C. (2018). Saturation in qualitative research: exploring its conceptualization and operationalization. Quality & quantity , 52 (4), 1893–1907. https://doi.org/10.1007/s11135-017-0574-8
Scarduzio, J. A. (2017). Emic approach to qualitative research. The International Encyclopedia of Communication Research Methods, 1–2 . https://doi.org/10.1002/9781118901731.iecrm0082
Schreier, M. (2012). Qualitative content analysis in practice / Margrit Schreier.
Shenton, A. K. (2004). Strategies for ensuring trustworthiness in qualitative research projects. Education for Information, 22 , 63–75.
Starks, H., & Trinidad, S. B. (2007). Choose your method: a comparison of phenomenology, discourse analysis, and grounded theory. Qualitative health research , 17 (10), 1372–1380. https://doi.org/10.1177/1049732307307031
Tenny, S., Brannan, J. M., & Brannan, G. D. (2022). Qualitative Study. In StatPearls. StatPearls Publishing.
Tobin, G. A., & Begley, C. M. (2004). Methodological rigour within a qualitative framework. Journal of Advanced Nursing, 48, 388–396. doi:10.1111/j.1365-2648.2004.03207.x
Vaismoradi, M., Turunen, H., & Bondas, T. (2013). Content analysis and thematic analysis: Implications for conducting a qualitative descriptive study. Nursing & health sciences , 15 (3), 398-405. https://doi.org/10.1111/nhs.12048
Wood L. A., Kroger R. O. (2000). Doing discourse analysis: Methods for studying action in talk and text. Sage.
Yilmaz, K. (2013). Comparison of Quantitative and Qualitative Research Traditions: epistemological, theoretical, and methodological differences. European journal of education , 48 (2), 311-325. https://doi.org/10.1111/ejed.12014
- AI & NLP
- Churn & Loyalty
- Customer Experience
- Customer Journeys
- Customer Metrics
- Feedback Analysis
- Product Experience
- Product Updates
- Sentiment Analysis
- Surveys & Feedback Collection
- Text Analytics
- Try Thematic
Welcome to the community

The Primary Methods of Qualitative Data Analysis
In academic research as well as in the business landscape, qualitative data analysis plays a crucial role in understanding and interpreting non-numerical data.
Qualitative data analysis helps us make sense of the stories and personal narratives. In the business context, qualitative data analysis turns customer feedback into an in-depth understanding of what matters to customers. Sharing the insights from this analysis with decision-makers helps them drive initiatives that improve customer experiences.
While quantitative data analysis focuses on numerical measurement and statistical analysis, qualitative data analysis delves into the rich and complex nature of human experiences and perceptions. When analyzed effectively, customer feedback can be transformed into actionable insights for every team across the company.
This guide will provide an in-depth exploration of the different methods employed in qualitative data analysis, as well as the steps involved and challenges encountered. We’ll also have a look at what QDA means in the business context and how to turn it into a high-powered tool for CX and product teams.
Understanding Qualitative Data Analysis Methods
Definition and importance of qualitative data analysis.
Qualitative data analysis refers to the systematic process of examining and interpreting non-numerical data to gain meaningful insights and generate new knowledge. It’s what happens when you put a year’s worth of Amazon reviews into a thematic analysis engine, and end up with a thorough understanding of how users interact with your product (and half a dozen actionable insights to boot).
It involves dissecting text, images, videos, and other forms of qualitative data to identify patterns, themes, and relationships.
By capturing the nuances and depth of human experiences, the qualitative data analysis approach allows researchers to explore complex social phenomena that quantitative approaches cannot fully capture. It provides a rich and detailed understanding of social contexts, individual perspectives, and subjective experiences.
Qualitative data analysis methods offer an in-depth exploration of the hows and whys behind social phenomena, enabling researchers to gain a comprehensive understanding of complex social issues. It is incredibly valuable in fields such as sociology, anthropology, psychology, and education, where human behavior and social interactions are studied.
In these fields, researchers often seek to understand the intricacies of human experiences, and qualitative data analysis allows them to capture the complexity of these phenomena.
In the world of business & product development, qualitative data analysis methods can work to improve user experiences. Suddenly, you’ve got the opportunity to reach a comprehensive understanding of just what your products mean on the social landscape.
User feedback gets transformed into big-picture knowledge that offers a 360-degree view of how a product performs in the real world. Product teams get a solid, reliable basis on which to make decisions , and guesswork becomes a thing of the past.
Key Principles of Qualitative Data Analysis
Before delving into the various methods of qualitative data analysis, let’s look at the key principles that underpin these analysis techniques. Qualitative data analysis is guided by the following principles:
- Inductive Reasoning: Qualitative research focuses on specific observations and gradually develops broader interpretations and theories. It allows for the discovery of new patterns and relationships through an iterative process of data investigation.
- Contextual Understanding: Qualitative data analysis emphasizes the importance of understanding the research context and the social, cultural, and historical factors that shape it. Context provides meaning and helps researchers identify themes as well as interpret and make sense of the data.
- Subjectivity and Reflexivity: When research is human-led, the researchers acknowledge and critically reflect upon their own beliefs, biases, and experiences throughout the qualitative data analysis process. Where research is AI-driven, humans get a chance to view the actual data each insight is based on and check to see if it makes objective sense.
- Active Engagement: A qualitative data analysis method is an active and dynamic process that involves constant engagement with the data. Thematic analysis works most effectively as an ongoing process, thoroughly examining and interpreting all available data, while continually questioning and refining the research questions and analysis as new data points are added.
Inductive reasoning is a fundamental principle of qualitative data analysis. It allows researchers to start with specific observations and gradually develop broader interpretations and theories. Through this iterative process of data investigation, new patterns and relationships can be discovered. When you’ve got AI-driven data analysis software, this inductive reasoning is going on under the hood.
Contextual understanding is another key principle of the qualitative analysis process. It emphasizes the importance of understanding the research context and the social, cultural, and historical factors that shape it.
By considering the context when analyzing qualitative data, researchers can gain a deeper understanding of the data and interpret it more accurately. Well-designed thematic analysis software has this built in.
Subjectivity and reflexivity are essential principles in qualitative data analysis. Qualitative data analysis research must be repeatable if it is to be relied on, and there should always be ways to check just what qualitative feedback particular trends and insights come from. When qualitative data analysis is done right, transparency and rigor can be maintained throughout the process, from the initial selection of research questions and gathering of raw data to final analysis techniques.
Active engagement is a crucial aspect of qualitative feedback interpretation. It involves constant engagement with the data, as researchers thoroughly examine and interpret it. This active and dynamic process allows researchers to continually question and refine their qualitative analysis, ensuring a comprehensive understanding of the data.
Different Qualitative Data Analysis Methods
Just how does qualitative analysis work out in practice? In this article, we will explore five commonly used qualitative analysis methods: content analysis, narrative analysis, discourse analysis, grounded theory, and thematic analysis.
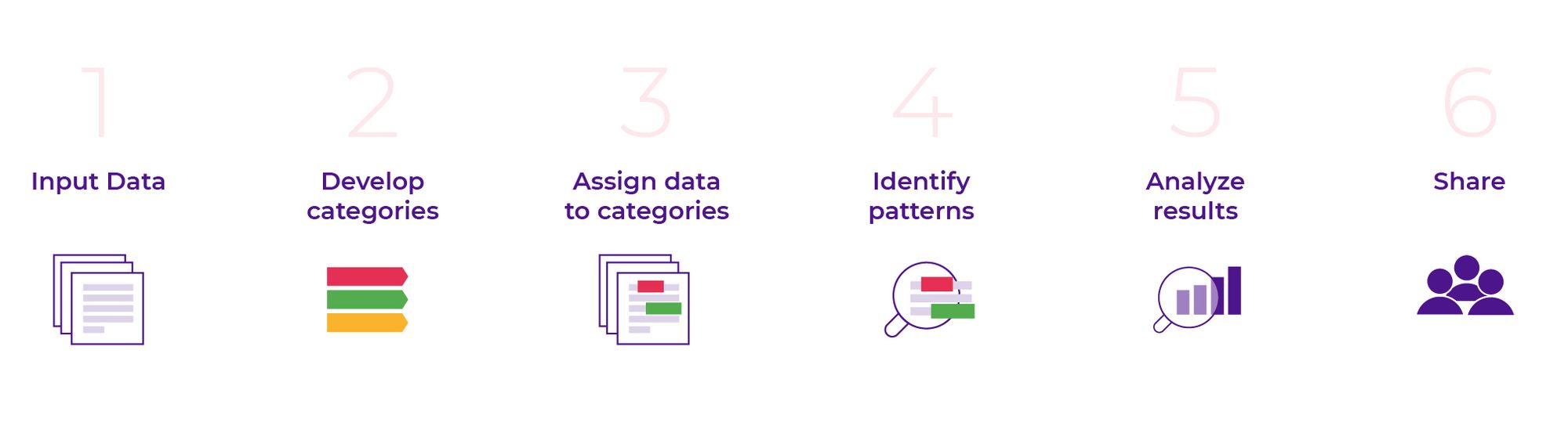
- Content Analysis
Content analysis is a systematic and objective approach to analyzing data by categorizing, coding, and quantifying specific words, themes, or concepts within a text. It involves identifying patterns, frequencies, and relationships in the content, which can be textual, visual, or auditory.
Researchers can employ content analysis techniques to examine interviews, focus group discussions, newspaper articles, social media posts, and other forms of textual data. By assigning codes to different segments of the text, researchers can identify recurring themes, sentiments, or messages.
This same qualitative data analysis approach can be used by CX and product teams to analyze customer feedback or support tickets.
For example, in an analysis of public response to a new product, a PX team might use content analysis to analyze social media posts discussing the topic.
By categorizing the posts based on their stance (e.g., positive, negative, neutral) and identifying recurring themes (e.g., user experience, look and feel), a company could gain insights into the dominant narratives and public perceptions surrounding the product launch.

- Narrative Analysis
Narrative analysis focuses on interpreting and understanding the stories and personal narratives shared by individuals. Researchers analyze the structure, content, and meaning of these narratives to gain insights into how individuals make sense of their experiences, construct identities, and communicate their perspectives.
Through narrative analysis techniques, qualitative researchers explore the plot, characters, setting, and themes within a narrative. They examine how the narrator constructs meaning, conveys emotions, and positions themselves within the story.
This same narrative analysis method is often used in psychology, sociology, and anthropology to understand identity formation, life histories, and personal narratives. It can be used in a business setting to analyze long-form responses and user interviews or descriptions of user behavior.
For instance, in a study on the experiences of cancer survivors, researchers may conduct narrative analysis on interviews with survivors. By examining the narratives, researchers can identify common themes such as coping strategies, support systems, and personal growth.
This qualitative analysis process can provide valuable insights into the lived experiences of cancer survivors and inform interventions and support programs.

- Discourse Analysis
Discourse analysis examines the social, cultural, and power relations that shape language use in different contexts. It focuses on the ways in which language constructs and reflects social reality, identities, and ideologies.
Researchers employing discourse analysis analyze data that includes spoken or written language, including interviews, speeches, media articles, and conversations.
They examine linguistic features such as metaphors, power dynamics, framing, and silences to uncover underlying social structures and processes.
For example, in a study on gender representation in media, researchers may use discourse analysis to analyze television advertisements. By examining the language, visual cues, and narratives used in the advertisements, researchers can identify how gender roles and stereotypes are constructed and reinforced.
It can shed light on the ways in which media perpetuates or challenges societal norms and expectations.
Another example might be using discourse analysis to analyze Tik Tok and YouTube videos to understand the societal responses to a rebranding; for instance, that from Twitter to X. Customer interviews are another good source for this analysis method.
- Grounded Theory
Grounded theory is an approach to qualitative analysis that aims to develop theories and concepts grounded in data. It involves iterative data collection and analysis to develop an inductive theory that emerges from the unstructured data itself.
Researchers using grounded theory analyze interviews, observations, and textual data to generate concepts and categories.
These concepts are continually refined and developed through theoretical sampling and constant comparison. Grounded theory analysis is particularly useful when exploring complex social phenomena where existing theories may be limited.
For instance, in a study on the experiences of individuals living with chronic pain, researchers may use grounded theory to analyze interviews with participants. Through iterative analysis, researchers can identify key concepts such as pain management strategies, social support networks, and psychological coping mechanisms.
These concepts can then be used to develop a theoretical framework within grounded theory that captures the multidimensional nature of living with chronic pain.
Although historically grounded theory analysis has been primarily used in the social sciences, grounded theory has also been used successfully for business inquiry.
- Thematic Analysis
Thematic analysis is a widely used method in qualitative data analysis that involves identifying, analyzing, and reporting patterns or themes within data. It is a flexible approach that can be applied across a variety of qualitative data, such as interview transcripts, survey responses, and observational notes.
When thematic analysis is done manually, researchers initially familiarize themselves with the raw data, reading through the material multiple times to gain a deep understanding.
Following this, they begin manual coding. The first step is to generate initial codes, which are tags or labels that identify important features of the data relevant to the research question.
These codes are then collated into potential themes, which are broader patterns that emerge across the data set.
Each theme is then reviewed and refined to ensure it accurately represents the coded data and the overall data set. The final step involves defining and naming the themes, during which researchers provide detailed analysis, including how themes relate to each other and to the research question.
Sound complicated? The great news is that advances in artificial intelligence mean we no longer have to do all that by hand.
Thematic analysis software can process thousands of pieces of consumer feedback in a matter of minutes, providing a user-friendly view of the themes and trends in the customer data pool.
What’s more, this type of software can be programmed to do content analysis, discourse analysis, and narrative analysis at the same time. The best comprehensive business solution for thematic analysis today is Thematic; a comprehensive feedback analysis that is designed for customer-centric businesses. It makes qualitative user analysis accessible to anyone, and is able to process feedback at scale.
Across disciplines, thematic analysis is particularly valued for its ability to provide a rich and detailed, yet complex account of data. It's a method that is accessible to researchers across different levels of qualitative research experience and can be applied to a variety of theoretical and epistemological approaches, making it a versatile tool in qualitative work.

Steps in Qualitative Data Analysis
Data collection.
Data collection is the initial phase of qualitative research and data analysis. It involves selecting appropriate methods to gather data such as interviews, observations, focus groups, or archival research.
Researchers may employ various techniques to collect data. These can include developing interview protocols, conducting observations, or collecting data using audio-visual recording devices.
They may need to consider ethical considerations, ensure informed consent, and establish rapport with participants to obtain rich and reliable data. The goal is to gather qualitative data that is relevant, comprehensive, and representative of the research topic.
Qualitative research questions can be more open-ended than those used for gathering quantitative data, and the research findings have the potential to be far more extensive.
In a business context, much of the work is done for you by customers who provide feedback in reviews, on support tickets, and on social media. Customer interviews are another possible source of rich data.
Data Coding
Data coding is the process of categorizing and organizing qualitative data into meaningful segments. When this is done manually, researchers assign codes to different parts of the data based on the emerging patterns, themes, or concepts identified during analysis. This coding process helps researchers manage and make sense of large amounts of qualitative data.
There are different types of codes used in analyzing raw data, including descriptive codes, interpretive codes, and conceptual codes.
Descriptive codes capture the content and surface-level meaning of all the data, while interpretive codes delve deeper into the underlying meanings and interpretations. A conceptual coding system further abstracts the research data by identifying broader concepts or theories.
Data Interpretation
Data interpretation involves making sense of the coded data and exploring the relationships, themes, and patterns that emerge from the analysis. Researchers critically examine the data, compare different codes, and then identify themes and connections between categories and concepts.
During data interpretation, researchers may engage in constant comparison, where they continually compare new data to existing codes and categories. This iterative process helps refine the analysis and identify theoretical insights.
It involves synthesizing the findings of qualitative and quantitative data and crafting a narrative that presents a comprehensive understanding of the research phenomenon.
Both data coding and data interpretation can be done by your qualitative analytics software, either in a research or business setting. In a corporate setting, CX /PX teams and customer service can then use information gained through the data interpretation step to drive favourable outcomes.
Performing Qualitative Data Analysis with Generative AI and LLM
Running manual grounded theory analysis or content analysis on a large amount of consumer feedback has never been a practical option. But that doesn’t mean qualitative research doesn’t make sense in a business context.
Generative AI, based on large language models (LLMs) can work with qualitative data at scale, analyze it, and derive the themes, connections and insights that can inform business decisions.
An LLM is a powerful machine learning model, based on deep learning and neural networks. It’s able to process and identify the complex relationships in natural language, and it can also understand user questions and moods and even generate text.
A natural languague processing LLM, trained on huge amounts of text data, could do all the work of a QDA researcher with the added benefits of easily verifiable, repeatable results.
Companies with extensive in-house talent may be able to build an in-house AI engine to analyze customer feedback and make sense of it— on a small scale. Those who are serious about getting real insights, though, will want to go with professional tools that have been trained on massive amounts of data and give reliable, dependable results.
Thematic is probably the best example of such a tool. Built to make sense of any amount of feedback data, it works in a highly transparent way that will leave you confident in every insight you derive.
It’s also incredibly user-friendly, with helpful visualizations and an easy-to-use dashboard that enables you to keep constant tabs on exactly what your users feel about the company. It’s never been easier to transform your user experience.
Modern Methods of Qualitative Data Analysis in Action: A Case Study

Instacart is one example of a company that discovered the power of qualitative data analysis. This company has 10 million end users, 500,000 personal shoppers, and more than 40,000 retailers. Processing all this qualitative data the traditional way would have been impossible, but Ant Marty, product operations team manager, found a method that worked.
Plugging data from the app into Thematic, she got real time information on everything happening among those millions of users: trends, themes, and deep understanding of what mattered to the people who made the company run.
Data collection is easy when you have an app with numerous feedback collection options. Data coding is automated by Thematic. And Thematic makes the first move in interpretation as well, providing insights that can be transformed by product teams into action plans and even a long-term vision.
Challenges Facing Qualitative Data Analysis Methods
Ensuring data validity and reliability.
One of the main challenges to a qualitative approach is ensuring the validity and reliability of the findings. Validity refers to the accuracy, truthfulness, and credibility of the data collected and analysis, while reliability refers to the consistency and replicability of the research process and findings.
Researchers address these challenges by employing rigorous data collection methods, ensuring data saturation, conducting member checks, and establishing inter-rater reliability. They also maintain reflexivity by critically reflecting on their assumptions, biases, and interpretations throughout the analysis process.
If you are a business using software to conduct qualitative research, your data validation check may be somewhat different, but it’s just as important. Some software, like Thematic, has validation built in, and the whole process is so transparent you can easily check and double-check where each insight comes from .
With other software options, you may have to run manual checks to ensure every piece of information provided has a firm basis.
Dealing with Subjectivity and Bias
Subjectivity and bias used to be considered inherent to qualitative research methods due to the interpretive nature of the process. Researchers bring their own perspectives, beliefs, and experiences, which can influence the analysis and interpretations.
To mitigate subjectivity and bias, researchers maintain transparency in their analytical processes by documenting their decision-making, providing detailed justifications for their interpretations, and engaging in peer debriefing and member checking. Using multiple researchers or an expert panel can also increase the credibility and reliability of the analysis.
Another way to decrease subjectivity is through thematic analysis software, which produces results that are repeatable and verifiable.
When it is all said and done, qualitative analysis offers a powerful and nuanced examination of human experiences and social phenomena. By employing diverse methods, adhering to key principles, and addressing potential limitations, researchers can harness the full potential of qualitative data to uncover rich insights and contribute to the advancement of knowledge.
Benefits of Qualitative Data Analysis Methods
Rich, in-depth insights.
A primary benefit of qualitative research techniques is their ability to provide rich, in-depth insights into complex phenomena. These methods delve deeply into human experiences, emotions, beliefs, and behaviors, offering a comprehensive understanding that is often unattainable through quantitative methods.
By exploring the nuances and subtleties of social interactions and personal experiences, qualitative analysis can uncover the layers of meaning that underpin human behavior. This depth of understanding is particularly valuable in fields like psychology, sociology, and anthropology, where the intricacies of human experience are central to the research question.
It is even more important for customer-focused businesses and enables them to create a product and a CX that meets their customer’s needs and desires. Quantitative analysis can provide a one-dimensional understanding of user behavior based on quantitative data, but when analysing qualitative data you get the why to every what.
Flexibility and Contextual Understanding
Another significant advantage of these analysis techniques is their inherent flexibility and capacity to provide contextual understanding. Unlike quantitative research, which relies on rigid structures and predefined hypotheses, qualitative research is adaptable to the evolving nature of the study.
This flexibility allows researchers to explore unexpected themes and patterns that emerge during the data collection process. Qualitative analysis is how businesses like Atlassian have created infinite customer feedback loops and powered their own infinitely evolving products.
Additionally, qualitative methods are sensitive to the context in which the data is collected, acknowledging and incorporating the environmental, cultural, and social factors that influence the data. The context-rich approach used to collect qualitative data ensures a more holistic understanding of the subject matter, making it particularly useful in cross-cultural studies, community research, and exploratory investigations.
Your product may have global reach, and users in different areas may interact with it in different ways– but qualitative techniques can take all that into account.
This considered, it should be no surprise that qualitative analysis techniques have become powerful tools for researchers seeking to understand the complexities of human behavior and social phenomena. Their ability to provide depth, context, and rich narrative data makes them indispensable tools in the arsenal of social science research, and there’s no better way to gain solid information to guide your business decisions.
Whether you’re a researcher keen on analyzing and interpreting qualitative data or an entrepreneur keen on making your business more customer-centric, this research method is likely to become your next best friend.
If you’re in academia, you may want to do it all manually, and that’s totally okay. But if it’s business intelligence you’re after— try out Thematic. Your future self will thank you, as will everyone else who views the end-of-year reports.
What are the five methods to analyze qualitative data?
The five chief methods of qualitative data analysis are:
The right analysis method for your use case will depend on what context, your research questions, and the form of data available to you.
What are good sources of data for qualitative data analysis?
In a business context user reviews, support tickets, customer surveys and social media posts are all great sources of data for qualitative analysis. In a research project, gathering qualitative data may mean conducting interviews, surveys, or focus groups.
What are the benefits of qualitative data analysis?
Two big benefits of qualitative data analysis include:
- Rich, in-depth insights
- Flexibility and contextual understanding
In a business context, this translates into a loyal, well-satisfied user base, a successful product, and an upwards-ticking revenue curve. Research objectives for social sciences may include a better understanding of social dynamics or human relations.
What are the challenges of qualitative data analysis?
The two prime challenges of qualitative data analysis techniques are:
- Ensuring data validity and reliability
- Dealing with subjectivity and bias
What is the best tool for qualitative data analysis?
While a number of other options do exist, the best comprehensive software for qualitative data analysis in a business context today is Thematic.

Digital Marketing
Tyler manages Thematic's content & aims to create useful & informative content for CX, Product & Insight professionals.
We make it easy to discover the customer and product issues that matter.
Unlock the value of feedback at scale, in one platform. Try it for free now!
- Questions to ask your Feedback Analytics vendor
- How to end customer churn for good
- Scalable analysis of NPS verbatims
- 5 Text analytics approaches
- How to calculate the ROI of CX
Our experts will show you how Thematic works, how to discover pain points and track the ROI of decisions. To access your free trial, book a personal demo today.
Recent posts
Become a qualitative theming pro! Creating a perfect code frame is hard, but thematic analysis software makes the process much easier.
Discover the power of thematic analysis to unlock insights from qualitative data. Learn about manual vs. AI-powered approaches, best practices, and how Thematic software can revolutionize your analysis workflow.
When two major storms wreaked havoc on Auckland and Watercare’s infrastructurem the utility went through a CX crisis. With a massive influx of calls to their support center, Thematic helped them get inisghts from this data to forge a new approach to restore services and satisfaction levels.
Extract insights from Customer & Employee Interviews. At Scale.
Comprehensive guide to data analysis methods for qualitative research.
Home » Comprehensive guide to data analysis methods for qualitative research
Qualitative Analysis Techniques serve as the backbone of understanding complex human behaviors and perspectives. These techniques allow researchers to explore meanings, experiences, and social contexts that quantitative methods often overlook. By harnessing interviews, focus groups, and observations, one can capture rich, detailed narratives that reveal the subtleties of participants’ thoughts and feelings.
Engaging with qualitative data requires a clear framework for analysis. Researchers must select appropriate methods, such as thematic analysis or grounded theory, to interpret the collected information effectively. Understanding these techniques is crucial for drawing insightful conclusions and ultimately enhancing the research outcome. This guide aims to equip readers with essential knowledge for utilizing qualitative analysis effectively.
Common Qualitative Analysis Techniques
Qualitative analysis techniques are essential in uncovering deep insights from qualitative data. These methods help researchers interpret and understand complexities in human behavior, thoughts, and experiences. Common techniques include thematic analysis, grounded theory, and content analysis. Each technique has its strengths and is suited for different research questions.
Thematic analysis involves identifying and analyzing patterns within qualitative data. Researchers categorize data into themes to reveal important insights. Grounded theory, conversely, generates new theories from collected data rather than testing existing ones. Content analysis quantitatively assesses the presence of specific words or themes, allowing for a systematic examination of the data corpus. Employing these qualitative analysis techniques can significantly enhance the depth and quality of qualitative research, ensuring careful consideration of context and meaning in findings.
Thematic Analysis in Qualitative Research
Thematic analysis is a popular technique in qualitative research that enables researchers to identify, analyze, and report patterns within data. This method provides an accessible framework for understanding the insights derived from participant responses or textual materials. By focusing on themes, researchers can uncover significant meanings and connections that might be obscured in quantitative approaches.
The process of thematic analysis typically unfolds in several stages. First, researchers familiarize themselves with the data by immersing themselves in the content. Next, they systematically code relevant data segments that highlight significant features. Following this, themes are constructed from these codes, allowing for broader patterns to emerge. Finally, a detailed analysis of these themes leads to an insightful report, revealing the relationships between various data elements. This approach is particularly effective for qualitative analysis techniques, as it emphasizes the richness of participant perspectives while providing a clear structure for interpretation.
Grounded Theory: Building From Data
Grounded Theory offers a systematic approach to qualitative analysis techniques by building theories directly from the data collected. Researchers begin by gathering extensive qualitative data through interviews, focus groups, or observations. This data serves as the foundation for developing concepts and frameworks that explain the phenomena under study. It emphasizes the importance of data-driven insights, enabling researchers to explore patterns and relationships organically.
Through coding processes, researchers identify key themes and categories that emerge from the data. This iterative approach allows for constant comparison, refining theories as new data is collected. Grounded Theory not only simplifies analysis but also ensures that findings are deeply rooted in actual experiences. By engaging with participants’ perspectives, researchers effectively reveal the complex social contexts that shape behaviors and attitudes. This method enhances the richness and validity of qualitative research, offering truly impactful insights into human behavior.
Advanced Qualitative Analysis Techniques
Advanced qualitative analysis techniques play a crucial role in extracting deeper insights from qualitative data. These methods enable researchers to identify patterns and themes within complex datasets, enhancing understanding of participant perspectives. Techniques such as thematic analysis, grounded theory, and narrative analysis facilitate a rich exploration of qualitative data, allowing for a comprehensive interpretation.
One effective approach involves thematic analysis, where data is coded and categorized into meaningful themes. Grounded theory, on the other hand, is essential for developing theories based on the data itself. Additionally, narrative analysis focuses on how stories are constructed and the meanings behind them. Each of these qualitative analysis techniques offers unique benefits, ensuring a robust understanding of nuanced data and its implications. By mastering these methods, researchers can significantly enrich their analysis, ultimately leading to more impactful findings.
Narrative Analysis in Depth
Narrative analysis delves deep into the stories that qualitative data can tell, highlighting the significance of individual experiences. Recognizing how participants express their thoughts through narratives provides unique insights into their motivations and behaviors. This approach emphasizes understanding context rather than just viewing data as disconnected facts.
In narrative analysis, there are several important aspects to consider. First, focus on the structure of the story, as this reveals how individuals organize their thoughts. Second, analyze the themes present in the narratives, which can shed light on shared experiences or concerns. Finally, consider the role of the storyteller, as this often influences the interpretation of the narrative. By paying attention to these factors, researchers can extract valuable insights that support overall qualitative analysis techniques.
Discourse Analysis: Understanding Language Use
Discourse analysis serves as a profound method within qualitative analysis techniques, aiming to unravel the complexities of language use. By examining how language shapes social interactions and conveys meaning, researchers can gain deeper insights into the dynamics of communication. This technique focuses not only on the words themselves but also on the context and cultural nuances surrounding the conversations. It reveals how expressions can reflect social identities, power structures, and group relationships.
The process of discourse analysis often unfolds in several steps: identifying patterns in language, contextualizing the dialogues, and interpreting the underlying meanings. First, researchers look for recurring themes within the data. Next, they observe how external factors impact these interactions, such as cultural or situational contexts. Finally, the interpretations can lead to a richer understanding of the subjects at hand, making discourse analysis an invaluable tool for qualitative research. This holistic approach enables scholars to comprehensively explore human behavior, beliefs, and practices through the lens of language.
Conclusion: Mastering Qualitative Analysis Techniques
Mastering qualitative analysis techniques is essential for extracting meaningful insights from data. By employing a range of strategies, researchers can better understand their subjects, allowing for a deeper exploration of human experiences and perspectives. These techniques not only enhance data interpretation but also foster a greater appreciation of the nuances that come with qualitative research.
Effective qualitative analysis requires a thoughtful approach to coding data, recognizing patterns, and synthesizing information into coherent themes. This mastery empowers researchers to present their findings clearly, making the insights accessible and actionable for diverse audiences. By focusing on these techniques, scholars and practitioners alike can significantly improve the quality and impact of their qualitative research endeavors.
Turn interviews into actionable insights
On this Page
Coding for qualitative research: AI-powered tools
You may also like, how to cite qualitative research with author guidelines.
Effective Qualitative Study Data Collection Methods
Designing effective research design for qualitative studies.
Unlock Insights from Interviews 10x faster
- Request demo
- Get started for free
- MS in the Learning Sciences
- Tuition & Financial Aid

Qualitative vs. quantitative data analysis: How do they differ?

Learning analytics have become the cornerstone for personalizing student experiences and enhancing learning outcomes. In this data-informed approach to education there are two distinct methodologies: qualitative and quantitative analytics. These methods, which are typical to data analytics in general, are crucial to the interpretation of learning behaviors and outcomes. This blog will explore the nuances that distinguish qualitative and quantitative research, while uncovering their shared roles in learning analytics, program design and instruction.
What is qualitative data?
Qualitative data is descriptive and includes information that is non numerical. Qualitative research is used to gather in-depth insights that can't be easily measured on a scale like opinions, anecdotes and emotions. In learning analytics qualitative data could include in depth interviews, text responses to a prompt, or a video of a class period. 1
What is quantitative data?
Quantitative data is information that has a numerical value. Quantitative research is conducted to gather measurable data used in statistical analysis. Researchers can use quantitative studies to identify patterns and trends. In learning analytics quantitative data could include test scores, student demographics, or amount of time spent in a lesson. 2
Key difference between qualitative and quantitative data
It's important to understand the differences between qualitative and quantitative data to both determine the appropriate research methods for studies and to gain insights that you can be confident in sharing.
Data Types and Nature
Examples of qualitative data types in learning analytics:
- Observational data of human behavior from classroom settings such as student engagement, teacher-student interactions, and classroom dynamics
- Textual data from open-ended survey responses, reflective journals, and written assignments
- Feedback and discussions from focus groups or interviews
- Content analysis from various media
Examples of quantitative data types:
- Standardized test, assessment, and quiz scores
- Grades and grade point averages
- Attendance records
- Time spent on learning tasks
- Data gathered from learning management systems (LMS), including login frequency, online participation, and completion rates of assignments
Methods of Collection
Qualitative and quantitative research methods for data collection can occasionally seem similar so it's important to note the differences to make sure you're creating a consistent data set and will be able to reliably draw conclusions from your data.
Qualitative research methods
Because of the nature of qualitative data (complex, detailed information), the research methods used to collect it are more involved. Qualitative researchers might do the following to collect data:
- Conduct interviews to learn about subjective experiences
- Host focus groups to gather feedback and personal accounts
- Observe in-person or use audio or video recordings to record nuances of human behavior in a natural setting
- Distribute surveys with open-ended questions
Quantitative research methods
Quantitative data collection methods are more diverse and more likely to be automated because of the objective nature of the data. A quantitative researcher could employ methods such as:
- Surveys with close-ended questions that gather numerical data like birthdates or preferences
- Observational research and record measurable information like the number of students in a classroom
- Automated numerical data collection like information collected on the backend of a computer system like button clicks and page views
Analysis techniques
Qualitative and quantitative data can both be very informative. However, research studies require critical thinking for productive analysis.
Qualitative data analysis methods
Analyzing qualitative data takes a number of steps. When you first get all your data in one place you can do a review and take notes of trends you think you're seeing or your initial reactions. Next, you'll want to organize all the qualitative data you've collected by assigning it categories. Your central research question will guide your data categorization whether it's by date, location, type of collection method (interview vs focus group, etc), the specific question asked or something else. Next, you'll code your data. Whereas categorizing data is focused on the method of collection, coding is the process of identifying and labeling themes within the data collected to get closer to answering your research questions. Finally comes data interpretation. To interpret the data you'll take a look at the information gathered including your coding labels and see what results are occurring frequently or what other conclusions you can make. 3
Quantitative analysis techniques
The process to analyze quantitative data can be time-consuming due to the large volume of data possible to collect. When approaching a quantitative data set, start by focusing in on the purpose of your evaluation. Without making a conclusion, determine how you will use the information gained from analysis; for example: The answers of this survey about study habits will help determine what type of exam review session will be most useful to a class. 4
Next, you need to decide who is analyzing the data and set parameters for analysis. For example, if two different researchers are evaluating survey responses that rank preferences on a scale from 1 to 5, they need to be operating with the same understanding of the rankings. You wouldn't want one researcher to classify the value of 3 to be a positive preference while the other considers it a negative preference. It's also ideal to have some type of data management system to store and organize your data, such as a spreadsheet or database. Within the database, or via an export to data analysis software, the collected data needs to be cleaned of things like responses left blank, duplicate answers from respondents, and questions that are no longer considered relevant. Finally, you can use statistical software to analyze data (or complete a manual analysis) to find patterns and summarize your findings. 4
Qualitative and quantitative research tools
From the nuanced, thematic exploration enabled by tools like NVivo and ATLAS.ti, to the statistical precision of SPSS and R for quantitative analysis, each suite of data analysis tools offers tailored functionalities that cater to the distinct natures of different data types.
Qualitative research software:
NVivo: NVivo is qualitative data analysis software that can do everything from transcribe recordings to create word clouds and evaluate uploads for different sentiments and themes. NVivo is just one tool from the company Lumivero, which offers whole suites of data processing software. 5
ATLAS.ti: Similar to NVivo, ATLAS.ti allows researchers to upload and import data from a variety of sources to be tagged and refined using machine learning and presented with visualizations and ready for insert into reports. 6
SPSS: SPSS is a statistical analysis tool for quantitative research, appreciated for its user-friendly interface and comprehensive statistical tests, which makes it ideal for educators and researchers. With SPSS researchers can manage and analyze large quantitative data sets, use advanced statistical procedures and modeling techniques, predict customer behaviors, forecast market trends and more. 7
R: R is a versatile and dynamic open-source tool for quantitative analysis. With a vast repository of packages tailored to specific statistical methods, researchers can perform anything from basic descriptive statistics to complex predictive modeling. R is especially useful for its ability to handle large datasets, making it ideal for educational institutions that generate substantial amounts of data. The programming language offers flexibility in customizing analysis and creating publication-quality visualizations to effectively communicate results. 8
Applications in Educational Research
Both quantitative and qualitative data can be employed in learning analytics to drive informed decision-making and pedagogical enhancements. In the classroom, quantitative data like standardized test scores and online course analytics create a foundation for assessing and benchmarking student performance and engagement. Qualitative insights gathered from surveys, focus group discussions, and reflective student journals offer a more nuanced understanding of learners' experiences and contextual factors influencing their education. Additionally feedback and practical engagement metrics blend these data types, providing a holistic view that informs curriculum development, instructional strategies, and personalized learning pathways. Through these varied data sets and uses, educators can piece together a more complete narrative of student success and the impacts of educational interventions.
Master Data Analysis with an M.S. in Learning Sciences From SMU
Whether it is the detailed narratives unearthed through qualitative data or the informative patterns derived from quantitative analysis, both qualitative and quantitative data can provide crucial information for educators and researchers to better understand and improve learning. Dive deeper into the art and science of learning analytics with SMU's online Master of Science in the Learning Sciences program . At SMU, innovation and inquiry converge to empower the next generation of educators and researchers. Choose the Learning Analytics Specialization to learn how to harness the power of data science to illuminate learning trends, devise impactful strategies, and drive educational innovation. You could also find out how advanced technologies like augmented reality (AR), virtual reality (VR), and artificial intelligence (AI) can revolutionize education, and develop the insight to apply embodied cognition principles to enhance learning experiences in the Learning and Technology Design Specialization , or choose your own electives to build a specialization unique to your interests and career goals.
For more information on our curriculum and to become part of a community where data drives discovery, visit SMU's MSLS program website or schedule a call with our admissions outreach advisors for any queries or further discussion. Take the first step towards transforming education with data today.
- Retrieved on August 8, 2024, from nnlm.gov/guides/data-glossary/qualitative-data
- Retrieved on August 8, 2024, from nnlm.gov/guides/data-glossary/quantitative-data
- Retrieved on August 8, 2024, from cdc.gov/healthyyouth/evaluation/pdf/brief19.pdf
- Retrieved on August 8, 2024, from cdc.gov/healthyyouth/evaluation/pdf/brief20.pdf
- Retrieved on August 8, 2024, from lumivero.com/solutions/
- Retrieved on August 8, 2024, from atlasti.com/
- Retrieved on August 8, 2024, from ibm.com/products/spss-statistics
- Retrieved on August 8, 2024, from cran.r-project.org/doc/manuals/r-release/R-intro.html#Introduction-and-preliminaries
Return to SMU Online Learning Sciences Blog
Southern Methodist University has engaged Everspring , a leading provider of education and technology services, to support select aspects of program delivery.
This will only take a moment
In order to utilize all of the features of this web site, JavaScript must be enabled in your browser.
Interpretative Phenomenological Analysis: Theory, Method and Research
Digital Logos Edition
In production
This book presents a comprehensive guide to interpretative phenomenological analysis (IPA) which is an increasingly popular approach to qualitative inquiry taught to undergraduate and postgraduate students today. The first chapter outlines the theoretical foundations for IPA. It discusses phenomenology, hermeneutics, and idiography and how they have been taken up by IPA. The next four chapters provide detailed, step by step guidelines to conducting IPA research: study design, data collection and interviewing, data analysis, and writing up. In the next section, the authors give extended worked examples from their own studies in health, sexuality, psychological distress, and identity to illustrate the breadth and depth of IPA research. The final section of the book considers how IPA connects with other contemporary qualitative approaches like discourse and narrative analysis and how it addresses issues to do with validity.
Key Features
- Presents a comprehensive guide to interpretative phenomenological analysis.
- Outlines the theoretical foundations for IPA.
- Provides detailed, step by step guidelines to conducting IPA research.
Product Details
- Title : Interpretative Phenomenological Analysis: Theory, Method and Research
- Authors : Jonathan A. Smith , Paul Flowers , Michael Larkin
- Edition: 2nd Edition
- Publisher : SAGE
- Print Publication Date: 2022
- Logos Release Date: 2024
- Era: era:contemporary
- Language : English
- Resources: 1
- Format : Digital › Logos Research Edition
- Subjects : Phenomenological psychology; Psychology › Research
- ISBNs : 9781529753806 , 9781529753790 , 1529753805 , 1529753791
- Resource ID: LLS:NTRPRTTVPHRSRCH
- Resource Type: Monograph
- Metadata Last Updated: 2024-08-16T22:26:51Z
Sign in with your Logos account
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Can J Hosp Pharm
- v.68(3); May-Jun 2015

Qualitative Research: Data Collection, Analysis, and Management
Introduction.
In an earlier paper, 1 we presented an introduction to using qualitative research methods in pharmacy practice. In this article, we review some principles of the collection, analysis, and management of qualitative data to help pharmacists interested in doing research in their practice to continue their learning in this area. Qualitative research can help researchers to access the thoughts and feelings of research participants, which can enable development of an understanding of the meaning that people ascribe to their experiences. Whereas quantitative research methods can be used to determine how many people undertake particular behaviours, qualitative methods can help researchers to understand how and why such behaviours take place. Within the context of pharmacy practice research, qualitative approaches have been used to examine a diverse array of topics, including the perceptions of key stakeholders regarding prescribing by pharmacists and the postgraduation employment experiences of young pharmacists (see “Further Reading” section at the end of this article).
In the previous paper, 1 we outlined 3 commonly used methodologies: ethnography 2 , grounded theory 3 , and phenomenology. 4 Briefly, ethnography involves researchers using direct observation to study participants in their “real life” environment, sometimes over extended periods. Grounded theory and its later modified versions (e.g., Strauss and Corbin 5 ) use face-to-face interviews and interactions such as focus groups to explore a particular research phenomenon and may help in clarifying a less-well-understood problem, situation, or context. Phenomenology shares some features with grounded theory (such as an exploration of participants’ behaviour) and uses similar techniques to collect data, but it focuses on understanding how human beings experience their world. It gives researchers the opportunity to put themselves in another person’s shoes and to understand the subjective experiences of participants. 6 Some researchers use qualitative methodologies but adopt a different standpoint, and an example of this appears in the work of Thurston and others, 7 discussed later in this paper.
Qualitative work requires reflection on the part of researchers, both before and during the research process, as a way of providing context and understanding for readers. When being reflexive, researchers should not try to simply ignore or avoid their own biases (as this would likely be impossible); instead, reflexivity requires researchers to reflect upon and clearly articulate their position and subjectivities (world view, perspectives, biases), so that readers can better understand the filters through which questions were asked, data were gathered and analyzed, and findings were reported. From this perspective, bias and subjectivity are not inherently negative but they are unavoidable; as a result, it is best that they be articulated up-front in a manner that is clear and coherent for readers.
THE PARTICIPANT’S VIEWPOINT
What qualitative study seeks to convey is why people have thoughts and feelings that might affect the way they behave. Such study may occur in any number of contexts, but here, we focus on pharmacy practice and the way people behave with regard to medicines use (e.g., to understand patients’ reasons for nonadherence with medication therapy or to explore physicians’ resistance to pharmacists’ clinical suggestions). As we suggested in our earlier article, 1 an important point about qualitative research is that there is no attempt to generalize the findings to a wider population. Qualitative research is used to gain insights into people’s feelings and thoughts, which may provide the basis for a future stand-alone qualitative study or may help researchers to map out survey instruments for use in a quantitative study. It is also possible to use different types of research in the same study, an approach known as “mixed methods” research, and further reading on this topic may be found at the end of this paper.
The role of the researcher in qualitative research is to attempt to access the thoughts and feelings of study participants. This is not an easy task, as it involves asking people to talk about things that may be very personal to them. Sometimes the experiences being explored are fresh in the participant’s mind, whereas on other occasions reliving past experiences may be difficult. However the data are being collected, a primary responsibility of the researcher is to safeguard participants and their data. Mechanisms for such safeguarding must be clearly articulated to participants and must be approved by a relevant research ethics review board before the research begins. Researchers and practitioners new to qualitative research should seek advice from an experienced qualitative researcher before embarking on their project.
DATA COLLECTION
Whatever philosophical standpoint the researcher is taking and whatever the data collection method (e.g., focus group, one-to-one interviews), the process will involve the generation of large amounts of data. In addition to the variety of study methodologies available, there are also different ways of making a record of what is said and done during an interview or focus group, such as taking handwritten notes or video-recording. If the researcher is audio- or video-recording data collection, then the recordings must be transcribed verbatim before data analysis can begin. As a rough guide, it can take an experienced researcher/transcriber 8 hours to transcribe one 45-minute audio-recorded interview, a process than will generate 20–30 pages of written dialogue.
Many researchers will also maintain a folder of “field notes” to complement audio-taped interviews. Field notes allow the researcher to maintain and comment upon impressions, environmental contexts, behaviours, and nonverbal cues that may not be adequately captured through the audio-recording; they are typically handwritten in a small notebook at the same time the interview takes place. Field notes can provide important context to the interpretation of audio-taped data and can help remind the researcher of situational factors that may be important during data analysis. Such notes need not be formal, but they should be maintained and secured in a similar manner to audio tapes and transcripts, as they contain sensitive information and are relevant to the research. For more information about collecting qualitative data, please see the “Further Reading” section at the end of this paper.
DATA ANALYSIS AND MANAGEMENT
If, as suggested earlier, doing qualitative research is about putting oneself in another person’s shoes and seeing the world from that person’s perspective, the most important part of data analysis and management is to be true to the participants. It is their voices that the researcher is trying to hear, so that they can be interpreted and reported on for others to read and learn from. To illustrate this point, consider the anonymized transcript excerpt presented in Appendix 1 , which is taken from a research interview conducted by one of the authors (J.S.). We refer to this excerpt throughout the remainder of this paper to illustrate how data can be managed, analyzed, and presented.
Interpretation of Data
Interpretation of the data will depend on the theoretical standpoint taken by researchers. For example, the title of the research report by Thurston and others, 7 “Discordant indigenous and provider frames explain challenges in improving access to arthritis care: a qualitative study using constructivist grounded theory,” indicates at least 2 theoretical standpoints. The first is the culture of the indigenous population of Canada and the place of this population in society, and the second is the social constructivist theory used in the constructivist grounded theory method. With regard to the first standpoint, it can be surmised that, to have decided to conduct the research, the researchers must have felt that there was anecdotal evidence of differences in access to arthritis care for patients from indigenous and non-indigenous backgrounds. With regard to the second standpoint, it can be surmised that the researchers used social constructivist theory because it assumes that behaviour is socially constructed; in other words, people do things because of the expectations of those in their personal world or in the wider society in which they live. (Please see the “Further Reading” section for resources providing more information about social constructivist theory and reflexivity.) Thus, these 2 standpoints (and there may have been others relevant to the research of Thurston and others 7 ) will have affected the way in which these researchers interpreted the experiences of the indigenous population participants and those providing their care. Another standpoint is feminist standpoint theory which, among other things, focuses on marginalized groups in society. Such theories are helpful to researchers, as they enable us to think about things from a different perspective. Being aware of the standpoints you are taking in your own research is one of the foundations of qualitative work. Without such awareness, it is easy to slip into interpreting other people’s narratives from your own viewpoint, rather than that of the participants.
To analyze the example in Appendix 1 , we will adopt a phenomenological approach because we want to understand how the participant experienced the illness and we want to try to see the experience from that person’s perspective. It is important for the researcher to reflect upon and articulate his or her starting point for such analysis; for example, in the example, the coder could reflect upon her own experience as a female of a majority ethnocultural group who has lived within middle class and upper middle class settings. This personal history therefore forms the filter through which the data will be examined. This filter does not diminish the quality or significance of the analysis, since every researcher has his or her own filters; however, by explicitly stating and acknowledging what these filters are, the researcher makes it easer for readers to contextualize the work.
Transcribing and Checking
For the purposes of this paper it is assumed that interviews or focus groups have been audio-recorded. As mentioned above, transcribing is an arduous process, even for the most experienced transcribers, but it must be done to convert the spoken word to the written word to facilitate analysis. For anyone new to conducting qualitative research, it is beneficial to transcribe at least one interview and one focus group. It is only by doing this that researchers realize how difficult the task is, and this realization affects their expectations when asking others to transcribe. If the research project has sufficient funding, then a professional transcriber can be hired to do the work. If this is the case, then it is a good idea to sit down with the transcriber, if possible, and talk through the research and what the participants were talking about. This background knowledge for the transcriber is especially important in research in which people are using jargon or medical terms (as in pharmacy practice). Involving your transcriber in this way makes the work both easier and more rewarding, as he or she will feel part of the team. Transcription editing software is also available, but it is expensive. For example, ELAN (more formally known as EUDICO Linguistic Annotator, developed at the Technical University of Berlin) 8 is a tool that can help keep data organized by linking media and data files (particularly valuable if, for example, video-taping of interviews is complemented by transcriptions). It can also be helpful in searching complex data sets. Products such as ELAN do not actually automatically transcribe interviews or complete analyses, and they do require some time and effort to learn; nonetheless, for some research applications, it may be a valuable to consider such software tools.
All audio recordings should be transcribed verbatim, regardless of how intelligible the transcript may be when it is read back. Lines of text should be numbered. Once the transcription is complete, the researcher should read it while listening to the recording and do the following: correct any spelling or other errors; anonymize the transcript so that the participant cannot be identified from anything that is said (e.g., names, places, significant events); insert notations for pauses, laughter, looks of discomfort; insert any punctuation, such as commas and full stops (periods) (see Appendix 1 for examples of inserted punctuation), and include any other contextual information that might have affected the participant (e.g., temperature or comfort of the room).
Dealing with the transcription of a focus group is slightly more difficult, as multiple voices are involved. One way of transcribing such data is to “tag” each voice (e.g., Voice A, Voice B). In addition, the focus group will usually have 2 facilitators, whose respective roles will help in making sense of the data. While one facilitator guides participants through the topic, the other can make notes about context and group dynamics. More information about group dynamics and focus groups can be found in resources listed in the “Further Reading” section.
Reading between the Lines
During the process outlined above, the researcher can begin to get a feel for the participant’s experience of the phenomenon in question and can start to think about things that could be pursued in subsequent interviews or focus groups (if appropriate). In this way, one participant’s narrative informs the next, and the researcher can continue to interview until nothing new is being heard or, as it says in the text books, “saturation is reached”. While continuing with the processes of coding and theming (described in the next 2 sections), it is important to consider not just what the person is saying but also what they are not saying. For example, is a lengthy pause an indication that the participant is finding the subject difficult, or is the person simply deciding what to say? The aim of the whole process from data collection to presentation is to tell the participants’ stories using exemplars from their own narratives, thus grounding the research findings in the participants’ lived experiences.
Smith 9 suggested a qualitative research method known as interpretative phenomenological analysis, which has 2 basic tenets: first, that it is rooted in phenomenology, attempting to understand the meaning that individuals ascribe to their lived experiences, and second, that the researcher must attempt to interpret this meaning in the context of the research. That the researcher has some knowledge and expertise in the subject of the research means that he or she can have considerable scope in interpreting the participant’s experiences. Larkin and others 10 discussed the importance of not just providing a description of what participants say. Rather, interpretative phenomenological analysis is about getting underneath what a person is saying to try to truly understand the world from his or her perspective.
Once all of the research interviews have been transcribed and checked, it is time to begin coding. Field notes compiled during an interview can be a useful complementary source of information to facilitate this process, as the gap in time between an interview, transcribing, and coding can result in memory bias regarding nonverbal or environmental context issues that may affect interpretation of data.
Coding refers to the identification of topics, issues, similarities, and differences that are revealed through the participants’ narratives and interpreted by the researcher. This process enables the researcher to begin to understand the world from each participant’s perspective. Coding can be done by hand on a hard copy of the transcript, by making notes in the margin or by highlighting and naming sections of text. More commonly, researchers use qualitative research software (e.g., NVivo, QSR International Pty Ltd; www.qsrinternational.com/products_nvivo.aspx ) to help manage their transcriptions. It is advised that researchers undertake a formal course in the use of such software or seek supervision from a researcher experienced in these tools.
Returning to Appendix 1 and reading from lines 8–11, a code for this section might be “diagnosis of mental health condition”, but this would just be a description of what the participant is talking about at that point. If we read a little more deeply, we can ask ourselves how the participant might have come to feel that the doctor assumed he or she was aware of the diagnosis or indeed that they had only just been told the diagnosis. There are a number of pauses in the narrative that might suggest the participant is finding it difficult to recall that experience. Later in the text, the participant says “nobody asked me any questions about my life” (line 19). This could be coded simply as “health care professionals’ consultation skills”, but that would not reflect how the participant must have felt never to be asked anything about his or her personal life, about the participant as a human being. At the end of this excerpt, the participant just trails off, recalling that no-one showed any interest, which makes for very moving reading. For practitioners in pharmacy, it might also be pertinent to explore the participant’s experience of akathisia and why this was left untreated for 20 years.
One of the questions that arises about qualitative research relates to the reliability of the interpretation and representation of the participants’ narratives. There are no statistical tests that can be used to check reliability and validity as there are in quantitative research. However, work by Lincoln and Guba 11 suggests that there are other ways to “establish confidence in the ‘truth’ of the findings” (p. 218). They call this confidence “trustworthiness” and suggest that there are 4 criteria of trustworthiness: credibility (confidence in the “truth” of the findings), transferability (showing that the findings have applicability in other contexts), dependability (showing that the findings are consistent and could be repeated), and confirmability (the extent to which the findings of a study are shaped by the respondents and not researcher bias, motivation, or interest).
One way of establishing the “credibility” of the coding is to ask another researcher to code the same transcript and then to discuss any similarities and differences in the 2 resulting sets of codes. This simple act can result in revisions to the codes and can help to clarify and confirm the research findings.
Theming refers to the drawing together of codes from one or more transcripts to present the findings of qualitative research in a coherent and meaningful way. For example, there may be examples across participants’ narratives of the way in which they were treated in hospital, such as “not being listened to” or “lack of interest in personal experiences” (see Appendix 1 ). These may be drawn together as a theme running through the narratives that could be named “the patient’s experience of hospital care”. The importance of going through this process is that at its conclusion, it will be possible to present the data from the interviews using quotations from the individual transcripts to illustrate the source of the researchers’ interpretations. Thus, when the findings are organized for presentation, each theme can become the heading of a section in the report or presentation. Underneath each theme will be the codes, examples from the transcripts, and the researcher’s own interpretation of what the themes mean. Implications for real life (e.g., the treatment of people with chronic mental health problems) should also be given.
DATA SYNTHESIS
In this final section of this paper, we describe some ways of drawing together or “synthesizing” research findings to represent, as faithfully as possible, the meaning that participants ascribe to their life experiences. This synthesis is the aim of the final stage of qualitative research. For most readers, the synthesis of data presented by the researcher is of crucial significance—this is usually where “the story” of the participants can be distilled, summarized, and told in a manner that is both respectful to those participants and meaningful to readers. There are a number of ways in which researchers can synthesize and present their findings, but any conclusions drawn by the researchers must be supported by direct quotations from the participants. In this way, it is made clear to the reader that the themes under discussion have emerged from the participants’ interviews and not the mind of the researcher. The work of Latif and others 12 gives an example of how qualitative research findings might be presented.
Planning and Writing the Report
As has been suggested above, if researchers code and theme their material appropriately, they will naturally find the headings for sections of their report. Qualitative researchers tend to report “findings” rather than “results”, as the latter term typically implies that the data have come from a quantitative source. The final presentation of the research will usually be in the form of a report or a paper and so should follow accepted academic guidelines. In particular, the article should begin with an introduction, including a literature review and rationale for the research. There should be a section on the chosen methodology and a brief discussion about why qualitative methodology was most appropriate for the study question and why one particular methodology (e.g., interpretative phenomenological analysis rather than grounded theory) was selected to guide the research. The method itself should then be described, including ethics approval, choice of participants, mode of recruitment, and method of data collection (e.g., semistructured interviews or focus groups), followed by the research findings, which will be the main body of the report or paper. The findings should be written as if a story is being told; as such, it is not necessary to have a lengthy discussion section at the end. This is because much of the discussion will take place around the participants’ quotes, such that all that is needed to close the report or paper is a summary, limitations of the research, and the implications that the research has for practice. As stated earlier, it is not the intention of qualitative research to allow the findings to be generalized, and therefore this is not, in itself, a limitation.
Planning out the way that findings are to be presented is helpful. It is useful to insert the headings of the sections (the themes) and then make a note of the codes that exemplify the thoughts and feelings of your participants. It is generally advisable to put in the quotations that you want to use for each theme, using each quotation only once. After all this is done, the telling of the story can begin as you give your voice to the experiences of the participants, writing around their quotations. Do not be afraid to draw assumptions from the participants’ narratives, as this is necessary to give an in-depth account of the phenomena in question. Discuss these assumptions, drawing on your participants’ words to support you as you move from one code to another and from one theme to the next. Finally, as appropriate, it is possible to include examples from literature or policy documents that add support for your findings. As an exercise, you may wish to code and theme the sample excerpt in Appendix 1 and tell the participant’s story in your own way. Further reading about “doing” qualitative research can be found at the end of this paper.
CONCLUSIONS
Qualitative research can help researchers to access the thoughts and feelings of research participants, which can enable development of an understanding of the meaning that people ascribe to their experiences. It can be used in pharmacy practice research to explore how patients feel about their health and their treatment. Qualitative research has been used by pharmacists to explore a variety of questions and problems (see the “Further Reading” section for examples). An understanding of these issues can help pharmacists and other health care professionals to tailor health care to match the individual needs of patients and to develop a concordant relationship. Doing qualitative research is not easy and may require a complete rethink of how research is conducted, particularly for researchers who are more familiar with quantitative approaches. There are many ways of conducting qualitative research, and this paper has covered some of the practical issues regarding data collection, analysis, and management. Further reading around the subject will be essential to truly understand this method of accessing peoples’ thoughts and feelings to enable researchers to tell participants’ stories.
Appendix 1. Excerpt from a sample transcript
The participant (age late 50s) had suffered from a chronic mental health illness for 30 years. The participant had become a “revolving door patient,” someone who is frequently in and out of hospital. As the participant talked about past experiences, the researcher asked:
- What was treatment like 30 years ago?
- Umm—well it was pretty much they could do what they wanted with you because I was put into the er, the er kind of system er, I was just on
- endless section threes.
- Really…
- But what I didn’t realize until later was that if you haven’t actually posed a threat to someone or yourself they can’t really do that but I didn’t know
- that. So wh-when I first went into hospital they put me on the forensic ward ’cause they said, “We don’t think you’ll stay here we think you’ll just
- run-run away.” So they put me then onto the acute admissions ward and – er – I can remember one of the first things I recall when I got onto that
- ward was sitting down with a er a Dr XXX. He had a book this thick [gestures] and on each page it was like three questions and he went through
- all these questions and I answered all these questions. So we’re there for I don’t maybe two hours doing all that and he asked me he said “well
- when did somebody tell you then that you have schizophrenia” I said “well nobody’s told me that” so he seemed very surprised but nobody had
- actually [pause] whe-when I first went up there under police escort erm the senior kind of consultants people I’d been to where I was staying and
- ermm so er [pause] I . . . the, I can remember the very first night that I was there and given this injection in this muscle here [gestures] and just
- having dreadful side effects the next day I woke up [pause]
- . . . and I suffered that akathesia I swear to you, every minute of every day for about 20 years.
- Oh how awful.
- And that side of it just makes life impossible so the care on the wards [pause] umm I don’t know it’s kind of, it’s kind of hard to put into words
- [pause]. Because I’m not saying they were sort of like not friendly or interested but then nobody ever seemed to want to talk about your life [pause]
- nobody asked me any questions about my life. The only questions that came into was they asked me if I’d be a volunteer for these student exams
- and things and I said “yeah” so all the questions were like “oh what jobs have you done,” er about your relationships and things and er but
- nobody actually sat down and had a talk and showed some interest in you as a person you were just there basically [pause] um labelled and you
- know there was there was [pause] but umm [pause] yeah . . .
This article is the 10th in the CJHP Research Primer Series, an initiative of the CJHP Editorial Board and the CSHP Research Committee. The planned 2-year series is intended to appeal to relatively inexperienced researchers, with the goal of building research capacity among practising pharmacists. The articles, presenting simple but rigorous guidance to encourage and support novice researchers, are being solicited from authors with appropriate expertise.
Previous articles in this series:
Bond CM. The research jigsaw: how to get started. Can J Hosp Pharm . 2014;67(1):28–30.
Tully MP. Research: articulating questions, generating hypotheses, and choosing study designs. Can J Hosp Pharm . 2014;67(1):31–4.
Loewen P. Ethical issues in pharmacy practice research: an introductory guide. Can J Hosp Pharm. 2014;67(2):133–7.
Tsuyuki RT. Designing pharmacy practice research trials. Can J Hosp Pharm . 2014;67(3):226–9.
Bresee LC. An introduction to developing surveys for pharmacy practice research. Can J Hosp Pharm . 2014;67(4):286–91.
Gamble JM. An introduction to the fundamentals of cohort and case–control studies. Can J Hosp Pharm . 2014;67(5):366–72.
Austin Z, Sutton J. Qualitative research: getting started. C an J Hosp Pharm . 2014;67(6):436–40.
Houle S. An introduction to the fundamentals of randomized controlled trials in pharmacy research. Can J Hosp Pharm . 2014; 68(1):28–32.
Charrois TL. Systematic reviews: What do you need to know to get started? Can J Hosp Pharm . 2014;68(2):144–8.
Competing interests: None declared.
Further Reading
Examples of qualitative research in pharmacy practice.
- Farrell B, Pottie K, Woodend K, Yao V, Dolovich L, Kennie N, et al. Shifts in expectations: evaluating physicians’ perceptions as pharmacists integrated into family practice. J Interprof Care. 2010; 24 (1):80–9. [ PubMed ] [ Google Scholar ]
- Gregory P, Austin Z. Postgraduation employment experiences of new pharmacists in Ontario in 2012–2013. Can Pharm J. 2014; 147 (5):290–9. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Marks PZ, Jennnings B, Farrell B, Kennie-Kaulbach N, Jorgenson D, Pearson-Sharpe J, et al. “I gained a skill and a change in attitude”: a case study describing how an online continuing professional education course for pharmacists supported achievement of its transfer to practice outcomes. Can J Univ Contin Educ. 2014; 40 (2):1–18. [ Google Scholar ]
- Nair KM, Dolovich L, Brazil K, Raina P. It’s all about relationships: a qualitative study of health researchers’ perspectives on interdisciplinary research. BMC Health Serv Res. 2008; 8 :110. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Pojskic N, MacKeigan L, Boon H, Austin Z. Initial perceptions of key stakeholders in Ontario regarding independent prescriptive authority for pharmacists. Res Soc Adm Pharm. 2014; 10 (2):341–54. [ PubMed ] [ Google Scholar ]
Qualitative Research in General
- Breakwell GM, Hammond S, Fife-Schaw C. Research methods in psychology. Thousand Oaks (CA): Sage Publications; 1995. [ Google Scholar ]
- Given LM. 100 questions (and answers) about qualitative research. Thousand Oaks (CA): Sage Publications; 2015. [ Google Scholar ]
- Miles B, Huberman AM. Qualitative data analysis. Thousand Oaks (CA): Sage Publications; 2009. [ Google Scholar ]
- Patton M. Qualitative research and evaluation methods. Thousand Oaks (CA): Sage Publications; 2002. [ Google Scholar ]
- Willig C. Introducing qualitative research in psychology. Buckingham (UK): Open University Press; 2001. [ Google Scholar ]
Group Dynamics in Focus Groups
- Farnsworth J, Boon B. Analysing group dynamics within the focus group. Qual Res. 2010; 10 (5):605–24. [ Google Scholar ]
Social Constructivism
- Social constructivism. Berkeley (CA): University of California, Berkeley, Berkeley Graduate Division, Graduate Student Instruction Teaching & Resource Center; [cited 2015 June 4]. Available from: http://gsi.berkeley.edu/gsi-guide-contents/learning-theory-research/social-constructivism/ [ Google Scholar ]
Mixed Methods
- Creswell J. Research design: qualitative, quantitative, and mixed methods approaches. Thousand Oaks (CA): Sage Publications; 2009. [ Google Scholar ]
Collecting Qualitative Data
- Arksey H, Knight P. Interviewing for social scientists: an introductory resource with examples. Thousand Oaks (CA): Sage Publications; 1999. [ Google Scholar ]
- Guest G, Namey EE, Mitchel ML. Collecting qualitative data: a field manual for applied research. Thousand Oaks (CA): Sage Publications; 2013. [ Google Scholar ]
Constructivist Grounded Theory
- Charmaz K. Grounded theory: objectivist and constructivist methods. In: Denzin N, Lincoln Y, editors. Handbook of qualitative research. 2nd ed. Thousand Oaks (CA): Sage Publications; 2000. pp. 509–35. [ Google Scholar ]

Research Design and Methods
Acknowledgments, a qualitative study of barriers to medication-taking among people with type 2 diabetes using the theoretical domains framework.
- Split-Screen
- Article contents
- Figures & tables
- Supplementary Data
- Peer Review
- Open the PDF for in another window
- Cite Icon Cite
- Get Permissions
Michael Vallis , Susie Jin , Agnieszka Klimek-Abercrombie , Ginnie Ng , Noah M. Ivers; A Qualitative Study of Barriers to Medication-Taking Among People With Type 2 Diabetes Using the Theoretical Domains Framework. Diabetes Spectr 15 August 2024; 37 (3): 264–272. https://doi.org/10.2337/ds23-0038
Download citation file:
- Ris (Zotero)
- Reference Manager
We aimed to better understand the challenges related to type 2 diabetes medication-taking through Theoretical Domains Framework (TDF)-guided interviews with people with type 2 diabetes with varying degrees of medication-taking.
One-on-one qualitative interviews following a semistructured discussion guide informed by the TDF were conducted. Thirty people with type 2 diabetes in Canada were interviewed, with representation from across the country, of both sexes (47% female), of people with various diabetes durations (mean 12.9 ± 7.9 years), with different types of medication plans ( n = 15 on polypharmacy), and with various medication-taking levels ( n = 10 each for low-, medium-, and high-engagement groups).
Themes related to medication-taking from interviews mapped to 12 of the 14 TDF theme domains, with the exclusion of the knowledge and skills domains. The most prominent domains, as determined by high-frequency themes or themes for which people with low and high medication-taking had contrasting perspectives, were 1 ) emotion; 2 ) memory, attention, and decision processes; 3 ) behavioral regulation; 4 ) beliefs about consequences; 5 ) goals; and 6 ) environmental context and resources.
Through our interviews, several areas of focus emerged that may help efforts to increase medication-taking. To validate these findings, future quantitative research is warranted to help support people with type 2 diabetes in overcoming psychological and behavioral barriers to medication-taking.
Approximately 3.4 million Canadians were estimated to be living with diabetes in 2015, corresponding to 9.3% of the total Canadian population, and this figure is predicted to rise to 12.1% of the population by 2025 ( 1 ). Nine out of 10 people living with diabetes in Canada have type 2 diabetes ( 2 ).
The management of type 2 diabetes is multifaceted, and treatment guidelines recommend behavior change interventions focusing on healthy eating and increased physical activity, concurrent cardiorenal protective medications, and the initiation of glucose-lowering agents as needed ( 3 ). Low levels of medication-taking can contribute to unstable glucose levels ( 3 ) and has been associated with increased risk of type 2 diabetes–related complications, hospitalizations, and mortality ( 4 , 5 ). Unstable glycemic outcomes resulting from low medication-taking have also been associated with increases in health care resource utilization and costs (e.g., for outpatient care, emergency room visits, hospitalizations, and managing diabetes-related complications) ( 4 ). It is estimated that approximately half of Canadian adults with type 2 diabetes have unstable glucose levels (i.e., an A1C ≥7.0% [53 mmol/mol]) ( 6 ).
Medication-taking is defined as the extent to which people engage with their prescribed medication dosing plan ( 7 ). In its 2003 report on medication-taking, the World Health Organization stated that increasing medication-taking engagement may have a far greater impact on the health of the population than making changes to specific medical treatments ( 8 ). A systematic review on medication-taking for people with type 2 diabetes found engagement rates ranging from 36 to 93% for oral hypoglycemic agents and from 62 to 64% for insulin, with variations resulting, in part, from the variety of measurement methods used ( 9 ).
Several reviews published in recent years have examined the body of literature on medication-taking in relation to people with type 2 diabetes and established many factors affecting medication-taking engagement, including age, race, complexity of dosing plans, safety and tolerability of medications, perceptions of risks and benefits of medications (including injection phobia and concerns about long-term use), costs and economic considerations, insulin use, health literacy, and patient-provider interactions ( 4 , 10–13 ). Previous efforts to increase medication-taking have focused on reducing treatment complexity through fixed-dose combination pills and less frequent dosing plans, identifying health beliefs regarding acceptance of medication, and behavioral habit formation regarding medication-taking behavior ( 4 , 13 ). In 2015, Sapkota et al. ( 14 ) published a systematic review exploring 52 studies that addressed engagement with blood glucose–lowering medications in people with type 2 diabetes. They found that no single type of intervention increased medication-taking consistently and that increments in improved engagement were achieved by most strategies in one or more of the outcomes assessed. Another 2015 review by Sapkota et al. ( 15 ) found that interventions addressing several factors contributing to low medication-taking were comparatively more effective in improving medication-taking and meeting glycemic targets in people with type 2 diabetes than interventions addressing single factors (educational, behavioral, or economic). Yet, it was also observed that educational strategies were the most common intervention type, followed by behavior change strategies ( 15 ). A review summarizing the challenges associated with diabetes self-management similarly identified targets of intervention across three domains: 1 ) knowledge, beliefs, and related cognitive constructs; 2 ) emotional distress and well-being; and 3 ) behavioral skills and coping ( 16 ). As part of their conclusion, the authors accentuated the need for continued research on the psychosocial aspects of living with diabetes and, specifically, the context underlying self-management behaviors ( 16 ). More recently, a study interviewing people with type 2 diabetes in the United States identified some psychosocial factors that influence the way people take medications, including their belief in medicine, and emphasized the need to understand individuals’ life experiences and behaviors to increase their engagement with medication-taking ( 17 ).
Identifying factors related to not taking medications through the use of behavior change theories and frameworks is imperative to develop credible scientific evidence and inform evidence-based interventions. The Theoretical Domains Framework (TDF) is a comprehensive and commonly used theory-informed approach to guide qualitative data collection to better understand barriers and facilitators to behavior change ( 18 ). The TDF summarizes 33 theories of behavior change in the 14 theoretical domains listed in Table 1 . This framework has been applied in the design of different interview studies (e.g., to increase the understanding of medication-taking behaviors among patients and decision-making in health care professionals [ 19 , 20 ]). Taken together, research to date showcases that factors contributing to medication-taking are multifaceted and highlights a gap in understanding the behavioral influences of medication-taking, especially among people with type 2 diabetes, in Canada.
TDF Domains and Definitions (18)
| Domain . | Definition . |
|---|---|
| Knowledge | An awareness of the existence of something |
| Skills | An ability or proficiency acquired through practice |
| Social/professional role and identity | A coherent set of behaviors and displayed personal qualities of an individual in a social or work setting |
| Beliefs about capabilities | Acceptance of the truth, reality, or validity about an ability, talent, or facility that a person can put to constructive use |
| Optimism | The confidence that things will happen for the best or that desired goals will be attained |
| Beliefs about consequences | Acceptance of the truth, reality, or validity about outcomes of a behavior in a given situation |
| Reinforcement | Increasing the probability of a response by arranging a dependent relationship, or contingency, between the response and a given stimulus |
| Intentions | A conscious decision to perform a behavior or a resolve to act in a certain way |
| Goals | Mental representations of outcomes or end states that an individual wants to achieve |
| Memory, attention, and decision processes | The ability to retain information, focus selectively on aspects of the environment, and choose between two or more alternatives |
| Environmental context and resources | Any circumstance of a person’s situation or environment that discourages or encourages the development of skills and abilities, independence, social competence, and adaptive behavior |
| Social influences | Those interpersonal processes that can cause individuals to change their thoughts, feelings, or behaviors |
| Emotion | A complex reaction pattern involving experiential, behavioral, and physiological elements, by which the individual attempts to deal with a personally significant matter or event |
| Behavioral regulation | Anything aimed at managing or changing objectively observed or measured actions |
| Domain . | Definition . |
|---|---|
| Knowledge | An awareness of the existence of something |
| Skills | An ability or proficiency acquired through practice |
| Social/professional role and identity | A coherent set of behaviors and displayed personal qualities of an individual in a social or work setting |
| Beliefs about capabilities | Acceptance of the truth, reality, or validity about an ability, talent, or facility that a person can put to constructive use |
| Optimism | The confidence that things will happen for the best or that desired goals will be attained |
| Beliefs about consequences | Acceptance of the truth, reality, or validity about outcomes of a behavior in a given situation |
| Reinforcement | Increasing the probability of a response by arranging a dependent relationship, or contingency, between the response and a given stimulus |
| Intentions | A conscious decision to perform a behavior or a resolve to act in a certain way |
| Goals | Mental representations of outcomes or end states that an individual wants to achieve |
| Memory, attention, and decision processes | The ability to retain information, focus selectively on aspects of the environment, and choose between two or more alternatives |
| Environmental context and resources | Any circumstance of a person’s situation or environment that discourages or encourages the development of skills and abilities, independence, social competence, and adaptive behavior |
| Social influences | Those interpersonal processes that can cause individuals to change their thoughts, feelings, or behaviors |
| Emotion | A complex reaction pattern involving experiential, behavioral, and physiological elements, by which the individual attempts to deal with a personally significant matter or event |
| Behavioral regulation | Anything aimed at managing or changing objectively observed or measured actions |
In this study, we aimed to 1 ) identify barriers to medication-taking faced by people with type 2 diabetes and 2 ) explore potential strategies to address these barriers. Particularly, we believed that using the TDF to guide the conduct and analysis of semi-structured one-on-one telephone interviews with people with type 2 diabetes in Canada would help to elucidate the situational context and behavioral influences that contribute to medication-taking.
Participants
Thirty individuals were recruited for 45-minute one-on-one interviews. Participants were recruited from a general sample of Canadians who have previously consented to be contacted to complete interviews for research. These people were initially recruited through referrals, social media, advertising, and patient advocacy groups. To participate in this study, individuals were required to have been living with type 2 diabetes for at least 1 year (i.e., since 2020) and to be currently on medication(s) to manage their type 2 diabetes. Quota sampling was used to ensure that the distribution of individuals included representation from various geographical regions in Canada, different medication types, and different levels of medication-taking.
Study Design
An interview discussion guide was developed, informed by the results from our previous scoping review ( 21 ), inputs from a national steering committee of clinical experts in diabetes, prior TDF medication-taking research ( 19 ), and the TDF ( 18 ) ( Supplementary Material ). The discussion guide included background questions on participants’ history with type 2 diabetes and medications and questions exploring issues related to medication-taking for each of the 14 TDF domains. One-on-one, 45-minute telephone interviews were conducted from 8 July to 12 August 2021 in English or French by one of three interviewers. After receiving consent from participants, the interviews were digitally recorded, and themes (barriers) were identified for analysis. Participants were classified by sex, geographical region (Atlantic Provinces, Quebec, Ontario, or Western Provinces), years since type 2 diabetes diagnosis, types of medication(s) (metformin only, oral medications other than or in addition to metformin, insulin with or without metformin or other oral medications, or noninsulin injectable medications with or without metformin, other oral medications, or insulin), and levels of medication-taking (low, medium, or high).
Levels of medication-taking were determined based on answers to the Morisky, Green, Levine Medication Adherence Questionnaire (MGL-MAQ) ( 22 ). The MGL-MAQ is a concise, reliable, and validated self-reported scale to measure medication-taking behaviors in people with chronic diseases ( 23 , – 25 ). This four-item scale includes yes/no questions, which are framed in reverse wording to overcome yes-saying bias. These items are 1 ) Do you ever forget to take your medicine? 2 ) Are you careless at times about taking your medicine? 3 ) When you feel better, do you sometimes stop taking your medicine? and 4 ) Sometimes if you feel worse when you take the medicine, do you stop taking it? Individuals who answered “No” to all questions were considered to have high engagement, those who answered “Yes” to one or two questions were considered to have medium engagement, and those who answered “Yes” to three or four questions were considered to have low engagement.
An inductive approach was used to analyze the qualitative data collected from the interviews ( 18 ). Transcripts from all interviews were reviewed and condensed into brief summaries and further summarized into overarching belief statements (i.e., themes) by two reviewers (G.N. and Andrean Bunko of IQVIA Solutions Canada, Inc.) ( 26 ). The themes were then mapped to the best fitting of the 14 TDF domains listed in Table 1 by a single reviewer (G.N.). The mapping of themes into domains was interrogated and confirmed by a second reviewer (Andrean Bunko) ( 18 ) and subsequently reviewed by all authors. Domains were considered relevant if they had a high frequency (i.e., including several belief statements mentioned by multiple respondents) or included belief statements that contrasted between respondents who were considered high versus low engagement.
Participant Characteristics
The demographic characteristics of the 30 people with type 2 diabetes interviewed for this study are presented in Table 2 . Of the included individuals, 14 (47%) were female, with the largest proportion from Ontario (43%) and smallest proportion from the Atlantic Provinces (7%). The average time since diagnosis of type 2 diabetes for all individuals was 12.9 ± 7.9 years (range 1–30 years), and half (50%) of the individuals reported currently taking more than one medication to manage their type 2 diabetes. Among all participants, 9 (30%) were taking metformin only, 9 (30%) were taking oral medication(s) other than or in addition to metformin, 3 (10%) were taking insulin with or without metformin or another oral medications, and 9 (30%) were taking a noninsulin injectable medication with or without metformin, other oral medication(s), or insulin. In all, 10 individuals each were classified into the low, medium, and high medication-taking categories. Table 2 presents the participant characteristics stratified by level of medication-taking.
Demographics of Study Participants
| Characteristic . | Total ( = 30) . | Low Engagement ( = 10) . | Medium Engagement ( = 10) . | High Engagement ( = 10) . |
|---|---|---|---|---|
| Female sex | 14 (47) | 5 (50) | 3 (30) | 6 (60) |
| Geographic region | ||||
| Ontario | 13 (43) | 5 (50) | 5 (50) | 3 (30) |
| Quebec | 8 (27) | 3 (30) | 1 (10) | 4 (40) |
| Western Provinces | 7 (23) | 2 (20) | 2 (20) | 3 (30) |
| Atlantic Provinces | 2 (7) | 0 (0) | 2 (20) | 0 (0) |
| Years since type 2 diabetes diagnosis, mean ± SD (range) | 12.9 ± 7.9 (1–30) | 9.2 ± 7.2 (1–22) | 11.3 ± 7.4 (2–21) | 18.1 ± 6.9 (5–30) |
| Takes more than one diabetes medication | 15 (50) | 4 (40) | 5 (50) | 6 (60) |
| Medications | ||||
| Metformin only | 9 (30) | 4 (40) | 3 (30) | 2 (20) |
| Oral medication(s) other than or in addition to metformin | 9 (30) | 3 (30) | 3 (30) | 3 (30) |
| Insulin with or without metformin or other oral medication(s) | 3 (10) | 0 (0) | 2 (20) | 1 (10) |
| Noninsulin injectable with or without metformin or other oral medication(s) or insulin | 9 (30) | 3 (30) | 2 (20) | 4 (40) |
| Characteristic . | Total ( = 30) . | Low Engagement ( = 10) . | Medium Engagement ( = 10) . | High Engagement ( = 10) . |
|---|---|---|---|---|
| Female sex | 14 (47) | 5 (50) | 3 (30) | 6 (60) |
| Geographic region | ||||
| Ontario | 13 (43) | 5 (50) | 5 (50) | 3 (30) |
| Quebec | 8 (27) | 3 (30) | 1 (10) | 4 (40) |
| Western Provinces | 7 (23) | 2 (20) | 2 (20) | 3 (30) |
| Atlantic Provinces | 2 (7) | 0 (0) | 2 (20) | 0 (0) |
| Years since type 2 diabetes diagnosis, mean ± SD (range) | 12.9 ± 7.9 (1–30) | 9.2 ± 7.2 (1–22) | 11.3 ± 7.4 (2–21) | 18.1 ± 6.9 (5–30) |
| Takes more than one diabetes medication | 15 (50) | 4 (40) | 5 (50) | 6 (60) |
| Medications | ||||
| Metformin only | 9 (30) | 4 (40) | 3 (30) | 2 (20) |
| Oral medication(s) other than or in addition to metformin | 9 (30) | 3 (30) | 3 (30) | 3 (30) |
| Insulin with or without metformin or other oral medication(s) | 3 (10) | 0 (0) | 2 (20) | 1 (10) |
| Noninsulin injectable with or without metformin or other oral medication(s) or insulin | 9 (30) | 3 (30) | 2 (20) | 4 (40) |
Data are n (%) except where noted.
Key Medication-Taking Themes by Theme Domain
Forty-two themes were identified and mapped to 12 of the 14 theme domains, with the knowledge and skills domains being the exceptions. The most prominent domains, as determined by high-frequency themes or themes for which people with low and high medication-taking had contrasting perspectives were emotion (2 themes); memory, attention, and decision processes (6 themes); behavioral regulation (5 themes); beliefs about consequences (8 themes); goals (3 themes); and environmental context and resources (4 themes). The key themes from the most prominent domains are discussed below, along with illustrative quotes captured during the interviews. A list of all of the identified themes is provided in Supplementary Table S1 .
Primary themes by theme domain
“Because I take so many medications—at times, I just get frustrated with it—that I’m so ill all the time and I get an attitude like, ‘Oh, well. Who cares? I’m going to do what I want, eat what I want. Sort of like a lack of total awareness of what happens when I don’t take the medicine. I’m just in a mood that I don’t care . . . . I am very tired of taking loads of pills every day after many years.” (low engagement)
“The fact that I have to take it at all—no, doesn’t make me feel good at all—makes me feel like a total failure. Diabetes was pretty much my own fault because of the huge weight gain I had when I was a kid. . . . but it’s my own fault.” (low engagement)
“For me, it is about accountability and being more responsible in my day-to-day life for how I’m feeling and how I’m doing. Medication is a huge part of that. Being responsible and organized and taking the medication is definitely a part of who I am now. It’s a normal, everyday occurrence, but it’s something I have to do.” (high engagement)
Memory, attention, and decision processes
“I would typically travel every 6 months . . . and, absolutely, sometimes I will forget to take the medication with me. Or, just because you’re traveling—you’re outside with family and friends—you’re not able to stick to your regimen. You forget it.” (low engagement)
“All of my medications are very stable and portable, so whenever I have traveled, it’s easy to take everything with me.” (high engagement)
“If work is too busy, I tend to skip one of my doses. Usually there will be social events with friends or long trips or a busy work schedule—they are the activities that interfere with my medication . . . . I skip the doses.” (low engagement)
“I babysit my granddaughter, and she takes up a lot of my time. I forget for a while, but I always take it after.” (high engagement)
“The most important is eating healthy because even if you don’t exercise and you don’t take your medication, if you’re eating healthy, you’re still going to be able to control your sugar levels.” (low engagement)
“Sometimes, I just wake up the next day and go, ‘Oh, I didn’t take the meds last night, did I?’” (low engagement)
Behavioral regulation
“I made everything habit. Once it’s habit, it becomes automatic—you just do it.” (medium engagement)
“I take it in the morning and in the evening—it’s routine. I do go to bed every evening, so I know that I have to take my medication. And in the morning, when I wake up, I take the other medications.” (high engagement)
“I could set alarms on my phone, but I don’t. I don’t use anything, no.” (low engagement)
“[I have] notifications on the phone, blister packs, and . . . Post-It notes and having my family around me, who ask, ‘Did you remember to take this? Did you remember to do that?’” (high engagement)
“Meeting with friends, or any kind of social events, or if I go on long trips—these are activities that are interfering with my medications.” (low engagement)
“It’s a timing thing. It [medication] causes gurgling and gas and all kinds of nastiness, so if I’m going to a restaurant, I would’ve thought ‘I’m going to wait until I’m done with the restaurant to take the pill’ . . . steering around my schedule a little bit. I try not to miss, but if I miss, I wait until the next time. I don’t double dose.” (low engagement)
“In the morning, if my routine is broken, sometimes I can forget, but it’s rare. Almost like a step-by-step thing in the morning. So, I do A, B, C . . . D is taking the pills.” (high engagement)
Beliefs about consequences
“Medications come with their own side effects, and they ruin other things, like your liver and kidney and whatever.” (low engagement)
“I was told by the doctor to take [my medication] 3 times a day. At first, I really wanted to follow those instructions from the doctor, but then I would get side effects, like I would have diarrhea and metallic taste . . .. It’s not practical. It’s very uncomfortable . . . . I would only take the medicine if I’m staying at home . . . but if I’m working or doing errands outside . . . I stop taking it.” (low engagement)
“I wish there was just one magic pill or one magic injection that will take care of all of it.” (low engagement)
“This medication is helping me. I’m not going to stop taking it. I’m a much happier person. I’m a much better person to my family and everyone as long as I take my medication.” (high engagement)
Environmental context and resources
“The cost is getting to be huge for diabetics. Right now, when you haven’t been working for a year . . . someone on a fixed income . . . and it’s been going up in price—let’s say an average of $170–180 every 2 months for your supplies. And that’s not including your test strips. That is a lot of money.” (medium engagement)
“Initially, I was scared of the drug because I have read [online] that metformin usually causes Alzheimer’s, but later on, when I kept searching, I said, ‘Okay, it’s safe.’ But still, I’m having my concerns about how metformin side effects are going to be in the long term. I was concerned taking it long term.” (low engagement)
Through qualitative interviews, we identified themes and barriers affecting medication-taking among people with type 2 diabetes in Canada using the TDF, with six prominent theme domains identified. These domains were 1 ) emotion; 2 ) memory, attention, and decision processes; 3 ) behavioral regulation; 4 ) beliefs about consequences; 5 ) goals; and 6 ) environmental context and resources. To our knowledge, this is the first study to apply the TDF to qualitative one-on-one interviews with people with type 2 diabetes to provide a greater understanding of the situational context and drivers of medication-taking behavior.
The results from our study extend those of prior studies in several ways, providing further context for the ways in which complex dosing plans, tolerability of medications, and perceptions of risks and benefits of medications ( 13 ) contribute to engagement. These findings also present additional context for how both unintentional nonengagement behaviors, such as forgetfulness and beliefs about the need for medication, and intentional nonengagement behaviors contribute to taking medication as recommended ( 27 , 28 ).
Polonsky and Henry ( 4 ) emphasized that innovative strategies to improve the attitudes of people with type 2 diabetes toward medication or encourage habit formation regarding medication-taking may help increase engagement. Our study used the TDF to identify specific intentional and unintentional behavioral influences on medication nonengagement that were common among people with low medication-taking. This information was further enriched by contrasting the attitudes and behaviors of people with higher medication-taking to derive recommended areas of change. By leveraging findings from our approach, strategies to increase medication-taking can be tailored to target specific behavioral influences and contextual challenges.
Our results can be applied to the development and application of effective interventions to increase medication-taking, and these should take into account the broader TDF theme domains we identified, as well as target specific themes within each domain ( 29 ). For example, people with type 2 diabetes with low engagement experience emotional challenges associated with being overwhelmed by living with diabetes and needing to take medication. Therefore, diabetes self-management support interventions might benefit from incorporating emotion management strategies for people who struggle with diabetes and treatment acceptance ( Supplementary Table S1 , theme 13.1). Diabetes self-management education that provides concise and reliable diabetes and medication knowledge might mitigate issues associated with individuals’ past experiences of having unreliable information sources ( Supplementary Table S1 , theme 11.3) and also address feelings about medication-taking ( Supplementary Table S1 , theme 6.2). The individuals expressed a desire for information that is simple and easily accessible, supports setting expectations regarding side effects, and increases their understanding of the long-term benefits of treatment. Furthermore, we suggest that improving access to resources such as diabetes clinics, pharmacists, and support groups where people with type 2 diabetes can ask questions, receive coaching, and get essential education and training would be useful. These strategies would provide an opportunity to reframe negative feelings about diabetes and medications ( Supplementary Table S1 , theme 13.2). These approaches could also specifically target identified themes such as the importance of long-term goal setting ( Supplementary Table S1 , theme 9.3) and understanding the role of medications in type 2 diabetes treatment and the progressive nature of the disease (i.e., that it usually requires long-term medication use) ( Supplementary Table S1 , themes 9.2 and 10.3).
Likewise, practical support that facilitates medication-taking and increases accessibility may help to increase engagement. It was apparent in this study that disruptions to routines affected individuals who reported low engagement more than those with medium or high engagement, resulting in unintentionally or intentionally forgetting, skipping, or delaying medication doses ( Supplementary Table S1 , themes 10.1, 10.2, and 14.5). This finding highlights an important consideration when selecting medication plans, which should incorporate individuals’ convenience and lifestyle considerations to encourage engagement.
Our findings also highlight the importance of people with type 2 diabetes establishing a medication-taking routine. A number of participants reporting low engagement did not use reminders, whereas those with high engagement had a clear and defined process, including organization resources such as pill boxes and pharmacy-prepared personalized blister packs and tracking resources such as phone alarms, calendars, and diaries ( Supplementary Table S1 , theme 14.2). Thus, encouraging or enabling the use of such tools among people with type 2 diabetes may increase engagement, although success in using these tools may be more a result of individuals’ motivations and organizational skills rather than the mere availability of the tools.
There is also a role for pharmacist teams to identify people with or at risk for low engagement and to support these individuals through actions such as sending refill reminders, dispensing medication in personalized blister packs, and accommodating emergency medication needs, in addition to having conversations with and supporting people with type 2 diabetes in individualized appointments.
Finally, financial barriers such as high costs of medications and glucose monitoring supplies ( Supplementary Table S1 , theme 11.1) can be diminished through health system changes (specifically, covering diabetes-related care products and medications) and through support programs.
Limitations
There are limitations to our study that should be considered for proper interpretation of the results. First, although quota sampling was conducted to ensure diversity among the participants, little diversity was reflected in recruited participants. Therefore, our participants may not have been representative of all people with type 2 diabetes in Canada. Second, social desirability bias, in which respondents tend to provide answers that overreport desirable and underreport less desirable attributes is a known concern with interviews. However, this problem was mitigated by our comprehensive discussion guide, which approached the topic of medication-taking from many perspectives, as well as by contrasting the responses of people with lower and higher medication-taking engagement. Third, although generating data using the TDF has been shown to be a comprehensive and inclusive approach to exploratory research, it is still a descriptive framework rather than a theory ( 30 ). The results generated do not specify relationships between the domains and do not generate testable hypotheses ( 31 ). Finally, we recognize that qualitative interviews about behavioral influences represent the perceptions of the individuals interviewed and may not reflect the actual causes of their behaviors or be generalizable to broader populations ( 30 ).
Our study identified behavioral influences contributing to lower medication-taking engagement, highlighting key areas for change among a small group of people with type 2 diabetes. Future research is warranted to validate these findings within a larger sample and to explore the suitability of various intervention and implementation options to improve medication-taking ( 32 ). Additionally, the results of this study can inform the development of medical education and training programs for health care providers supporting people with type 2 diabetes in Canada.
This article contains supplementary material online at https://doi.org/10.2337/figshare.25270123 .
The authors thank the people with type 2 diabetes who took part in qualitative interviews for this study. They also thank Carole Hamersky, PhD, and Cory Gamble, MD, of Novo Nordisk Canada, Inc., for expert advice on developing the qualitative discussion guide. The authors thank the following individuals from Real World Solutions, IQVIA Solutions Canada, Inc.: Andrean Bunko, MSc, and Pierre-François Meyer, PhD, for their support conducting interviews, coding, and reviewing theme domain mapping; Atif Kukaswadia, PhD, and Calum S. Neish, PhD, for their guidance and oversight throughout the project; and Vibha Dhamija, MSc, and Saurabh Trikha, PhD, for their assistance with medical writing.
This study was funded by Novo Nordisk Canada, Inc.
Duality of Interest
M.V., S.J., and N.M.I. received advisory fees from IQVIA Solutions Canada, Inc., for contributions to the design and interpretation of the study but received no compensation for development of the manuscript. A.K.-A. is an employee of Novo Nordisk Canada, Inc. G.N. is a consultant employed by IQVIA Solutions Canada, Inc., to support this study sponsored by Novo Nordisk Canada, Inc. No other potential conflicts of interest relevant to this article were reported.
Author Contributions
All authors reviewed the TDF mapping results and participated in the interpretation of results, reviewed and revised the manuscript, and approved the final manuscript. G.N. conducted the interviews, coded the themes, and conducted the theme domain mapping. G.N. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Prior Presentation
Parts of this article were presented in abstract form at the American Diabetes Association’s 82nd Scientific Sessions, 3–7 June 2022.
Email alerts
- Online ISSN 1944-7353
- Print ISSN 1040-9165
- Diabetes Care
- Clinical Diabetes
- Diabetes Spectrum
- Standards of Medical Care in Diabetes
- Scientific Sessions Abstracts
- BMJ Open Diabetes Research & Care
- ShopDiabetes.org
- ADA Professional Books
Clinical Compendia
- Clinical Compendia Home
- Latest News
- DiabetesPro SmartBrief
- Special Collections
- DiabetesPro®
- Diabetes Food Hub™
- Insulin Affordability
- Know Diabetes By Heart™
- About the ADA
- Journal Policies
- For Reviewers
- Advertising in ADA Journals
- Reprints and Permission for Reuse
- Copyright Notice/Public Access Policy
- ADA Professional Membership
- ADA Member Directory
- Diabetes.org
- X (Twitter)
- Cookie Policy
- Accessibility
- Terms & Conditions
- Get Adobe Acrobat Reader
- © Copyright American Diabetes Association
This Feature Is Available To Subscribers Only
Sign In or Create an Account
This paper is in the following e-collection/theme issue:
Published on 21.8.2024 in Vol 26 (2024)
Comparing GPT-4 and Human Researchers in Health Care Data Analysis: Qualitative Description Study
Authors of this article:

Kevin Danis Li 1, 2 , BS ; Adrian M Fernandez 1 , MD ; Rachel Schwartz 3, 4 , PhD ; Natalie Rios 1 , BS ; Marvin Nathaniel Carlisle 1 , BS ; Gregory M Amend 5 , MD ; Hiren V Patel 1 , MD, PhD ; Benjamin N Breyer 1, 2 , MAS, MD
1 Department of Urology, University of California San Francisco, San Francisco, CA, United States
2 Department of Epidemiology and Biostatistics, University of California San Francisco, San Francisco, CA, United States
3 Department of Anesthesia and Perioperative Care, University of California San Francisco, San Francisco, CA, United States
4 Division of General Internal Medicine, Department of Medicine, University of California San Francisco, San Francisco, CA, United States
5 Department of Urology, Icahn School of Medicine at Mount Sinai, New York, NY, United States
Corresponding Author:
- Kevin Danis Li , BS
- Department of Urology
- University of California San Francisco
- 400 Parnassus Ave
- San Francisco , CA
- United States
- Phone: 1 415-353-2200
- Email: [email protected]
- Open access
- Published: 22 August 2024
Factors influencing fidelity to guideline implementation strategies for improving pain care at cancer centres: a qualitative sub-study of the Stop Cancer PAIN Trial
- Tim Luckett 1 ,
- Jane Phillips 2 ,
- Meera Agar 1 , 3 ,
- Linda Richards 4 ,
- Najwa Reynolds 5 ,
- Maja Garcia 1 ,
- Patricia Davidson 6 ,
- Tim Shaw 7 ,
- David Currow 6 ,
- Frances Boyle 8 , 9 ,
- Lawrence Lam 10 ,
- Nikki McCaffrey 11 &
- Melanie Lovell 5 , 9
BMC Health Services Research volume 24 , Article number: 969 ( 2024 ) Cite this article
27 Accesses
Metrics details
The Stop Cancer PAIN Trial was a phase III pragmatic stepped wedge cluster randomised controlled trial which compared effectiveness of screening and guidelines with or without implementation strategies for improving pain in adults with cancer attending six Australian outpatient comprehensive cancer centres ( n = 688). A system for pain screening was introduced before observation of a ‘control’ phase. Implementation strategies introduced in the ‘intervention’ phase included: (1) audit of adherence to guideline recommendations, with feedback to clinical teams; (2) health professional education via an email-administered ‘spaced education’ module; and (3) a patient education booklet and self-management resource. Selection of strategies was informed by the Capability, Opportunity and Motivation Behaviour (COM-B) Model (Michie et al., 2011) and evidence for each strategy’s stand-alone effectiveness. A consultant physician at each centre supported the intervention as a ‘clinical champion’. However, fidelity to the intervention was limited, and the Trial did not demonstrate effectiveness. This paper reports a sub-study of the Trial which aimed to identify factors inhibiting or enabling fidelity to inform future guideline implementation initiatives.
The qualitative sub-study enabled in-depth exploration of factors from the perspectives of personnel at each centre. Clinical champions, clinicians and clinic receptionists were invited to participate in semi-structured interviews. Analysis used a framework method and a largely deductive approach based on the COM-B Model.
Twenty-four people participated, including 15 physicians, 8 nurses and 1 clinic receptionist. Coding against the COM-B Model identified ‘capability’ to be the most influential component, with ‘opportunity’ and ‘motivation’ playing largely subsidiary roles. Findings suggest that fidelity could have been improved by: considering the readiness for change of each clinical setting; better articulating the intervention’s value proposition; defining clinician roles and responsibilities, addressing perceptions that pain care falls beyond oncology clinicians’ scopes of practice; integrating the intervention within existing systems and processes; promoting patient-clinician partnerships; investing in clinical champions among senior nursing and junior medical personnel, supported by medical leaders; and planning for slow incremental change rather than rapid uptake.
Conclusions
Future guideline implementation interventions may require a ‘meta-implementation’ approach based on complex systems theory to successfully integrate multiple strategies.
Trial registration
Registry: Australian New Zealand Clinical Trials Registry; number: ACTRN 12615000064505; data: https://www.anzctr.org.au/Trial/Registration/TrialReview.aspxid=367236&isReview=true .
Peer Review reports
Pain is a common and burdensome symptom in people with cancer [ 1 ]. Barriers to pain care occur at all ‘levels’, including the patient and family (e.g., misconceptions regarding opioids), clinician (e.g. lack of expertise), service (e.g. inadequate referral processes) and healthcare system (e.g. lack of coordination) [ 2 , 3 , 4 , 5 , 6 , 7 , 8 ]. A recent systematic review suggests that around 40% of cancer patients with pain may not receive adequate management [ 9 ]. Research has demonstrated that routine screening and implementation of evidence-based guidelines has potential to improve quality of cancer pain care and outcomes [ 10 , 11 , 12 , 13 , 14 ]. However, experience suggests that clinicians are unlikely to utilise screening results or follow guidelines unless these are supported by targeted strategies [ 15 , 16 ].
The Stop Cancer PAIN Trial (ACTRN 12615000064505) was a phase III pragmatic stepped wedge cluster randomised controlled trial conducted between 2014 and 2019 which compared the effectiveness of screening and guidelines with or without implementation strategies for improving pain in adults with cancer attending six outpatient comprehensive cancer centres in Australia ( n = 688) [ 17 , 18 ]. A pen/paper system to screen for pain using 0–10 numerical rating scales (NRS) for worst and average intensity over the past 24 h was introduced to each centre prior to observation of a ‘control’ phase, in which clinicians were also made aware of the Australian Cancer Pain Management in Adults guidelines [ 19 ]. At the beginning of the training phase, trial investigators presented at staff meetings on the importance of better managing pain and the rationale and evidence base for the intervention components. Implementation strategies (collectively termed the ‘intervention’) were then introduced in a ‘training’ phase and maintained during an ‘intervention’ phase as follows: (1) audit of adherence to key guideline recommendations [ 19 ] and feedback delivered to clinical teams in one or two cycles; (2) health professional education via a ‘Qstream’ email-administered ‘spaced education’ module [ 20 ]; and (3) a patient education booklet and self-management resource for completion together with a clinician that included goal setting, a pain diary and pain management plan [ 21 , 22 ]. Selection of these strategies was informed by the Capability, Opportunity and Motivation Behaviour (COM-B) Model of behaviour change [ 23 ], and evidence that each strategy had been separately effective for supporting guideline implementation for other health conditions. The intervention was supported at each centre by a consultant physician who agreed to be a ‘clinical champion’ [ 24 ].
As reported previously [ 18 ], the Stop Cancer PAIN Trial found no significant differences between the intervention and the control phases on the trial’s primary outcome - the proportion of patients with moderate-severe worst pain intensity who reported a 30% decrease at 1-week follow-up. Fidelity to the intervention was lower than anticipated and variable between centres: only 2/6 centres had two audit cycles rather than one; completion rates for the health professional spaced education varied from 12% to 74% between centres; and the proportion of patients reporting receipt of written information of any kind rose to an average of only 30% (20-44%) versus 22% (2-30%) in the control phase. Unexpectedly, secondary measures of mean, worst and average pain over a 4-week follow-up period improved by 0.5 standard deviation during control as well as intervention phases. However, the lack of a comparison group with no screening system made it difficult to conclude whether improvement in the control phase was due to effects from screening, a Hawthorne effect, or some other explanation.
The current paper reports a sub-study of the Stop Cancer PAIN Trial which aimed to identify factors influencing fidelity to the intervention that might warrant consideration by similar initiatives in the future.
The intervention, methods and results of the Stop Cancer PAIN trial have been described in previous open-access articles [ 17 , 18 ]. The sub-study used a qualitative approach with pragmatic orientation to enable in-depth exploration of factors influencing success from the perspectives of clinicians at each participating centre [ 25 ]. Clinician views canvassed at interview were considered the most efficient means of identifying barriers and enablers among complex contextual factors at each centre, including personnel’s knowledge, attitudes and beliefs towards pain care and the intervention.
The sub-study was approved by the Southwestern Sydney Local Health District Human Research Ethics Committee (HREC/14/LPOOL/479) as part of the overall trial. All participants gave written informed consent to participate.
Reporting adheres to the consolidated criteria for reporting qualitative research (COREQ) [ 26 ].
Participants
Participants were eligible if they were employed on a permanent basis either full- or part-time at a participating centre in a role that provided clinical care to cancer patients or patient-focused administrative support. The clinical champion at each centre was invited to participate by the research team. Other personnel were invited by means of email circulars and verbal invitations during meetings. Given the diverse range of roles at each centre, no limit was set on sample size to canvass as many perspectives as possible.
Data collection
Data were collected by means of semi-structured interviews conducted by one of two researchers, a female pharmacist with experience of medical education for pain management (LR), and a male social scientist with a doctorate (TL). Both interviewers had prior experience in qualitative research and knew some participants through their project roles.
Participants were fully aware of the study purpose before consenting. Interviews were conducted face-to-face or by telephone, with the participant and interviewer being the only people present. Interviews began with open questions about ‘what worked’ and ‘didn’t work’ across the intervention before focusing on each implementation strategy in more detail and important contextual factors at their centre (see Table 1 for a topic guide, which was developed specifically for this study). Interviewers explicitly invited criticism, expressing a tone of open enquiry and neutrality throughout. Prompts were used as necessary to explore factors identified by participants in more detail. Factors identified at previous interviews were raised at subsequent ones for verification, inviting participants to disagree or agree as they felt appropriate. No requests were received to return transcripts to participants for comment. Interviews were audio-recorded and transcribed verbatim.
Analysis used the framework method [ 27 ] and a largely deductive approach based on the same theoretical framework used during intervention design - the COM-B Model [ 23 ]. Based on a systematic review, the COM-B Model posits that behaviour change requires three conditions, namely ‘capability’ (including both psychological and physical capacity), ‘opportunity’ (all the factors that lie outside the individual that make the behaviour possible or prompt it) and ‘motivation’ (including habitual processes, emotional responding, as well as analytical decision-making). Initial line-by-line coding categorized data against these conditions according to which best described relationships between factors and behaviours within and across implementation strategies and the levels of patient, clinician and centre. While the COM-B model originally focused directly on human behaviour, it became clear during coding that behaviour was substantially influenced by centre, specialty and disciplinary factors, so these were also considered appropriate foci for coding against COM conditions. To enhance credibility, the same data were coded in different ways where multiple interpretations seemed plausible until coding of further interviews identified consistencies to help with disambiguation. Charting of codes for data within and between centres enabled mapping between the relative contributions made by each condition, summarised as lessons learned for guiding similar initiatives in the future. Dependability was increased by ensuring coding was conducted by two members of the research team (NR, MG) who had no previous involvement in the project but were experienced in qualitative research. Review and discussion with two team members who were involved in the project throughout (TL and ML) was intended to balance ‘outsider’ and ‘insider’ perspectives to guard against bias from preconceived interpretations whilst also referencing contextual understanding. Both Excel 2019 (Microsoft) and NVivo V12 (QSR) software were used to help manage different stages of the analytic process.
Twenty-four people participated across the six centres, ranging from one to six participants. Fifteen were physicians (of whom six were clinical champions), eight were nurses, and one was a clinic receptionist. This response rate ranged from 2 to 27% of eligible personnel at each centre. See Table 2 for a more detailed summary of participant roles at each centre. Interviews were a median of 20 min long, with an inter-quartile range of 13 to 28 min.
Capability, opportunity and motivation
Coding against the COM-B Model identified ‘capability’ to be the component having most influence over intervention success, with ‘opportunity’ and ‘motivation’ playing largely subsidiary roles.
Capabilities: Pertinent capabilities were reported to include: a pre-existing, centre-level culture of continuous improvement, communication pathways between senior management and other personnel, established roles and responsibilities for pain care among disciplines and specialties, systems and processes that could readily accommodate the intervention, and a culture of involving patients as partners in care. These capabilities influenced the degree to which personnel and patients had the opportunity and motivation to fully engage with the intervention.
Opportunity and motivation: These elements were most frequently discussed by participants in terms of ‘time’ that personnel could commit to pain care relative to other responsibilities. Clinical champions were perceived to play a critical role in supporting intervention success but were under-resourced at every centre and challenged by turnover in the role at two. In addition to more systemic drivers, individual personnel’s motivation was influenced by the degree to which they accepted the intervention’s value proposition at the outset and perceived this to be demonstrated over time.
Interactions between capability, opportunity and motivation are explored below in terms of their implications for similar future initiatives. Findings suggest that fidelity could have been improved by: considering the readiness for change of each clinical setting; better articulating the intervention’s value proposition; defining clinician roles and responsibilities, addressing perceptions that pain care falls beyond oncology clinicians’ scopes of practice; integrating the intervention within existing systems and processes; promoting patient-clinician partnerships; investing in clinical champions among senior nursing and junior medical personnel, supported by medical leaders; and planning for slow incremental change rather than rapid uptake.
Consider centres’ readiness for change
The degree to which centres had a pre-existing culture of continuous improvement was considered important in providing a fertile context for the intervention. At Centre 5, there was a consensus that change of any kind was difficult to instigate, even according to the head of department: “… because it’s new - because we’re so entrenched in our ways ” (C5P04 [Centre 5, participant 04] medical oncologist, head of department and clinical champion). At another, the complex centre-level nature of the intervention was perceived to pose particular challenges compared to oncology drug trials with which they were more familiar: “ we haven’t been a principal site [in a trial of this kind] previously and I think that’s sort of opened up some gaps in knowledge for us and some opportunities for learning in the future … what kind of support we’d need to come with that trial to help it be a success in this culture ” (C3P02 palliative care physician and clinical champion).
Articulate and deliver on the intervention’s value proposition
Interviews highlighted the importance of articulating the intervention’s value proposition to every member of the workforce and maintaining engagement by demonstrating benefits over time. At Centre 5, some participants perceived that the intervention had been imposed by management rather than generated from clinical priorities: “…senior staff say [to researchers] ‘come to our clinics, but we expect everyone else to do the work’ ” (C5P05 radiation oncologist). This was compounded by a perceived lack of communication about the project, which limited personnels’ opportunity to take a more active role even when they were motivated to do so: “ I would have facilitated [the intervention] … but I didn’t know about it ” (C5P01 nurse practitioner). Eliciting and maintaining engagement was said to be additionally challenged at this centre by high staff turnover, especially among junior medical officers on rotation: “ it was very accepted by the junior medical staff [but] I think, unfortunately, when there’s a relatively high turnover of staff … ” (C5P07 radiation oncology trainee). At two other centres, turnover among personnel required a transition in the role of clinical champion, interrupting support for the intervention while the new incumbents familiarised themselves with the role.
Across centres, participants reported reservations among some of their colleagues regarding the project’s fundamental premises, including the assumption that pain care needed improving at their centre (“ they actually felt this trial was a little bit insulting for their clinical skills. There was a bit of eye rolling and ‘of course we do that already!’ ” (C3P02 palliative care physician and clinical champion)) or that pain warranted a specific focus rather than symptoms more generally: “ I find it more useful when more than one symptom is targeted ” (C5P06 palliative care physician).
More specific criticism was also levelled at each of the intervention strategies as follows.
Pain screening
In the case of screening, two participants questioned the validity of a 0–10 numerical rating scale (NRS) for different reasons: “ sometimes getting the numbers breaks the flow of the narrative” (C6P04 medical oncologist); “they [patients] would say, ‘no, I’m not in pain but I have a lot of discomfort when I swallow’ - it was in the wording ” C5P02 registered nurse). Even one of the clinical champions felt that screening was redundant where pain was very severe: “ if someone is clearly in a pain crisis, you don’t need to be asking … you kind of know what number - they might tell you it’s 15 [out of 10] ” (C6P02 palliative care physician and clinical champion). Perceptions of the value of screening were also influenced by the degree to which it led to demonstrable improvements in pain care, which was undermined by problems with establishing an efficient process at some centres: “ I think I’ve still probably got stray [pain screening] forms on my desk ” (C3P06 palliative care physician). A lack of understanding among personnel and patients about how screening might lead to better pain outcomes was said to result in “ fatigue ” (C5P03 clinical nurse consultant [clinical nurse consultant]; C1P01 palliative care physician and clinical champion), manifest as a downward spiral of effort in, and value from, screening.
Audit and feedback
The audit and feedback strategy attracted limited attention from personnel at most centres: “ I don’t think that the audit and feedback were terribly noticeable ” (C4P01 medical oncologist and clinical champion). At the centre where only the palliative care department participated, one participant perceived baseline audit results to be acceptable and therefore demotivating for change: “[ the audit results showed] we were doing a good job even ahead of time … it did sort of make you think – ‘well where do we go from here?’ ” (C6P04 pain medicine physician). At another centre, motivation among personnel to improve on less favourable audit findings was perceived to depend on whether they prioritised pain care to start with: “ people have come up to me and said, ‘Gee, we really did very badly didn’t we?’ … but they’re not necessarily the people who don’t treat pain well - that’s the problem ” (C1P01 palliative care physician and clinical champion).
Spaced education for health professionals
Participants’ opinion on the value of the online spaced education depended on discipline and seniority, with nurses and junior medical officers reporting benefits “( it gave me a bit more confidence that I was on the right track” (C5P01 nurse practitioner)) but consultant physicians perceiving the knowledge level too “basic” (C6P04 pain medicine physician) or questioning advice from online spaced education that their responses were ‘wrong’: “…some of the multiple answers could have been equally valid” (C504 medical oncologist and clinical champion). Where consultants remained engaged, motivation was said to rely on cultivating “ competition” between colleagues (C602 palliative care physician and clinical champion). Inevitably, the voluntary nature of online spaced education also meant that only motivated personnel engaged to begin with.
Patient self-management resource
All participants who had used the patient self-management resource perceived at least some value. However, its use was limited by barriers relating to role and process considered below.
Define roles and responsibilities
Among the most commonly voiced barriers was a lack of clarity about which specialties and disciplines should be responsible for pain screening, patient education and management. This was usually described in terms of a ‘lack of time’ for pain care relative to other duties afforded greater priority within their scope of practice. Perspectives on roles and responsibilities are considered separately for each aspect of pain care as follows.
While most centres allocated the clinical task of pain screening to clinic receptionists, there was widespread reflection that this had been suboptimal. The only participating clinic receptionist felt that pain screening fell outside her area of responsibility: “but I’m an administrative person - I don’t have anything to do with pain management ” (C2P03 clinic receptionist). Clinician participants across disciplines similarly perceived that pain screening required clinical expertise to assist patients with reporting their pain and triage for urgent follow-up: “ you need somebody talking to the patients, rather than just handing the form, say ‘fill this in’ ” (C2P04 clinical nurse consultant). One centre that recognised this early on reallocated screening from an administrative to a nursing role, leading to substantial improvements in the completeness and quality of data: “ it made a big difference and certainly improved our ability to recognise people who had pain and allowed access for those people who were in severe pain to medications or at least an assessment … implementation through the clerical staff was not a long-term strategy ” (C1P01 palliative care physician and clinical champion).
Patient education
There was little consensus on which disciplines should be responsible for supporting patients to use the self-management resource, with medical personnel deferring to nurses and vice-versa. Role allocation was challenged by the diverse components within the resource, with each perceived to fall within a different scope of practice: “ pain is something I always do as an assessment … [but] … I’m not managing the pain … I’ll review and make recommendations and talk about the pain diaries and discussing their diary with their palliative care doctor or their general practitioner. And I would encourage that process. [But] I wouldn’t be the one that’s setting the goals on their daily activities and stuff ” (C5P01 nurse practitioner). Some oncology nursing roles were perceived to focus on chemo- or radiotherapy protocols to the exclusion of supportive care unless symptoms arose from, or impeded, treatment. Meanwhile, oncologists tended to interpret their role as solely focused on prescribing rather than also encompassing patient education: “ junior doctors only [have] 15 minutes to take a history and everything. [They] could enter in meds [into the patient resource] if everything else is done by someone else … part of me knows it’s [patient resource] important, but the other part of me - I just - when will I have time in my clinical practice to do it? ” (C5P05 radiation oncologist).
Pain management
Some oncologists viewed even pharmacological pain management as peripheral to their scope of practice when consultation time was short, prioritising cancer treatment instead. These participants viewed their role as limited to referring to palliative medicine or pain specialists, especially where pain was believed to have causes other than cancer: “ if the pain is a complex pain where the patient doesn’t have evidence of cancer, and it may be treatment-related, then in those scenarios we tend to divert to the chronic pain team ” (C5P07 radiation oncology advanced trainee). While participants from palliative care and pain medicine welcomed referrals for complex cases, they felt that oncologists sometimes referred for pain they could have easily managed themselves: “ what about some regular paracetamol? … These are things that you’d expect any junior doctors, never mind consultants [to have provided advice on] ” (C5P06 palliative care physician).
Integrate within existing systems and processes
Participants from several centres expressed a view that the intervention’s complex nature had proven overwhelming for systems and processes at their centres. At two centres, integration was especially challenged by broader infrastructure shifts and process failures that limited receptiveness to further changes. Participants at several centres emphasised the process-driven nature of oncology services and the challenge of changing established processes: “ they have got a pro forma that they use for chemo-immunotherapy review, and pain is not part of it, and that perhaps needs more of an organisational nuance … why doesn’t pain feature as a clinical outcome as part of the chemotherapy, immunotherapy review?” (C6P01 clinical nurse consultant). Participants emphasised the need to integrate pain care into existing processes to help personnel understand what was expected of them: “…nursing staff were getting them [screening forms] in the patient’s files and going, ‘what am I supposed to do with this?’ ” (C2P04 clinical nurse consultant). Moreover, centres’ focus on cancer treatment meant that pain care struggled to gain traction even when a process could be instituted: “ unless pain is the presenting complaint and is at the forefront it goes into those, sorts of, you know, the ‘other details’ ” (C5P06 palliative care physician). For the palliative care centre, where pain care was already prioritised, there were doubts about how the proposed process improved on those already in place: “ I generally ask pretty detailed questions about pain anyway [so don’t need patients to be screened in the waiting room] ” (C6P04 pain medicine physician).
Suggestions for better integrating the intervention included “in-building” (C3P04 medical oncologist) responsibility for the strategies within new staff roles or introducing the strategies gradually by means of a “ multistep process” (C5P04 medical oncologist, head of department and clinical champion). Features of two strategies were singled out as having positive potential for supporting existing processes of care. The patient resource was said to “ facilitate communication between the oncology teams and the palliative care team ” (C5P05 radiation oncologist) and serve as a “ visual cue ” (C3P02 medical oncologist) to cover educational topics that “ they might have otherwise forgotten ” (C2P01 palliative care physician and clinical champion). Participants also found the spaced education email administration, spacing and repetition “ easy to manage ” (C2P01 palliative care physician and clinical champion) within their daily routines.
Promote patient-clinician partnership on pain care
Several participants expressed surprise at the prevalence of moderate-severe pain in screening results, and acknowledged that this revealed under-reporting of pain in usual care. Under-reporting was perceived to stem partly from patient expectations that pain from cancer was “ normal ” (C4P03 nurse practitioner) and to be especially common in the context of certain generational or cultural attitudes towards pain and opioids (“ I certainly think there’s a cultural element but there’s also your elderly patients who you know have been through the war and they’re just used to coping with things and you just suck it up … it’s like a badge of honour to be able to say ‘I’m not one of these pill-takers ’” (C3P03 registered nurse [RN])) or when patients were concerned that reporting pain might reduce their fitness for anti-cancer treatment: “[ patients might think that] if I tell them honestly how crappy I am with other symptoms and pain and everything, then they might stop my chemo” (C3P02 palliative care physician). Several participants perceived that under-reporting was also due to patients taking an overly passive role in consultations: “[clinicians assume that] if the patient doesn’t bring it up, it’s not a problem for them and … then the patient [is] thinking ‘the doctor will only talk about important things that are important for me and I won’t mention it because obviously it’s not important’ ” (C3P02 palliative care physician and clinical champion).
The screening component of the intervention was considered to address under-reporting by “ normal[ising] ” pain care, thus encouraging disclosure. The patient resource was also considered helpful for building patient capability to partner with clinicians on pain management by “ encouraging self-efficacy ” (C2P01 palliative care physician and clinical champion) through the tools it provided and its positive message that “ you can get control of your pain ” (C3P02 palliative care physician and clinical champion). It was also perceived to help patients “ keep a record ” (C5P03 clinical nurse consultant) of breakthrough pain and analgesia to discuss in their consultation. However, some participants delineated patient groups who might be less able to use the resource, including those with lower educational levels who struggled to set goals and identify an ‘acceptable’ level of pain balanced against side-effects from pharmacological management. For these patients, it was suggested that too many resources could be overwhelming rather than supportive: “ it’s almost like, the more resources they have, the less resourced there are ” (C5P06 RN). At one centre with an especially diverse demographic, patients were said to require substantial support even to understand the purpose and process of pain screening: “ most [patients] look at you going ‘oh, do I have to do anything?’ … They don’t want to read the [instruction] page which is relatively simple ” (C2P03 clinic receptionist).
Invest in clinical champions
All participants perceived the role of clinical champion to be pivotal to the intervention’s success. Champions were perceived to have two major responsibilities: advocating for the intervention among colleagues to boost motivation and providing practical support to build capability.
To be effective advocates, champions were perceived to need support from senior management ( “[leadership of change] it’s got to happen from the top ” (C5P02 RN)) as well as established, cordial relationships with colleagues they could leverage to motivate engagement: “ it also relies on the champion’s personal relationship with the staff which you’re asking to perform these roles and trying to change their management ” (C1P01 palliative care physician and clinical champion). Where champions felt under-supported by management, they relied on moral support from the project team to sustain their advocacy work: “ being the champion, and sometimes being the nagging champion, it actually felt quite nice to have the back-up of other people ” (C1P01 palliative care physician and clinical champion). Both physicians and nurses perceived the champion role might better suit the scope of practice of a junior doctor or senior nurse rather than consultants, based on their willingness to engage and approachability: “ realistically, you’re probably always going to get more engagement with registrars compared to consultants, unless it’s their own trial ” (C5P07 radiation oncologist); “ just give it [the role] to the CNCs [clinical nurse consultants] because as a general rule they’re the best at everything and have the best relationships with the patient ” (C3P04 medical oncologist).
From a practical perspective, clinical champions were expected to provide human resources for establishing and supporting pain screening and patient education: “ you need a body ” (C2P04 clinical nurse consultant). Unfortunately, however, champions across centres reported having limited time protected for the role within their usual duties: “ there just wasn’t the manpower to do that here ” (C3P02 palliative care physician and clinical champion). One suggestion for boosting capacity was to narrow the focus to one clinic and delegate practical tasks to less senior delegates than required for advocacy to render the time commitment more cost-effective: “[ it] might have been better to focus on one clinic and have full-time … junior nurse ” (C5P05 radiation oncologist). This presented an opportunity to train more than one clinical champion to provide better coverage across shifts and safeguard against the risk of losing champions to staff turnover.
Increasing pain awareness is the first step: Plan for slow incremental change rather than rapid uptake
While the barriers above meant only modest practice changes could be achieved, champions at half the centres perceived incremental progress had been made through increasing awareness among personnel regarding pain care as a focus for improvement: “ I think just trying to make pain something that people think about was probably one of the better strategies ” (C1P01 palliative care physician and clinical champion); it’s more at the top of our minds to remember, to screen the pain at every visit ” (C2P01 palliative care physician and clinical champion); “ I think it has highlighted those issues for us and we now need to take this on ” (C5P04 medical oncologist, head of department and clinical champion). Both nursing and medical participants at Centre 5 emphasized the need to be persistent in striving for continuous improvement: “ I think to get practice change, even for well-motivated people, I think it just needs to be pushed … they’ve done similar things with hand washing for doctors and it’s finally getting through ” (C504 medical oncologist and clinical champion); “ it would take more than just one of these kind of programs to get people to change ” (C5P03 clinical nurse consultant). Encouragingly, participants at this and one other centre expected some clinicians to continue using the patient education booklet and resource after the project ended: “ I’d just love to continue using these booklets ” (C5P02 RN); “[the] patient-held resource has been useful and has been taken up by people and I think they will continue to use those ” (C6P02 palliative care physician and clinical champion).
This qualitative sub-study of a cluster randomized controlled trial identified centre-level capabilities to be the most influential factors impeding or facilitating guideline implementation strategies for improving pain care for outpatients with cancer. Findings suggest that future initiatives of this kind should: consider centre readiness for change; articulate and deliver on the intervention’s value proposition; define clinician roles and responsibilities; integrate the intervention within existing systems and processes; promote patient partnership; invest in the clinical champion role, drawing from senior nurses and junior doctors, with support from medical leaders and management; and design the initiative around slow incremental change rather than rapid uptake.
Our findings are largely consistent with those from an ethnographic study exploring factors influencing implementation of cancer pain guidelines in Korean hospital cancer units, which identified a ‘lack of receptivity for change’ to be a key barrier [ 28 ]. However, observations from the Korean study suggested that a lack of centre leadership and cultural norms regarding nursing hierarchy were the most important underlying factors, whereas our Australian sample focused more on constraints imposed by centre systems and processes and a lack of clarity regarding disciplinary roles. These factors were consistently emphasized regardless of participants’ discipline and seniority, including by one centre’s head of department. Consistent with these findings, a recent Australian qualitative sub-study of anxiety/depression guideline implementation in oncology centres found greater role flexibility to be a key factor underpinning organisational readiness for change [ 29 ]. This team also provided quantitative evidence consistent with our finding that centres’ readiness for change is associated with personnel’s perception of benefit from guideline implementation [ 30 ]. Future initiatives should work harder to persuade clinicians of the intervention’s rationale and evidence base prior to commencement, given that perceptions of coherence and effectiveness are key dimensions of acceptability required for clinicians to invest time and effort [ 31 ]. Since our Trial was conducted, evidence has emerged for an impact from cancer symptom screening on survival that could be used persuasively [ 32 ]. Furthermore, the spaced education module might be more acceptable if made adjustable to the knowledge levels of a broader range of clinicians.
Other studies on implementation of cancer pain guidelines [ 11 , 13 ] suggest that structured approaches to process change tend to be more successful than less prescriptive approaches of the kind taken in the Stop Cancer PAIN Trial. We provided centres with guideline implementation strategies but no clear guidance on how to integrate these within existing contexts - i.e. implementation of the implementation, or ‘meta-implementation’. It was wrongly assumed that clinical champions could support integration with centre processes based on their knowledge of local context, but this turned out to be unreasonable given champions’ limited time for the role and lack of training in change management. Like most research to date [ 33 , 34 ], our trial focused largely on the advocacy role played by clinical champions, neglecting more practical and time consuming aspects that our interviews identified to be just as important. We join others in calling for more research on the mechanisms by which clinical champions can optimally facilitate change and ways to maximize their efficacy through training and support [ 24 ]. This should include exploration of optimal models by which different aspects of the champion role might be shared between more than one person where no-one is available with all the necessary attributes, as well as ways to ensure sustainability after support from the project team is withdrawn.
Theory-based research suggests that adding complex interventions to complex healthcare systems creates dynamic interplay and feedback loops, making consequences hard to predict [ 35 ]. In the current trial, this was likely exacerbated by our attempt to combine multiple strategies targeting patient, clinician and centre levels. We chose each strategy based on evidence for its stand-alone efficacy, and combined strategies rather than used them singly with the intent of leveraging complementary mechanisms, as recommended by the COM-B Model and US Institute of Medicine [ 36 ]. However, findings from our interviews suggest that interactions between the strategies and local processes separated their spheres of influence, precluding intended synergies. The Stop Cancer PAIN Trial is not alone in having over-estimated the value of combining guideline implementation strategies; a recent systematic review found that 8 other multi-component interventions similarly demonstrated limited effects on guideline adherence and patient outcomes [ 37 ]. Collectively, these findings suggest that future attempts at combining strategies should consider complex systems theory as well as behaviour change frameworks at each of a number of stages [ 38 ]. Alternatively, a more manageable approach for most cancer centres might be to focus on just one component at a time, periodically reviewing progress against SMART goals and, depending on results, supplementing with additional components using plan-do-study cycles [ 39 ].
Given the challenges with integrating screening into centre processes, it seems unlikely that improvements in pain scores during the control phase reported in our primary results article were due to the spontaneous use of screening data in consultations [ 18 ]. Indeed, while routine use of patient-reported outcome measures (PROMs) in oncology has been researched for more than a quarter-century [ 40 ], benefits to patient outcomes have only recently been demonstrated in the context of electronically-administered PROMs (ePROMs) that enable remote self-reporting, real-time feedback to clinicians, and clinician-patient telecommunication [ 12 ]. Further research is needed on how best to support clinician engagement with ePROMs, including training on how to use results in partnership with patients to assist shared decision-making and self-management [ 41 ].
A worrying finding from the current study was that some or all aspects of pain care were perceived to fall between the scopes of practice for oncology clinicians from each discipline. Clinical practice guidelines emphasize the need for pain care to be inter-disciplinary in recognition of the need for comprehensive assessment, non-pharmacological as well as pharmacological management, and patient education and support for self-management [ 42 ]. While the patient self-management resource included in the intervention was perceived to support communication between clinicians and patients, its potential for assisting coordination of care between disciplines was limited where roles and responsibilities were not previously established. Our findings and other research suggest that future initiatives may benefit from ‘process mapping’ with clinicians to identify where clinical workflow and roles might be reconfigured to incorporate the various aspects of pain care in the most efficient ways that do not substantially add to workload [ 41 ].
Patient education has been proven to improve pain outcomes by clinical trials [ 43 , 44 ], and we have argued previously that supporting pain self-management should be core business for all clinicians working in cancer care [ 45 ]. The ‘coaching’ approach needed to empower patients to recognize themselves as ‘experts’ on their pain and equal partners with clinicians in its management is iterative rather than a single event, and is ideally built on established and ongoing therapeutic relationships of trust with a particular team member. However, findings from patient education research more generally suggest that patient education and behaviour change is also optimally supported when key messages are reinforced by differing disciplinary perspectives [ 46 ]. Results from the current study suggest that these principles of pain care need more formal recognition within the scope of practice of oncology clinicians to ensure they are afforded sufficient time alongside anti-cancer treatment and related supportive care. Findings also indicate that clinicians may require training in the person-centred, partnership-oriented aspects of pain care beyond the educational approach used in the Stop Cancer PAIN Trial and other research [ 47 ]. Such training should be repeated regularly to ensure it reaches the majority of personnel at cancer centres, allowing for turnover.
Limitations
The current study had several limitations. Transferability even within Australia is limited by a focus on metropolitan services in only three out of eight jurisdictions. Data relied on clinician perspectives, and the response rate was less than one quarter of personnel at each centre, with the disciplines and specialties of participants being unrepresentative of centre workforces. Over-sampling of medical compared to nursing personnel likely reflects the fact that all clinical champions were medical consultants, while the predominance of palliative care physicians among medical participants presumably arises from the central focus this specialty has on pain care. Notably, our sample included no perspectives from allied health disciplines, despite the important roles these can play in non-pharmacological pain management. Confirmability was threatened by the potential for cognitive bias among researchers towards a favourable view of the intervention given their long-standing investment as members of the project team. We attempted to offset this by explicitly inviting criticism of the intervention from participants, and having the initial analysis conducted by researchers with no prior involvement in the project. A final limitation concerns reliance on the COM-B Model for analysis rather than an alternative framework or more inductive approach. While the COM-B has been widely used to explore barriers and facilitators across a wide range of healthcare interventions, we applied the model in a somewhat novel way to systems and processes as well as individuals’ behaviour after finding that participants perceived their agency to be majorly constrained by these. An implementation framework such as the integrated-Promoting Action on Research Implementation in Health Service (i-PARIHS) framework (iPARIHS) [ 48 ] or Consolidated Framework for Implementation Research (CFIR) [ 49 ] would have conceived of factors and their relationships in alternative ways that might have proven equally informative [ 50 ].
This qualitative sub-study elucidated important factors influencing the success of guideline implementation strategies at six cancer centres in the Stop Cancer PAIN Trial. Findings underscore the value that a qualitative approach offers for understanding the role of context when evaluating complex interventions [ 51 ]. Ultimately, the Stop Cancer PAIN Trial may have been overly ambitious in the scale of its intervention, especially given limited resources available at each centre. Further research is needed to understand how multi-component guideline implementation strategies can be optimally introduced within the context of local roles, systems and processes.
Availability of data and materials
The qualitative interview datasets generated and analysed during the current study are not publicly available due to the conditions of ethical approval which acknowledge the risk of participant re-identification.
Van Den Beuken-Van MH, Hochstenbach LMJ, Joosten EAJ, Tjan-Heijnen VCG, Janssen DJA. Update on prevalence of pain in patients with cancer: systematic review and meta-analysis. J Pain Symptom Manag. 2016;51(6):1070–90.
Article Google Scholar
Luckett T, Davidson PM, Green A, Boyle F, Stubbs J, Lovell M. Assessment and management of adult cancer pain: a systematic review and synthesis of recent qualitative studies aimed at developing insights for managing barriers and optimizing facilitators within a comprehensive framework of patient care. J Pain Symptom Manag. 2013;46(2):229–53.
Oldenmenger WH, Sillevis Smitt PAE, van Dooren S, Stoter G, van der Rijt CCD. A systematic review on barriers hindering adequate cancer pain management and interventions to reduce them: a critical appraisal. Eur J Cancer. 2009;45(8):1370–80.
Article PubMed Google Scholar
Jacobsen R, Sjogren P, Moldrup C, Christrup L. Physician-related barriers to cancer pain management with opioid analgesics: a systematic review. J Opioid Manag. 2007;3(4):207–14.
Jacobsen R, Moldrup C, Christrup L, Sjogren P. Patient-related barriers to cancer pain management: a systematic exploratory review. Scand J Caring Sci. 2009;23(1):190–208.
Jacobsen R, Liubarskiene Z, Moldrup C, Christrup L, Sjogren P, Samsanaviciene J. Barriers to cancer pain management: a review of empirical research. Medicina (Kaunas). 2009;45:427–33.
Fazeny B, Muhm M, Hauser I, Wenzel C, Mares P, Berzlanovich A, et al. Barriers in cancer pain management. Wiener Klinische Wochenschrift. 2000;112(22):978–81.
CAS PubMed Google Scholar
Makhlouf SM, Pini S, Ahmed S, Bennett MI. Managing pain in people with cancer—a systematic review of the attitudes and knowledge of professionals, patients, caregivers and Public. J Cancer Educ. 2020;35(2):214–40.
Roberto A, Greco MT, Uggeri S, Cavuto S, Deandrea S, Corli O, et al. Living systematic review to assess the analgesic undertreatment in cancer patients. Pain Pract. 2022;22(4):487–96.
Brink-Huis A, van Achterberg T, Schoonhoven L, Brink-Huis A, van Achterberg T, Schoonhoven L. Pain management: a review of organisation models with integrated processes for the management of pain in adult cancer patients. J Clin Nurs. 2008;17(15):1986–2000.
Du Pen SL, Du Pen AR, Polissar N, Hansberry J, Kraybill BM, Stillman M, et al. Implementing guidelines for cancer pain management: results of a randomized controlled clinical trial. J Clin Oncol. 1999;17(1):361–70.
Article CAS PubMed Google Scholar
Govindaraj R, Agar M, Currow D, Luckett T. Assessing patient-reported outcomes in routine cancer clinical care using electronic administration and telehealth technologies: Realist synthesis of potential mechanisms for improving health outcomes. J Med Internet Res. 2023;25:e48483.
Article PubMed PubMed Central Google Scholar
Koesel N, Tocchi C, Burke L, Yap T, Harrison A. Symptom distress: implementation of palliative care guidelines to improve pain, fatigue, and anxiety in patients with advanced cancer. Clin J Oncol Nurs. 2019;23(2):149–55.
PubMed Google Scholar
Dulko D, Hertz E, Julien J, Beck S, Mooney K. Implementation of cancer pain guidelines by acute care nurse practitioners using an audit and feedback strategy. J Am Acad Nurse Pract. 2010;22(1):45–55.
Samuelly-Leichtag G, Adler T, Eisenberg E. Something must be wrong with the implementation of cancer-pain treatment guidelines. A lesson from referrals to a pain clinic. Rambam Maimonides Med J. 2019;10(3):18.
Lopez Lopez R, Camps Herrero C, Khosravi-Shahi P, Guillem Porta V, Carrato Mena A, Garcia-Foncillas J, et al. Oncologist’s knowledge and implementation of guidelines for breakthrough cancer pain in Spain: CONOCE study. Clin Transl Oncol. 2018;20(5):613–8.
Luckett T, Phillips J, Agar M, Lam L, Davidson PM, McCaffrey N, et al. Protocol for a phase III pragmatic stepped wedge cluster randomised controlled trial comparing the effectiveness and cost-effectiveness of screening and guidelines with, versus without, implementation strategies for improving pain in adults with cancer attending outpatient oncology and palliative care services: the Stop Cancer PAIN trial. BMC Health Serv Res. 2018;18(1):1–13.
Lovell MR, Phillips JL, Luckett T, Lam L, Boyle FM, Davidson PM, et al. Effect of cancer pain guideline implementation on pain outcomes among adult outpatients with cancer-related pain: a stepped wedge cluster randomized trial. JAMA Netw Open. 2022;5(2):1–12.
Lovell M, Luckett T, Boyle F, Stubbs J, Phillips J, Davidson PM, et al. Adaptation of international guidelines on assessment and management of cancer pain for the Australian context. Asia-Pac J Clin Oncol. 2015;11(2):170–7.
Phillips JL, Heneka N, Bhattarai P, Fraser C, Shaw T. Effectiveness of the spaced education pedagogy for clinicians’ continuing professional development: a systematic review. Med Educ. 2019;53(9):886–902.
Cancer Council Australia. Overcoming cancer pain: A guide for people with cancer, their families and friends 2013 [Available from: http://www.cancercouncil.com.au/wp-content/uploads/2014/01/Can487-Overcoming-Pain-NSW-Lores2.pdf .
Luckett T, Davidson PM, Green A, Marie N, Birch MR, Stubbs J, et al. Development of a cancer pain self-management resource to address patient, provider, and health system barriers to care. Palliat Support Care. 2019;17(4):472–8.
Michie S, Atkins L, West R. The behaviour change wheel: a guide to designing interventions. London: Silverback Publishing; 2014.
Google Scholar
Santos WJ, Graham ID, Lalonde M, Demery Varin M, Squires JE. The effectiveness of champions in implementing innovations in health care: a systematic review. Implement Sci Commun. 2022;3(1):80.
Ramanadhan S, Revette AC, Lee RM, Aveling EL. Pragmatic approaches to analyzing qualitative data for implementation science: an introduction. Implement Sci Commun. 2021;2(1):70.
Tong A, Sainsbury P, Craig J. Consolidated criteria for reporting qualitative research (COREQ): a 32-item checklist for interviews and focus groups. Int J Qual Health Care. 2007;19(6):349–57.
Gale NK, Heath G, Cameron E, Rashid S, Redwood S. Using the framework method for the analysis of qualitative data in multi-disciplinary health research. BMC Med Res Methodol. 2013;13(1):117.
Kim M, Jeong S, McMillan M, Higgins I. Translation of policy and guidelines into practice: lessons learned from implementation of the cancer pain management guideline. J Hosp Manage Health Policy. 2020;4. https://doi.org/10.21037/jhmhp.2020.02.01 .
Geerligs L, Shepherd HL, Butow P, Shaw J, Masya L, Cuddy J, et al. What factors influence organisational readiness for change? Implementation of the Australian clinical pathway for the screening, assessment and management of anxiety and depression in adult cancer patients (ADAPT CP). Support Care Cancer. 2021;29(6):3235–44.
Faris MM, Shepherd HL, Butow PN, Kelly P, He S, Rankin N, et al. Staff- and service-level factors associated with organisational readiness to implement a clinical pathway for the identification, assessment, and management of anxiety and depression in adults with cancer. BMC Health Serv Res. 2023;23(1):866.
Sekhon M, Cartwright M, Francis JJ. Acceptability of healthcare interventions: an overview of reviews and development of a theoretical framework. BMC Health Serv Res. 2017;17(1):88.
Caminiti C, Maglietta G, Diodati F, Puntoni M, Marcomini B, Lazzarelli S, et al. The effects of patient-reported outcome screening on the survival of people with cancer: a systematic review and meta-analysis. Cancers. 2022;14(21):5470.
Morena AL, Gaias LM, Larkin C. Understanding the role of clinical champions and their impact on clinician behavior change: the need for causal pathway mechanisms. Front Health Serv. 2022;2:896885.
Miech EJ, Rattray NA, Flanagan ME, Damschroder L, Schmid AA, Damush TM. Inside help: an integrative review of champions in healthcare-related implementation. SAGE Open Med. 2018;6:2050312118773261.
Braithwaite J, Churruca K, Long JC, Ellis LA, Herkes J. When complexity science meets implementation science: a theoretical and empirical analysis of systems change. BMC Med. 2018;16:1–14.
Institute of Medicine (US) Committee on Standards for Developing Trustworthy Clinical Practice Guidelines. Clinical Practice Guidelines We Can Trust. In: Graham R, Mancher M, Miller Wolman D, Greenfield S, Steinberg E, editors. Washington, D.C.: National Academies Press; 2011.
Bora A-M, Piechotta V, Kreuzberger N, Monsef I, Wender A, Follmann M, et al. The effectiveness of clinical guideline implementation strategies in oncology—a systematic review. BMC Health Serv Res. 2023;23(1):347.
McGill E, Er V, Penney T, Egan M, White M, Meier P, et al. Evaluation of public health interventions from a complex systems perspective: a research methods review. Soc Sci Med. 2021;272: 113697.
Institute for Healthcare Improvement. Quality improvement essentials toolkit. 2017.
Trowbridge RDW, Jay SJ, et al. Determining the effectiveness of a clinical-practice intervention in improving the control of pain in outpatient with cancer. Acad Med. 1997;72:798–800.
Howell D, Rosberger Z, Mayer C, Faria R, Hamel M, Snider A, et al. Personalized symptom management: a quality improvement collaborative for implementation of patient reported outcomes (PROs) in ‘real-world’ oncology multisite practices. J Patient Rep Outcomes. 2020;4(1):47.
Australian Adult Cancer Pain Management Guideline Working Party. Autralian clinical pathway for screening, assessment and management of cancer pain in adults Sydney, Australia: Cancer Council Australia. 2013. http://wiki.cancer.org.au/australia/Guidelines:Cancer_pain_management/Flowchart_overview .
Marie N, Luckett T, Davidson P, Lovell M, Lal S. Optimal patient education for cancer pain: a systematic review and theory-based meta-analysis. Support Care Cancer. 2013;21:3529–37.
Abernethy AP, Currow DC, Shelby-James T, Rowett D, May F, Samsa GP, et al. Delivery strategies to optimize resource utilization and performance status for patients with advanced life-limiting illness: results from the palliative care trial [ISRCTN 81117481]. J Pain Symptom Manage. 2013;45(3):488–505.
Lovell MR, Luckett T, Boyle FM, Phillips J, Agar M, Davidson PM. Patient education, coaching, and self-management for cancer pain. J Clin Oncol. 2014;32(16):1712–20.
Luckett T, Roberts MM, Smith T, Swami V, Cho JG, Wheatley JR. Patient perspectives on how to optimise benefits from a breathlessness service for people with COPD. NPJ Prim Care Respir Med. 2020;30(1). https://doi.org/10.1038/s41533-020-0172-4 .
Phillips JL, Heneka N, Lovell M, Lam L, Davidson P, Boyle F, et al. A phase III wait-listed randomised controlled trial of novel targeted inter-professional clinical education intervention to improve cancer patients’ reported pain outcomes (the Cancer Pain Assessment (CPAS) Trial): study protocol. Trials. 2019;20:1–12.
Harvey G, Kitson A. PARIHS revisited: from heuristic to integrated framework for the successful implementation of knowledge into practice. Implement Sci. 2016;11(1):33.
Damschroder LJ, Reardon CM, Widerquist MAO, Lowery J. The updated consolidated framework for implementation research based on user feedback. Implement Sci. 2022;17(1):1–16.
Geerligs L, Rankin NM, Shepherd HL, Butow P. Hospital-based interventions: a systematic review of staff-reported barriers and facilitators to implementation processes. Implement Sci. 2018;13(1):36.
Skivington K, Matthews L, Simpson SA, Craig P, Baird J, Blazeby JM, et al. A new framework for developing and evaluating complex interventions: update of medical research council guidance. BMJ. 2021;374:n2061.
Download references
Acknowledgements
The authors would like to dedicate this article to the memory of Sally Fielding, who worked as a valued member of the project team throughout the Stop Cancer PAIN Trial. We would also like to acknowledge the contributions of project manager A/Prof Annmarie Hosie, data manager Dr Seong Cheah, and research assistant Layla Edwards.
This research was supported by a grant from the National Breast Cancer Foundation.
Author information
Authors and affiliations.
IMPACCT Centre—Improving Palliative, Aged and Chronic Care through Clinical Research and Translation, Faculty of Health, University of Technology Sydney (UTS), Building 10, 235 Jones St, Ultimo, Sydney, NSW, 2007, Australia
Tim Luckett, Meera Agar & Maja Garcia
School of Nursing and Centre for Healthcare Transformation, Queensland University of Technology (QUT), Brisbane, QLD, Australia
Jane Phillips
South West Sydney School of Clinical Medicine, University of New South Wales (UNSW), Sydney, NSW, Australia
The Limbic, Sydney, Australia
Linda Richards
Palliative Care Department, Greenwich Hospital, HammondCare, Sydney, NSW, Australia
Najwa Reynolds & Melanie Lovell
University of Wollongong, Wollongong, NSW, Australia
Patricia Davidson & David Currow
Charles Perkins Centre, School of Medical Sciences, The University of Sydney, Sydney, NSW, Australia
Patricia Ritchie Centre for Cancer Care and Research, The University of Sydney, Sydney, NSW, Australia
Frances Boyle
Northern Medical School, The University of Sydney, Sydney, NSW, Australia
Frances Boyle & Melanie Lovell
Macau University of Science and Technology, Macau, China
Lawrence Lam
Deakin Health Economics, Institute for Health Transformation, School of Health and Social Development, Deakin University, Melbourne, Australia
Nikki McCaffrey
You can also search for this author in PubMed Google Scholar
Contributions
TL, JP, MA, PMD, TS, DCC, FB, LL, NM and ML contributed to the concept and design of this research. TL, LR, MR, MG and ML contributed to the acquisition, analysis or interpretation of the data. TL and ML contributed to drafting of the manuscript. All authors contributed to revisions of the manuscript and approved the final version.
Corresponding author
Correspondence to Tim Luckett .
Ethics declarations
Ethics approval and consent to participate.
This research was approved by the Southwestern Sydney Local Health District Human Research Ethics Committee (HREC/14/LPOOL/479). All participants gave written informed consent to participate.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/ .
Reprints and permissions
About this article
Cite this article.
Luckett, T., Phillips, J., Agar, M. et al. Factors influencing fidelity to guideline implementation strategies for improving pain care at cancer centres: a qualitative sub-study of the Stop Cancer PAIN Trial. BMC Health Serv Res 24 , 969 (2024). https://doi.org/10.1186/s12913-024-11243-1
Download citation
Received : 10 April 2024
Accepted : 25 June 2024
Published : 22 August 2024
DOI : https://doi.org/10.1186/s12913-024-11243-1
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Implementation
- Qualitative
BMC Health Services Research
ISSN: 1472-6963
- General enquiries: [email protected]
- Open access
- Published: 20 August 2024
Exploring factors affecting the acceptance of fall detection technology among older adults and their families: a content analysis
- Hsin-Hsiung Huang 1 ,
- Ming-Hao Chang 1 ,
- Peng-Ting Chen 1 , 2 ,
- Chih-Lung Lin 3 ,
- Pi-Shan Sung 4 ,
- Chien-Hsu Chen 5 &
- Sheng-Yu Fan 6
BMC Geriatrics volume 24 , Article number: 694 ( 2024 ) Cite this article
164 Accesses
14 Altmetric
Metrics details
This study conducted in-depth interviews to explore the factors that influence the adoption of fall detection technology among older adults and their families, providing a valuable evaluation framework for healthcare providers in the field of fall detection, with the ultimate goal of assisting older adults immediately and effectively when falls occur.
The method employed a qualitative approach, utilizing semi-structured interviews with 30 older adults and 29 families, focusing on their perspectives and expectations of fall detection technology. Purposive sampling ensured representation from older adults with conditions such as Parkinson's, dementia, and stroke.
The results reveal key considerations influencing the adoption of fall-detection devices, including health factors, reliance on human care, personal comfort, awareness of market alternatives, attitude towards technology, financial concerns, and expectations for fall detection technology.
Conclusions
This study identifies seven key factors influencing the adoption of fall detection technology among older adults and their families. The conclusion highlights the need to address these factors to encourage adoption, advocating for user-centered, safe, and affordable technology. This research provides valuable insights for the development of fall detection technology, aiming to enhance the safety of older adults and reduce the caregiving burden.
Peer Review reports
Introduction
As the population of older adults grows, an emerging concern revolves around the prevalence of falls. Age-related gait and balance issues are prevalent and significant in the older adults, increasing the risk of falls and injuries [ 1 ]. Falls can result in a range of injuries, such as fracture or head injury [ 2 , 3 ]. Undoubtedly, the aging population faces a substantial risk related to falls, leading to both mortality and morbidity [ 4 ]. In the United States, statistics indicate that in 2018, 27.5% of adults aged 65 and older reported experiencing at least one fall in the previous year [ 5 ]. One out of five falls results in severe injuries, such as fractures or head trauma. These falls incurred a staggering $50 billion in total medical expenses in the US in 2015 [ 6 ]. There has been a concerning rise in the number of falls resulting in injuries over the years. One study revealed that only 39% of older individuals reported experiencing a fall [ 7 ]. Furthermore, research suggests that the impact of falls continues to affect both admitted and non-admitted older adults, leading to a reduced quality of life for up to nine months following the injury [ 8 ]. On the one hand, a study revealed significant concern and fear among individuals regarding the possibility of the older adults experiencing another fall [ 9 ]. On the other hand, time on the ground (TOG) has been identified as a crucial factor affecting prognosis after a fall. TOG refers to the duration an individual remains on the ground after falling. This factor has been specifically examined in dementia patients, as falls frequently occur in memory care facilities [ 10 ]. However, falls occurring within the home environment during old age often signal the presence of severe underlying health conditions, especially without intime assistance like memory care facilities [ 11 ]. Obviously, falls among older adults is an imperative issue that needs to be addressed.
Given the fact that falls pose a significant concern in healthcare and for family caregivers, there is a growing interest in the development of methods to detect falls. Previous studies on fall detection technology explore the use of sensors in detecting fall-related events among older individuals [ 12 , 13 , 14 ]. One study states that fall detection technology covers three dimensions, including wearable devices, camera-based devices, and ambiance devices. It's worth mentioning that many fall detection methods are already mature and commercially available. These include video-based systems using cameras to monitor movements, microwave-based methods with radar technology to detect falls, and acoustic monitoring that analyzes sounds to identify fall events. These technologies provide valuable alternatives and enhancements to sensor-based fall detection systems [ 15 ]. Wearable devices gather data on body posture and movement, utilizing algorithms to determine if a fall has occurred. Cameras strategically positioned enable ongoing monitoring of older adults, with captured data stored for subsequent analysis and reference. Ambience devices are placed in the surroundings, like walls, floors, and beds. Data from sensors are collected, and an algorithm analyzes the input to determine if a fall has occurred [ 14 ]. Another study found that many solutions also use mobile device sensors, particularly accelerometers, for fall detection in older adults [ 13 ]. The above literature review provides examples of fall detection technology application areas that already exist in the market. Therefore, fall detection technology among older adults has the potential to alleviate the societal burden. However, technology-based solutions, despite their potential benefits, often face resistance from older adults, creating barriers to the adoption of health-related information and communication technology. To address these barriers, we conducted a comprehensive literature review, examining the challenges that older adults may encounter when using fall detection technology.
In 1987, Ram introduced an innovation resistance model [ 16 ], aiming to address the reluctance of consumers to adopt new innovations, particularly when these innovations have the potential to disrupt their existing satisfaction levels or clash with their established beliefs. Building upon this framework, Ram and Sheth [ 17 ] (1989) identified a range of obstacles that hinder consumers' willingness to embrace innovations, classifying them into two main categories: functional barriers and psychological barriers. Functional barriers encompass aspects such as usage limitations, value considerations, and risk perceptions. We conducted a literature review on the barriers that older adults may face when using the technology. Among usage barriers, age-related factors, including hearing impairments, reduced dexterity, declining vision, and mild cognitive challenges, can significantly impact the ease with which users adopt new technologies [ 18 , 19 , 20 , 21 , 22 ]. Previous research [ 18 , 23 , 24 , 25 , 26 ] has emphasized that technical unfamiliarity, which includes inadequate technical skills, a lack of understanding about how to use technology, and limited computer literacy, poses significant challenges for older individuals in adopting new technologies. Additionally, a lack of clear and comprehensive instructions has been identified as a common obstacle for older adults in the literature [ 24 , 27 , 28 ]. Given that the value barrier concept suggests innovative products must offer greater value than existing ones to motivate consumers to switch, there is a scarcity of references related to this description. On the other hand, risk barriers encompass concerns about product reliability, including issues like false alarms and inaccurate data, which can be functional risks that older individuals may encounter [ 19 , 27 , 29 , 30 , 31 ]. High costs also contribute to risk barriers. Many older adults are concerned about the price of the product itself [ 22 , 30 , 32 ]. Furthermore, privacy concerns have been raised by many older individuals, adding to the array of issues related to risk barriers [ 18 , 21 , 22 , 33 , 34 ].
Psychological barriers encompass traditional belief barriers and image-related barriers. Older adults also encounter psychological barriers when using information and communication technology. Among older adults, attitude toward technology represents a common traditional belief barrier, reflecting issues related to trust in their ability to manage devices and their reluctance to adopt it [ 18 , 21 , 35 ]. Image barriers involve concerns about a product's appearance [ 27 ], with some older individuals perceiving certain products as designed for younger generations, which may deter their adoption [ 24 ].
While numerous articles have explored the barriers older individuals face in adopting information and communication technology (ICT) [ 18 , 22 , 36 ], it's essential to acknowledge that ICT encompasses a wide range of applications, making it a diverse and multifaceted topic. Within healthcare, various applications exist, which can make it challenging for healthcare providers to develop products that cater specifically to their target users. While the previous studies encompass fall prevalence, economic burden of falls, and the challenges older adults may face when using ICT, this study focuses more on barriers of these technological products used by older adults and their families, providing a valuable evaluation framework that can aid healthcare providers, particularly in the field of fall detection. Through this research, we aim to offer a valuable assessment framework for making the best use of ICT to help older adults immediately and effectively when falls happen.
Study design
In order to address our research inquiry on the perceived challenges associated with the adoption of fall detection technology and expectations of fall detection technology among older adults and their families, we employed a qualitative approach. Our primary sources of data analysis were semi-structured interviews from in-depth interviews. In-depth interviews are widely acknowledged and commonly used in qualitative research [ 37 ]. The semi-structured interview outline utilized in our study provided a well-defined yet flexible and open-ended framework for exploring the topic [ 38 ]. To align with the research objectives, we developed a semi-structured interview outline, including the background of participants, expectations of fall detection technology, and innovation resistance (see Tables 1 and 2 ). Face-to-face interviews were then conducted with older adults along with their families.
Study subject and recruitment
The aim of this study was to understand the perspectives of older adults with chronic disease, who are prone to falls [ 1 ], and their family caregivers, who are the older adults’ spouses or children. Purposive sampling was employed, and specific inclusion criteria were set for the study participants. These criteria consisted of: (1) healthy individuals over the age of 20 who agreed to participate; (2) participants aged 45 or above, including those affected by stroke, frailty, dementia, Parkinson's disease, and other diseases; (3) participants whose condition was stable, able to mobilize, and willing to take part in the study. We included participants younger than 60 years old in our study because they have chronic diseases such as stroke, dementia, and Parkinson's disease. Individuals with these conditions are more prone to falls compared to others. Although these diseases are typically associated with older adults, we believe that younger participants with these conditions are potential future users of fall detection technology. Therefore, our sample includes individuals under 60 years old and their respective family caregivers.
To ensure clear comprehension of the study's purpose, procedures, and potential risks, an individualized approach was adopted in explaining the study to each participant. Additionally, oral explanations were provided to ensure their understanding of the research instructions and terms outlined in the consent form. In total, interviews were conducted with 30 older adults and 29 families (with one family unable to attend).
Data collection
The study received ethical approval from the Human Research Ethics Review Committee, and the case number assigned was A-ER-110–211. From September 2022 to April 2023, in-depth interviews were conducted in NCKU outpatient hospital using a semi-structured interview outline. The interview process began with the researchers introducing themselves to the participants and providing a detailed explanation of the study's purpose, the interview procedure, and the rights of the participants. Privacy regulations were emphasized, assuring the interviewees that their personal data would be treated confidentially. Following comprehension of the study's objectives and their rights, the participants were informed about the recording of the interview. It was made clear that if they preferred not to be recorded, the investigators would respect their decision and take handwritten notes instead. Each interview lasted approximately 40–60 min. After each interview, research assistants were responsible for transcribing the recorded interview files to create a written transcript of the data. Prior to analysis, the researchers reviewed the verbatim transcripts of the interviews to ensure accuracy and identify any potential errors. If any inconsistencies or missing information were found, another researcher would review the audio recording and the transcript to ensure accuracy and correct any deviations from the original intended meaning.
Data analysis
The qualitative interview data in this study was subjected to content analysis. To streamline the content analysis process and identify themes within the qualitative responses, a panel consisting of four members was established. In addition, the whole process of data analysis was supervised by the professor. The panels include one doctoral researcher, one research assistant, and two graduate students. In employing the inductive approach, 4 researchers employed a systematic process that involved dividing the data into distinct units of meaning, condensing these units, assigning codes, categorizing the codes, and identifying overarching themes [ 39 , 40 ]. The analysis began with the researchers thoroughly reading and rereading the interview data, treating each segment as a unit of analysis. Similar statements within the text were identified and extracted to form meaning units. These meaning units were then condensed through a careful reduction process while ensuring the preservation of their core essence. Subsequently, the meaning units were systematically coded based on their content, with researchers assigning specific codes to each unit. Once the coding process was complete, all the codes were further organized into meaningful categories. Finally, the researchers identified and grouped together different categories that shared related underlying meanings, thereby forming overarching themes [ 41 ]. This rigorous approach to content analysis enabled a comprehensive exploration and interpretation of the qualitative interview data in the study.
Respondent characteristics
From September 2022 to April 2023, the study included 30 older adults and 29 family members, all recruited from NCKU Medical Center in Taiwan. These participants are referred to as N_ Interviewee (older adults /family). The older adults, primarily diagnosed with Parkinson's disease, dementia, or stroke, were selected based on their scores on the Morse Scale [ 42 ], Clinical Frailty Scales [ 43 ], and Barthel Index [ 44 ]. Additionally, the study documented the history of fall events and the relationship between the older adults and their family. Among the older adults, 19 older adults had experience using smartphones, while the remaining older adults did not have the experience (Table 3 ).
Based on the interviews conducted with older adults and their families, we have identified the primary considerations influencing the decision to use wearable fall-detection devices (as detailed in Fig. 1 ; Appendix). These considerations span various aspects, including (1) health considerations, (2) reliance on human care, (3) personal comfort issues, (4) market alternatives, (5) attitude towards technology, (6) financial concerns, and (7) expectations for fall detection technology. The main factors are described below.
Factors influencing adoption of fall detection technology in older adults and families
Health considerations
Concerns about potential health risks associated with wearable fall-detection devices emerged as a significant barrier to their adoption. older adults and their families expressed apprehensions about adverse effects such as dizziness, skin irritation, electrical leakage, and electromagnetic radiation. These concerns are particularly pronounced among older individuals, who tend to be more cautious about new technologies that interact directly with their bodies.
“Yeah, older adults won’t wear it if it's uncomfortable; it's just about avoiding dizziness.” (8_family)
For instance, some family members voiced worries about the possible radiation-related functions of these devices. Others were concerned about the risk of skin allergies and electrical leakage due to the close contact of these devices with the skin. These apprehensions highlight a broader fear of unknown health impacts, which can deter older adults from embracing new technological solutions for fall detection.
“Well, just now, it's just that I've heard that there might be some concerns about it. Because it's worn on the skin, so there's a fear of it having some impact on their skin. Also, there's the question of whether it might have electrical leakage.” (6_family)
“Perhaps, he has some kind of fear, like he might think that this thing could cause harm to the body? Or maybe he's worried about things like skin allergies or getting an electric shock, and so on.” (20_family)
Reliance on human care
Despite the potential benefits of fall-detection technology, many participants in the study emphasized a strong preference for human care and assistance. The majority believe that hiring caregivers or relying on family members is a more reliable and comforting approach. This trust in human assistance is deeply rooted and may significantly hinder the adoption of technological solutions.
Several older adults indicated that they felt no need for fall-detection devices because they were constantly accompanied by attentive family members or professional caregivers. For instance, some older adults mentioned that their spouses or foreign domestic workers were always available to assist them with daily activities, rendering the technology unnecessary. Others noted that their children, who are medical professionals, provided adequate care, further diminishing the perceived need for such devices.
Additionally, the cultural context plays a significant role in this reliance on human care. The close-knit family structure and the high value placed on personal interaction and caregiving contribute to the resistance against technological interventions. Many participants expressed a preference for investing in human care over spending money on devices, indicating that they view personal care as more effective and compassionate.
“Most people now hire foreign domestic workers to provide care. If he needs to get up to go to the bathroom, he'll definitely inform the foreign caregiver, saying, "I need this, I need that, please help me up.” (22_older adults)
“So instead of this, we might end up hiring someone to take care of him or considering long-term care services. Because rather than spending that money, it's the same as having someone look after you 24 h a day.” (2_family)
In summary, both health considerations and a strong reliance on human care are critical factors influencing the adoption of wearable fall-detection devices among older adults. Addressing these concerns through better education about the safety and benefits of these technologies, as well as integrating them into existing caregiving practices, may help in overcoming these barriers.
Personal comfort issues
The comfort and practicality of wearable devices are critical concerns for potential users, significantly impacting their adoption. Key issues identified include the weight and physical discomfort of these devices. Users are generally inclined to avoid technologies that cause inconvenience or discomfort in their daily lives, highlighting the necessity for user-friendly and ergonomic designs.
Participants indicated that the weight of the devices is a primary concern; many stated a preference for lightweight options. Physical discomfort, such as restrictions in movement, emerged as a significant factor. For example, older adults expressed concerns about devices causing discomfort when attached to the knee or foot, which could interfere with their mobility and overall comfort. There is a clear preference for devices that are unobtrusive and do not hinder daily activities.
“Fastened around the knee, I can't do it now. I'm afraid I'll get stuck when I'm walking.” (1_older adults)
“I care about the weight. It shouldn't be too heavy; it should be relatively lightweight.” (20_older adults)
Market alternatives
The preference for traditional fall prevention tools, such as canes and emergency buttons, was evident among many participants. These established solutions are familiar and trusted, making them more appealing than newer technological alternatives. Additionally, some participants believed that canes provide proactive assistance to prevent falls, whereas fall detection technology only alerts family members after a fall has occurred, which does not prevent the incident itself.
Participants noted that they already possess reliable fall prevention tools at home, such as emergency buttons, which they trust for their effectiveness in emergencies. The familiarity and simplicity of these tools make them a preferred choice over fall detection technology. Additionally, canes with stable bases are viewed as effective in ensuring personal safety and preventing falls, further reducing the perceived need for fall detection technology. To compete with traditional methods, fall-detection technology must not only match but surpass the reliability and convenience of existing tools.
“I currently have an emergency button installed in my home. If I have an accident, I can just press that button, and the security company will come to assist me.” (19_older adults)
“Because he just took the crutch and walked with it. Yes, if he wears this, he will still fall.” (8_family)
Attitude towards technology
A prevailing theme in the interviews is resistance to change, with some older individuals expressing a reluctance to adapt to new technologies. This resistance is often rooted in perceptions of inconvenience, unfamiliarity, and a general aversion to having devices attached to their bodies. Overcoming this resistance will require addressing user concerns and providing user-friendly solutions.
Elderly individuals frequently describe new devices as uncomfortable and cumbersome. For example, one older adult noted feeling "strange" and "not used to it" when considering wearing fall-detection devices. Others expressed outright resistance, emphasizing a strong preference for maintaining their current routines without the addition of new technological elements. This sentiment is further compounded by a dislike for the perceived hassle of wearing or carrying additional items, such as glasses or wearable devices.
“It's a strange feeling, doesn't feel like it, not used to it, feels weird.” (16_older adults)
“I'm just too lazy to wear glasses. We usually don't like having things hanging here and there.” (24_older adults)
“And to be honest, older people might have a greater psychological burden. If you ask them to carry something every day, they might not like it or feel that it restricts their mobility, and they might not want it.” (20_family)
Financial concerns
The cost of fall-detection devices is a significant consideration for many older adults and their families. Affordability is a key factor in their decision-making process, with financial capability greatly impacting the willingness to adopt new technology.
Many participants highlighted the financial burden that expensive fall-detection devices could impose. For families already managing substantial living expenses, the additional cost of advanced technology may be prohibitive. This financial strain is particularly acute for those on fixed incomes or with limited financial resources.
“I don’t want this if it’s too much money.” (9_older adults)
“I think financial capability comes first. If there are no issues with economic conditions, you have to make sure they have the financial ability to afford it. That's the main issue.” (5_family)
Expectations for fall detection technology
Participants highlighted several key expectations for fall detection technology, which, if met, could facilitate its adoption. These expectations include features such as remote notifications, physical support, real-time older adults status updates, and immediate assistance functions. Meeting these expectations can enhance the perceived value of fall detection technology and increase user willingness to adopt it.
A major expectation is the ability of the technology to provide real-time notifications to caregivers or family members when a fall occurs. Participants expressed a desire for systems that could alert them regardless of their location, ensuring timely intervention. For example, one family member emphasized the need for notifications even if older adults are far away, illustrating the importance of reliable and far-reaching communication capabilities.
Another expectation is for the technology to offer some form of physical support to prevent falls before they happen. Participants envisioned devices that could sense an impending fall and provide immediate physical assistance to prevent the incident. This proactive approach would not only enhance safety but also provide peace of mind for both users and their caregivers.
Real-time older adults’ status updates and the ability to monitor the condition of older adults remotely were also highly valued. For instance, having access to visual data or images of the older adults’ home environment was seen as a way to increase the sense of security and ensure timely responses to any issues. Comprehensive data on the older adults' health and activity levels could help in managing and understanding their overall condition.
“If we can assist her just before she falls, that would be the ideal scenario. Being able to support her right before the fall occurs.” (1_family)
“So, if we talk about it in terms of shoes, if it can sense that a person might slip or fall, can it prevent them from falling?” (2_family)
“It might be like this. If he wears it and triggers the alarm when he's far away, like what I just mentioned, if he's in Xitou and triggers the alarm, we're in Tainan.” (6_family)
“Data, as I just mentioned, is about being able to have a more immediate and clear understanding of the progression of the condition. And assuming that there is also the capability to capture images or, in a way, for me to see their condition at home, this might make me feel more at ease.” (10_family)
The adoption of fall-detection wearable devices among older individuals and their families is influenced by a complex interplay of factors, as revealed by the findings of this study. Understanding these factors is essential for the successful integration of such technologies into the lives of older adults. The participants' concerns about safety issues, such as skin irritation, dizziness, electrical leakage and radiation, may stem from a heightened awareness of the potential risks associated with electrical products, especially for wearable devices. These concerns can deter older adults from embracing wearable information and communications technology, implying that safety issue could be the potential barrier. Similarly, another study has identified safety factors, including concerns relate to radiation and the use of electricity [ 45 ]. Thus, to address this barrier, device designers should prioritize safety issues, reducing any safety-related risks. These considerations can help alleviate concerns and enhance user’s confidence. Another theme is the preference for human care over technology, with many participants believing that caregivers or family members provided more reliable support. One review study [ 30 ] emphasizes that companionship plays a crucial role in the context of having a source of support and presence in one's life. The preference for human care in taking care of older adults suggests that fall-detection devices should be viewed as complementary tools rather than replacements for caregivers. This aligns with concerns about the fear of losing social connections and experiencing loneliness [ 46 ]. In other words, while technology can aid in ensuring safety, the emotional and social aspects provided by human caregivers are irreplaceable. This is an important finding that emphasizing this perspective may decrease the barriers of using fall detection technology among older adults.
Issues related to device comfort and practicality were highlighted as significant factors influencing adoption as well. Concerns from stakeholders include device weight and physical discomfort. Obviously, user-friendly design is essential to mitigate these concerns [ 47 ]. Designers should aim to create lightweight, comfortable devices that seamlessly integrate into daily life, or design a fall detection technology that does not require older adults to wear. In addition, participants expressed a preference for traditional fall prevention tools, such as canes or emergency buttons, citing familiarity and trust in these established solutions. Several participants voiced the opinion that a cane is more beneficial than a fall detection device since a cane can provide support to older adults and reduce the risk of falls, whereas they believe that fall detection devices may not effectively prevent older adults from falling. This concept that the product is able to prevent falls is similar to fall prediction systems [ 48 ]. On the one hand, this factor may require fall detection technology to demonstrate its superiority over existing options or complement the characteristics of existing products. On the other hand, perception of inconvenience, unfamiliarity, and embarrassment were common attitudes among older adults [ 19 , 32 , 47 ]. In our study, some participants also stated that fall detection devices are troublesome. We suggest making fall detection devices easy to use by designing them to be simple and not bothersome.
The cost of fall detection devices emerged as a significant consideration for both older adults and their families. Affordability is a key factor in their decision-making process [ 22 , 27 , 30 , 32 , 47 ], highlighting the importance of exploring options for making these devices more accessible, such as through insurance coverage or subsidies. On the other hand, one study investigated the preferred specifications, perceived ease of use, and perceived usefulness of an automated fall detection device among older adults who rely on wheelchairs or scooters. It was noted that participants expressed a belief in the utility and user-friendliness of an automated fall detection device. The features include wireless charging, a wristwatch-like design, the option to change the emergency contact person in case of a fall, and the ability to deactivate notifications in case of false alarms [ 49 ]. In our study, participants emphasized the importance of comprehensive fall detection solutions, including remote notifications, real-time older adults’ status updates, and immediate assistance functions. It seems that the function of fall detection technology is oriented toward notifying the families, enabling them to assist immediately. Therefore, prioritizing the creation of devices that detect falls and provide added value through additional features is beneficial for enhancing overall safety and well-being.
Limitations
Although this study contributes to the field of fall detection technology, the study has several limitations. First, the sample of older adults comes from neurology outpatient. This limits the findings to this specific group and decreases their generalizability. Second, the findings of this study are based on the opinions and experiences of the respondents and may not be fully representative of all potential users of fall detection technology. The experiences and preferences of non-respondents remain unknown and might differ from those who participated in the study. In addition, the study involved respondents with varying levels of fall risk, as they suffered from different health conditions such as acute stroke, mild to moderate dementia, impaired cognitive function, and poor balance and gait. Third, as fall risk factors can significantly influence the perception and acceptance of fall detection technology, the results may not fully capture the nuances of specific subgroups within older population. The in-depth, face-to-face interviews were conducted in the outpatient area of the hospital. Although none of the interviewees discontinued the interviews due to privacy concerns, it is important to consider the potential influence of the interview setting. In addition, the outpatient waiting area in a hospital is an open and public space, which might have affected the responses of the interviewees. They may have been conscious of their surroundings and the presence of other individuals, possibly influencing the openness of their responses. Finally, the study focused on a specific population in Taiwan, and the findings may be influenced by cultural and regional factors unique to this context. Cultural differences and healthcare practices may lead to varying perspectives on fall detection technology in other regions or countries.
Conclusion and suggestions
In this study, we examined the factors influencing the adoption of wearable fall-detection devices among older adults and their caregivers. We identified several key considerations: concerns about potential health risks associated with these devices, the preference for human care over technology, the importance of device comfort and practicality, market alternatives, cost considerations, the attitude towards technology, and expectations of technology. Based on our evaluation framework, it is essential to consider safety, usability, affordability, and complementary to human care when developing fall detection products. In addition, meeting user expectations for comprehensive features like remote notifications and immediate assistance functions can further enhance adoption. Addressing these factors and challenges is expected to enhance the safety and quality of life for older adults, thereby relieving the burden of care.
Availability of data and materials
Data is provided within the manuscript.
Abbreviations
Information and communications technology
Mild Cognitive Impairment
Hypertension
Viswanathan A, Sudarsky L. Balance and gait problems in the elderly. Handb Clin Neurol. 2012;103:623–34.
Article PubMed Google Scholar
Institute of Medicine (US) Division of Health Promotion and Disease Prevention. Berg RL, Cassells JS, eds. The second fifty years: promoting health and preventing disability. Washington (DC): National Academies Press (US); 1992.
Centers for Disease Control and Prevention. Older adult fall prevention. https://www.cdc.gov/falls/data-research/facts-stats/?CDC_AAref_Val=https://www.cdc.gov/falls/facts.html . Accessed 22 Nov 2023.
Ambrose AF, Paul G, Hausdorff JM. Risk factors for falls among older adults: a review of the literature. Maturitas. 2013;75(1):51–61.
Moreland B, Kakara R, Henry A. Trends in nonfatal falls and fall-related injuries among adults aged ≥65 years - United States, 2012–2018. MMWR Morb Mortal Wkly Rep. 2020;69(27):875–81.
Article PubMed PubMed Central Google Scholar
Florence CS, Bergen G, Atherly A, Burns E, Stevens J, Drake C. Medical Costs of fatal and nonfatal falls in older adults. J Am Geriatr Soc. 2018;66(4):693–8.
Boongird C, Ross R. Views and expectations of community-dwelling thai elderly in reporting falls to their primary care physicians. J Appl Gerontol. 2017;36(4):480–98.
Hartholt KA, van Beeck EF, Polinder S, van der Velde N, van Lieshout EM, Panneman MJ, van der Cammen TJ, Patka P. Societal consequences of falls in the older population: injuries, healthcare costs, and long-term reduced quality of life. J Trauma. 2011;71(3):748–53.
PubMed Google Scholar
Dhar M, Kaeley N, Mahala P, Saxena V, Pathania M. The prevalence and associated risk factors of fear of fall in the elderly: a hospital-based, cross-sectional study. Cureus. 2022;14(3):e23479.
PubMed PubMed Central Google Scholar
Bayen E, Nickels S, Xiong G, Jacquemot J, Subramaniam R, Agrawal P, Hemraj R, Bayen A, Miller BL, Netscher G. Reduction of Time on the ground related to real-time video detection of falls in memory care facilities: observational study. J Med Internet Res. 2021;23(6):e17551.
Wild D, Nayak US, Isaacs B. How dangerous are falls in old people at home?. Br Med J (Clin Res Ed). 1981;282(6260):266–8.
Article PubMed CAS Google Scholar
Lapierre N, Neubauer N, Miguel-Cruz A, Rios Rincon A, Liu L, Rousseau J. The state of knowledge on technologies and their use for fall detection: a scoping review [published correction appears in int j med inform. 2018 aug; 116:9]. Int J Med Inform. 2018;111:58–71.
Mrozek D, Koczur A, Małysiak-Mrozek B. Fall detection in older adults with mobile IoT devices and machine learning in the cloud and on the edge. Inf Sci. 2020;537:132–47.
Article Google Scholar
Tanwar R, Nandal N, Zamani M, Manaf AA. Pathway of trends and technologies in fall detection: a systematic review. Healthcare (Basel). 2022;10(1):172.
Newaz NT, Hanada E. The methods of fall detection: a literature review. Sensors (Basel, Switzerland). 2023;23(11): 5212. https://doi.org/10.3390/s23115212 .
Ram S. A model of innovation resistance. Adv Consum Res. 1987;14(1):208–12.
Google Scholar
Ram S, Sheth JN. Consumer resistance to innovations: the marketing problem and its solutions. J Consum Mark. 1989;6(2):5–14.
Fischer SH, David D, Crotty BH, Dierks M, Safran C. Acceptance and use of health information technology by community-dwelling elders. Int J Med Inform. 2014;83(9):624–35.
Demiris G, Chaudhuri S, Thompson HJ. Older adults’ experience with a novel fall detection device. Telemed J E Health. 2016;22(9):726–32.
Guzman-Parra J, Barnestein-Fonseca P, Guerrero-Pertiñez G, Anderberg P, Jimenez-Fernandez L, Valero-Moreno E, Goodman-Casanova JM, Cuesta-Vargas A, Garolera M, Quintana M, García-Betances RI, Lemmens E, Sanmartin Berglund J, Mayoral-Cleries F. Attitudes and use of information and communication technologies in older adults with mild cognitive impairment or early stages of dementia and their caregivers: cross-sectional study. J Med Internet Res. 2020;22(6):e17253.
Wilson J, Heinsch M, Betts D, Booth D, Kay-Lambkin F. Barriers and facilitators to the use of e-health by older adults: a scoping review. BMC Public Health. 2021;21(1):1556–1556.
Zaman SB, Khan RK, Evans RG, Thrift AG, Maddison R, Islam SMS. Exploring barriers to and enablers of the adoption of information and communication technology for the care of older adults with chronic diseases: scoping review. JMIR Aging. 2022;5(1):e25251.
Saracchini R, Catalina C, Bordoni L. A mobile augmented reality assistive technology for the elderly. Comunicar. 2015;23:23.
Mercer K, Giangregorio L, Schneider E, Chilana P, Li M, Grindrod K. Acceptance of commercially available wearable activity trackers among adults aged over 50 and with chronic illness: a mixed-methods evaluation. JMIR Mhealth Uhealth. 2016;4(1):e7.
Jain SR, Sui Y, Ng CH, Chen ZX, Goh LH, Shorey S. Patients’ and healthcare professionals’ perspectives towards technology-assisted diabetes self-management education. A qualitative systematic review. PLoS One. 2020;15(8):e0237647.
Article PubMed PubMed Central CAS Google Scholar
O’Brien J, Mason A, Cassarino M, Chan J, Setti A. Older women’s experiences of a community-led walking programme using activity trackers. Int J Environ Res Public Health. 2021;18(18):9818.
Kononova A, Li L, Kamp K, Bowen M, Rikard RV, Cotten S, Peng W. The use of wearable activity trackers among older adults: focus group study of tracker perceptions, motivators, and barriers in the maintenance stage of behavior change. JMIR Mhealth Uhealth. 2019;7(4):e9832.
Jiwani R, Dennis B, Bess C, Monk S, Meyer K, Wang J, Espinoza S. Assessing acceptability and patient experience of a behavioral lifestyle intervention using fitbit technology in older adults to manage type 2 diabetes amid COVID-19 pandemic: a focus group study. Geriatr Nurs. 2021;42(1):57–64.
Ehn M, Eriksson LC, Åkerberg N, Johansson AC. Activity monitors as support for older persons’ physical activity in daily life: qualitative study of the users’ experiences. JMIR Mhealth Uhealth. 2018;6(2):e34.
Tsertsidis A, Kolkowska E, Hedström K. Factors influencing seniors’ acceptance of technology for ageing in place in the post-implementation stage: a literature review. Int J Med Inform. 2019;129:324–33.
Moore K, O’Shea E, Kenny L, Barton J, Tedesco S, Sica M, Crowe C, Alamäki A, Condell J, Nordström A, Timmons S. Older adults’ experiences with using wearable devices: qualitative systematic review and meta-synthesis. JMIR Mhealth Uhealth. 2021;9(6):e23832.
Chiu CJ, Liu CW. Understanding older adult’s technology adoption and withdrawal for elderly care and education: mixed method analysis from national survey. J Med Internet Res. 2017;19(11): e374.
Peek ST, Luijkx KG, Rijnaard MD, Nieboer ME, van der Voort CS, Aarts S, van Hoof J, Vrijhoef HJ, Wouters EJ. Older adults’ reasons for using technology while aging in place. Gerontology. 2016;62(2):226–37.
Perotti L, Stamm O, Mesletzky L, Vorwerg S, Fournelle M, Müller-Werdan U. Needs and attitudes of older chronic back pain patients towards a wearable for ultrasound biofeedback during stabilization exercises: a qualitative analysis. Int J Environ Res Public Health. 2023;20(6):4927.
Abouzahra M, Ghasemaghaei M. The antecedents and results of seniors’ use of activity tracking wearable devices. Health Policy Technol. 2020;9(2):213–7.
Finkelstein R, Wu Y, Brennan-Ing M. Older adults’ experiences with using information and communication technology and tech support services in New York City: findings and recommendations for post-pandemic digital pedagogy for older adults. Front Psychol. 2023;14:1129512.
Dickinson A, Horton K, Machen I, Bunn F, Cove J, Jain D, Maddex T. The role of health professionals in promoting the uptake of fall prevention interventions: a qualitative study of older people’s views. Age Ageing. 2011;40(6):724–30.
McIntosh MJ, Morse JM. Situating and constructing diversity in semi-structured interviews. Glob Qual Nurs Res. 2015;2:2333393615597674.
Graneheim UH, Lundman B. Qualitative content analysis in nursing research: concepts, procedures and measures to achieve trustworthiness. Nurse Educ Today. 2004;24(2):105–12.
Hsu YH, Lee TH, Chung KP, Tung YC. Determining the factors influencing the selection of post-acute care models by patients and their families: a qualitative content analysis. BMC Geriatr. 2023;23(1):179.
Elo S, Kyngäs H. The qualitative content analysis process. J Adv Nurs. 2008;62(1):107–15.
Morse JM, Black C, Oberle K, Donahue P. A prospective study to identify the fall-prone patient. Soc Sci Med. 1989;28(1):81–6.
Rockwood K, Theou O. Using the clinical frailty scale in allocating scarce health care resources. Can Geriatr J. 2020;23(3):210.
Mahoney FI, Barthel DW. Functional evaluation: the barthel index. Md State Med J. 1965;14:61–5.
PubMed CAS Google Scholar
Felber NA, Lipworth W, Tian YJA, Roulet Schwab D, Wangmo T. Informing existing technology acceptance models: a qualitative study with older persons and caregivers. Eur J Ageing. 2024;21(1):12.
Tian YJA, Felber NA, Pageau F, Schwab DR, Wangmo T. Benefits and barriers associated with the use of smart home health technologies in the care of older persons: a systematic review. BMC geriatr. 2024;24(1):152.
Puri A, Kim B, Nguyen O, Stolee P, Tung J, Lee J. User acceptance of wrist-worn activity trackers among community-dwelling older adults: mixed method study. JMIR Mhealth Uhealth. 2017;5(11):e173.
El-Bendary N, Tan Q, Pivot F, Lam A. Fall detection and prevention for the elderly: a review of trends and challenges. Int J Smart Sensing Intellig Syst. 2013;6:1230–66.
Rice LA, Fliflet A, Frechette M, Brokenshire R, Abou L, Presti P, Mahajan H, Sosnoff J, Rogers WA. Insights on an automated fall detection device designed for older adult wheelchair and scooter users: a qualitative study. Disabil Health J. 2022;15(1S):101207.
Download references
Acknowledgements
This research was made possible by the support and assistance of a number of people whom we would like to thank. We are very grateful to the anonymous referees for their valuable comments and constructive suggestions on interview and coding. We would like to thank all the respondents for their valuable opinions. This research was supported by the Ministry of Technology and Science under grant number NSTC 112-2628-E-006-008-MY3, NSTC 112-2627-M-006 -005, and the Medical Device Innovation Center (MDIC), National Cheng Kung University(NCKU) from the Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MoE) in Taiwan. This research was approved by the local Institutional Review Board of NCKUH (IRB Approval No. A-ER-110-211).
This research was supported by the National Science Council under grant number NSTC 112–2628-E-006–008-MY3 and NSTC 112–2627-M-006-005.
Author information
Authors and affiliations.
Department of Biomedical Engineering, National Cheng Kung University, Tainan, Taiwan, ROC
Hsin-Hsiung Huang, Ming-Hao Chang & Peng-Ting Chen
Medical Device Innovation Center, National Cheng Kung University, No.138, Shengli Rd., North District, Tainan City, 704, Taiwan, ROC
Peng-Ting Chen
Department of Electrical Engineering, National Cheng Kung University, Tainan, Taiwan, ROC
Chih-Lung Lin
Department of Neurology, National Cheng Kung University Hospital, Tainan, Taiwan, ROC
Pi-Shan Sung
Department of Industrial Design, National Cheng Kung University, Tainan, Taiwan, ROC
Chien-Hsu Chen
Institute of Gerontology, National Cheng Kung University, Tainan, Taiwan, ROC
Sheng-Yu Fan
You can also search for this author in PubMed Google Scholar
Contributions
Hsin-Hsiung Huang contributed significantly as the main interviewer, played a key role in coding, and contributed to the conception of the article. Ming-Hao Chang participated in designing interview questions, coding, and ensuring the quality of language in the article. Peng-Ting Chen assisted in conceptualizing research directions, overseeing the interview, coding, and the writing process, and shaped the article's concept. Chih-Lung Lin, Pi-Shan Sung, Chien-Hsu Chen, and Sheng-Yu Fan assisted in conceptualizing research directions.
Authors' information
Hsin-Hsiung Huang is pursuing his Ph.D. degree in the Department of Biomedical Engineering from National Cheng Kung University, Taiwan. His major research interests fall in medical device commercialization in the elderly market.
Ming-Hao Chang is pursuing his Master’s degree in the Department of Biomedical Engineering from National Cheng Kung University, Taiwan. His major research interests fall in medical device commercialization, especially in startups.
Professor Peng-Ting Chen received her Ph.D. in Technology Management from the University of National Chiao-Tung University, Taiwan. She is a professor in the Department of Biomedical Engineering, at National Cheng Kung University, Taiwan. Her current research interests include biomedical device-related business planning, strategies, and policies.
Corresponding author
Correspondence to Peng-Ting Chen .
Ethics declarations
Ethics approval and consent to participate.
The study was approved by the Institutional Review Board of NCKUH (IRB number: A-ER-110–211) before commencement. Informed consent was obtained from all subjects.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary material 1., rights and permissions.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Huang, HH., Chang, MH., Chen, PT. et al. Exploring factors affecting the acceptance of fall detection technology among older adults and their families: a content analysis. BMC Geriatr 24 , 694 (2024). https://doi.org/10.1186/s12877-024-05262-0
Download citation
Received : 06 March 2024
Accepted : 30 July 2024
Published : 20 August 2024
DOI : https://doi.org/10.1186/s12877-024-05262-0
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Information and Communications Technology (ICT)
- Innovation resistance
- Content analysis
BMC Geriatrics
ISSN: 1471-2318
- General enquiries: [email protected]

IMAGES
COMMENTS
In other words, qualitative isn't just limited to text-based data. So, how's that different from quantitative data, you ask? Simply put, qualitative research focuses on words, descriptions, concepts or ideas - while quantitative research focuses on numbers and statistics.
Qualitative research is a type of research methodology that focuses on exploring and understanding people's beliefs, attitudes, behaviors, and experiences through the collection and analysis of non-numerical data. It seeks to answer research questions through the examination of subjective data, such as interviews, focus groups, observations ...
Qualitative data analysis is a systematic process of examining non-numerical data to extract meaning, patterns, and insights. In contrast to quantitative analysis, which focuses on numbers and statistical metrics, the qualitative study focuses on the qualitative aspects of data, such as text, images, audio, and videos.
Qualitative research is the opposite of quantitative research, which involves collecting and analyzing numerical data for statistical analysis. Qualitative research is commonly used in the humanities and social sciences, in subjects such as anthropology, sociology, education, health sciences, history, etc.
This question is particularly relevant to researchers new to the field and practice of qualitative research and instructors and mentors who regularly introduce students to qualitative research practices. In this article, we seek to offer what we view as a useful starting point for learning how to do qualitative analysis.
Mapping the Field Data analysis is the central step in qualitative research. Whatever the data are, it is their analysis that, in a decisive way, forms the outcomes of the research. Sometimes, data collection is limited to recording and docu-menting naturally occurring phenomena, for example by recording interactions. Then qualitative research is concentrated on ana-lysing such recordings ...
Qualitative Data Analysis: Step-by-Step Guide (Manual vs. Automatic) Feedback Analysis Sentiment Analysis AI & NLP When we conduct qualitative methods of research, need to explain changes in metrics or understand people's opinions, we always turn to qualitative data. Qualitative data is typically generated through: Interview transcripts Surveys with open-ended questions Contact center ...
This paper aims to provide an overview of the use and assessment of qualitative research methods in the health sciences. Qualitative research can be defined as the study of the nature of phenomena and is especially appropriate for answering questions ...
Qualitative research designs are described after types of qualitative data and methods of analysis are described. The type of data collected and the approach to its analysis are more relevant to a researcher's compelling argument and sound conclusion than a category name placed on a general approach to data collection.
Aim The aim of this paper is to provide an overview of qualitative research methods, including hands-on information on how they can be used, reported and assessed. This article is intended for beginning qualitative researchers in the health sciences as well as experienced quantitative researchers who wish to broaden their understanding of qualitative research.
Qualitative research methods refer to techniques of investigation that rely on nonstatistical and nonnumerical methods of data collection, analysis, and evidence production. Qualitative research techniques provide a lens for learning about nonquantifiable phenomena such as people's experiences, languages, histories, and cultures.
Abstract Thematic analysis, often called Qualitative Content Analysis (QCA) in Europe, is one of the most commonly used methods for analyzing qualitative data. This paper presents the basics of this systematic method of qualitative data analysis, highlights its key characteristics, and describes a typical workflow. The aim is to present the main characteristics and to give a simple example of ...
Qualitative data analysis Analyzing qualitative data is the next step after you have completed the use of qualitative data collection methods. The qualitative analysis process aims to identify themes and patterns that emerge across the data. Qualitative data analysis focuses on organizing data into meaningful knowledge.
The purpose of this article is to provide an overview of some of the principles of data analysis used in qualitative research such as coding, interrater reliability, and thematic analysis.
Abstract The Oxford Handbook of Qualitative Research, second edition, presents a comprehensive retrospective and prospective review of the field of qualitative research. Original, accessible chapters written by interdisciplinary leaders in the field make this a critical reference work. Filled with robust examples from real-world research; ample discussion of the historical, theoretical, and ...
5 qualitative data analysis methods explained Qualitative data analysis ( QDA) is the process of organizing, analyzing, and interpreting qualitative research data—non-numeric, conceptual information, and user feedback—to capture themes and patterns, answer research questions, and identify actions to improve your product or website.
Qualitative data refers to non-numeric information such as interview transcripts, notes, video and audio recordings, images and text documents. Qualitative data analysis can be divided into the following five categories: 1. Content analysis. This refers to the process of categorizing verbal or ...
Qualitative research is a method of inquiry used in various disciplines, including social sciences, education, and health, to explore and understand human behavior, experiences, and social phenomena. It focuses on collecting non-numerical data, such as words, images, or objects, to gain in-depth insights into people's thoughts, feelings, motivations, and perspectives.
Qualitative research gathers participants' experiences, perceptions, and behavior. It answers the hows and whys instead of how many or how much. It could be structured as a standalone study, purely relying on qualitative data, or part of mixed-methods research that combines qualitative and quantitative data.
1 Chapter 8 1 Qualitative Data Analysis and Interpretatio n: Systematic Search for Meaning Patrick Ngulube 8.1 Introduction In this chapter, we will discuss the analysis and interpretation of ...
When qualitative data analysis is done right, transparency and rigor can be maintained throughout the process, from the initial selection of research questions and gathering of raw data to final analysis techniques. Active engagement is a crucial aspect of qualitative feedback interpretation.
This chapter provides an overview of selected qualitative data analysis strategies with a particular focus on codes and coding. Preparatory strategies for a qualitative research study and data management are first outlined. Six coding methods are then profiled using comparable interview data: process coding, in vivo coding, descriptive coding ...
Thematic Analysis in Qualitative Research. Thematic analysis is a popular technique in qualitative research that enables researchers to identify, analyze, and report patterns within data. This method provides an accessible framework for understanding the insights derived from participant responses or textual materials.
Qualitative research methods. Because of the nature of qualitative data (complex, detailed information), the research methods used to collect it are more involved. Qualitative researchers might do the following to collect data: Conduct interviews to learn about subjective experiences; Host focus groups to gather feedback and personal accounts
This book presents a comprehensive guide to interpretative phenomenological analysis (IPA) which is an increasingly popular approach to qualitative inquiry taught to undergraduate and postgraduate students today. The first chapter outlines the theoretical foundations for IPA. It discusses phenomenology, hermeneutics, and idiography and how they have been taken up by IPA. The next four chapters ...
In an earlier paper, 1 we presented an introduction to using qualitative research methods in pharmacy practice. In this article, we review some principles of the collection, analysis, and management of qualitative data to help pharmacists interested in doing research in their practice to continue their learning in this area.
One-on-one qualitative interviews following a semistructured discussion guide informed by the TDF were conducted. Thirty people with type 2 diabetes in Canada were interviewed, with representation from across the country, of both sexes (47% female), of people with various diabetes durations (mean 12.9 ± 7.9 years), with different types of medication plans (n = 15 on polypharmacy), and with ...
Background: Large language models including GPT-4 (OpenAI) have opened new avenues in health care and qualitative research. Traditional qualitative methods are time-consuming and require expertise to capture nuance. Although large language models have demonstrated enhanced contextual understanding and inferencing compared with traditional natural language processing, their performance in ...
Design. The intervention, methods and results of the Stop Cancer PAIN trial have been described in previous open-access articles [17, 18].The sub-study used a qualitative approach with pragmatic orientation to enable in-depth exploration of factors influencing success from the perspectives of clinicians at each participating centre [].Clinician views canvassed at interview were considered the ...
The method employed a qualitative approach, utilizing semi-structured interviews with 30 older adults and 29 families, focusing on their perspectives and expectations of fall detection technology. ... Graneheim UH, Lundman B. Qualitative content analysis in nursing research: concepts, procedures and measures to achieve trustworthiness. Nurse ...